Ammonia Inhalent
Active ingredient (in each ampule)
Ammonia 15%
Ammonia Inhalent
Purpose
Respiratory stimulant
Ammonia Inhalent
Uses
- to prevent or treat fainting
Ammonia Inhalent
Warnings
For external use only
Do not use
- if you have breathing problems such as asthma or emphysema
Stop use and ask a doctor if
Keep out of reach of children
- If swallowed get medical help or contact a Poison Control Center right away.
Ammonia Inhalent
Directions
- hold inhalant away from face and crush ampoule between thumb and forefinger at position indicated on sleeve.
- hold near nostrils for inhalation of volatile vapor
Ammonia Inhalent
Other information
- store at room temperature away from light
Ammonia Inhalent
Inactive ingredients
alcohol USP, FD&C red #40, lavender oil, lemon oil fcc, nutmeg oil, purified water
Ammonia Inhalent
Questions
1-800-430-5490
Eyewash
Active ingredient
Sterile Water 99%
Eyewash
Uses
- for flushing the eye to remove loose foreign material, air pollutants or chlorinated water
Eyewash
Warnings
For external use only Obtain immediate medical treatment for all open wounds in or near eyes. To avoid contamination, do not touch tip of container to any surface. Do not reuse. Once opened, discard.
Do not use
- if solution changes color or becomes cloudy
- if you have open wounds in or near the eyes, get medical help right away.
Stop use and ask a doctor if
- you experience eye pain
- changes in vision
- continued redness or irritation of the eye
- condition worsens or persists
Keep out of reach of children
- If swallowed, get medical help or contact a Poison Control Center right away.
Eyewash
Directions
- remove contacts before using
- twist top to remove
- flush the affected area as needed
- control rate of flow by pressure on the bottle
- if necessary, continue flushing with emergency eyewash or shower
Eyewash
Inactive ingredients
sodium chloride, sodium phosphate dibasic, sodium phosphate monobasic
Eyewash
Questions
1-800-430-5490
PVP
Active Ingredient
Povidone-iodine solution USP, 10% (equivalent to 1% titratable iodine)
PVP
Purpose
First aid antiseptic
PVP
Uses
- first aid to help prevent the risk of infection in minor cuts, scrapes, and burns
PVP
Warnings
For external use only
Do not use
- in the eyes
- over large areas of the body
- on individuals who are allergic or sensitive to iodine
Ask a doctor before use if you have
- deep or puncture wounds,
- animal bites
- serious burns
When using this product
- do not use longer than one wek unless directed by a doctor
Stop use and ask a doctor if
- conditions persists or gets worse
- irritation and redness develops
Keep out of reach of children.
- If swallowed, get medical help or contact a Poison Control Center right away
PVP
Directions
Reverse cardboard sleeve, then crush at dot between thumb and forefinger. Allow solution to saturate tip and apply solution to injury.
- clean affected area
- apply to affected area 1 to 3 times daily
- may be covered with a sterile bandage
- discard swab after single use
PVP
Other informatiion
- store at room temperature away from light
- keep from freezing or excessive heat
- do not use if package is torn or open
PVP
Inactive ingredient
citric acid, disodium phosphate,nonoxynol-9, sodium hydroxide, water
PVP
Questions
1-800-430-5490
Alcohol
Active ingredient
Isproppyl alcohol 70%
Alcohol
Purpose
First aid antiseptic
Alcohol
Uses
- first aid to help prevent infection in minor cuts, scrapes, and burns
Alcohol
Warnings
For external use only
Flammable, keep away from fire and flame
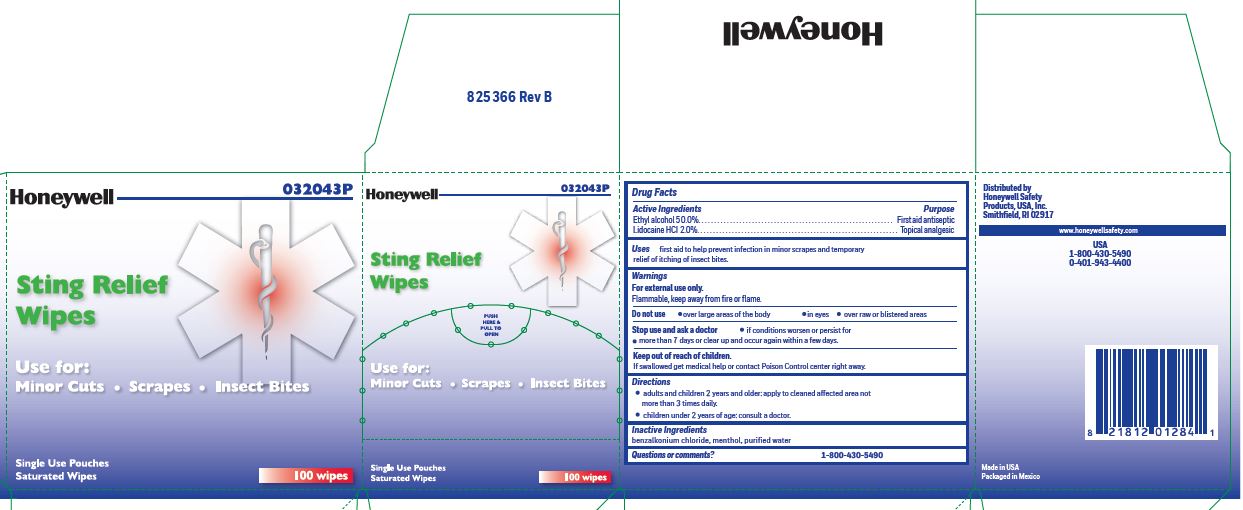
Sting Relief
Active ingredient (in each wipe)
Ethyl alcohol 50.0%
Lidocaine HCl 2.0%
Sting Relief
Purpose
Antiseptic
Topical pain relief
Sting Relief
Uses
- prevent infection in minor scrapes, and temporary relief of itching of insect bites
Sting Relief
Warnings
For external use only
Flammable, keep away from open fire or flame
Do not use
- over large areas of the body
- in eyes
- over raw or blistered areas
Stop use and ask a doctor
- if conditions worsen or persist for more than 7 days or clear up and occur again within a few days
Keep out of reach of children
If swallowed, get medical help or contact a Poison Control Center right away.
Sting Relief
Directions
- adults and children 2 years and older: Apply to cleaned affected area not more than 3 times daily.
- children under 2 years of age: consult a doctor.
Sting Relief
Inactive ingredients
benzalkonium chloride, menthol, and purified water
Sting Relief
Questions or comments?
1-800-430-5490
4190
010885-4514 Kit Contents
1 AMMONIA INHALANTS 10 PER
1 TRIANGULAR BDG, NON-STERILE
1 GAUZE COMPRESS, 1728 SQ IN 1
1 ADHESIVE TPE 1"X2-1/2 YD 2 PER
1 ADH BDG W/NOADHR PAD,1X3 32PER
1 BANDAGE COMP, 4" OFFSET, 1 PER
1 1 OZ EYE WASH W/PADS & STRIPS
1 PVP IODINE WIPES 10 PER
1 ELASTIC BANDAGE 3" X 4.5YD
1 MICROSHIELD W/VNL GLV/ALCL
1 NORTH RESPONSE REFILL/KIT
1 SCISSOR BDGE 4" RED PLS HDL
LBL STOCK 6-3/8"X4"
1 LBL STOCK 3"x1-7/8"
1 KIT STL 24 UN WHITE 01
1 LABL INSTR 24 & 36 UNIT KITS
1 STING RELIEF 10
Ammonia Inahalent
Principal Display Panel

Eyewash
Principal Display Panel

PVP
Principal Display Panel

Sting Relief
Principal Display Panel
4190 Kit Label
010885-4514





