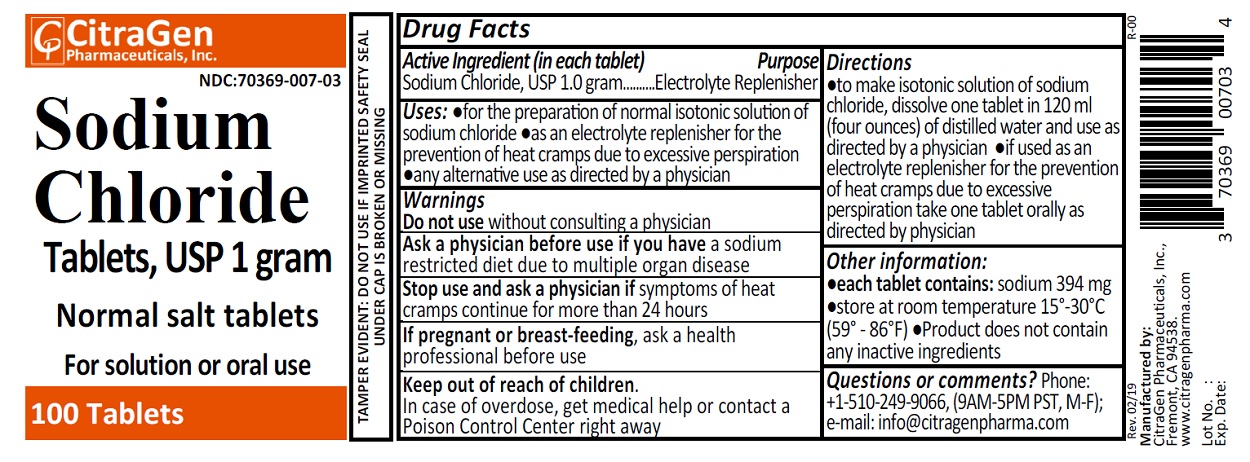
Label: SODIUM CHLORIDE NORMAL SALT- sodium chloride tablet
- NDC Code(s): 70369-007-02, 70369-007-03, 70369-007-07
- Packager: Citragen Pharmaceuticals Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 22, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Purpose
- Uses
- Warnings
-
Directions
- to make isontonic solution of sodium chloride, dissolve one tablet in 120 ml (four ounces) of distilled water and use as directed by a physician.
- if used as an electrolyte replenisher for the prevention of heat cramps due to excessive perspiration take one tablet orally as directed by your physician.
- Other information:
- Questions or comments?
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SODIUM CHLORIDE NORMAL SALT
sodium chloride tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70369-007 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) (CHLORIDE ION - UNII:Q32ZN48698) SODIUM CHLORIDE 1 g Product Characteristics Color white Score no score Shape ROUND Size 12mm Flavor Imprint Code CG007 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70369-007-02 500 in 1 BOTTLE; Type 0: Not a Combination Product 03/22/2019 2 NDC:70369-007-03 100 in 1 BOTTLE; Type 0: Not a Combination Product 03/22/2019 3 NDC:70369-007-07 300 in 1 BOTTLE; Type 0: Not a Combination Product 03/09/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part349 03/22/2019 Labeler - Citragen Pharmaceuticals Inc (024949457) Registrant - Citragen Pharmaceuticals Inc (024949457) Establishment Name Address ID/FEI Business Operations Citragen Pharmaceuticals Inc 024949457 manufacture(70369-007)