Label: LATANOPROST solution/ drops
- NDC Code(s): 68462-944-03
- Packager: GLENMARK PHARMACEUTICALS INC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated September 16, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use LATANOPROST OPHTHALMIC SOLUTION safely and effectively. See full prescribing information for LATANOPROST OPHTHALMIC SOLUTION ...
-
Table of ContentsTable of Contents
-
1 INDICATIONS AND USAGELatanoprost ophthalmic solution is indicated for the reduction of elevated intraocular pressure (IOP) in patients with open-angle glaucoma or ocular hypertension.
-
2 DOSAGE AND ADMINISTRATIONThe recommended dosage is one drop in the affected eye(s) once daily in the evening. If one dose is missed, treatment should continue with the next dose as normal. The dosage of latanoprost ...
-
3 DOSAGE FORMS AND STRENGTHSOphthalmic solution containing latanoprost 50 mcg/mL (0.005%).
-
4 CONTRAINDICATIONSKnown hypersensitivity to latanoprost, benzalkonium chloride, or any other ingredients in this product.
-
5 WARNINGS AND PRECAUTIONS5.1 Pigmentation - Latanoprost ophthalmic solution has been reported to cause changes to pigmented tissues. The most frequently reported changes have been increased pigmentation of the iris ...
-
6 ADVERSE REACTIONSThe following adverse reactions were reported in postmarketing experience and are discussed in greater detail in other sections of the label: • Iris pigmentation changes [see Warnings and ...
-
8 USE IN SPECIFIC POPULATIONS8.1 Pregnancy - Risk Summary - There are no adequate and well-controlled studies of latanoprost ophthalmic solution administration in pregnant women to inform drug-associated ...
-
10 OVERDOSAGEIV infusion of up to 3 mcg/kg of latanoprost in healthy volunteers produced mean plasma concentrations 200 times higher than during clinical treatment with latanoprost ophthalmic solution ...
-
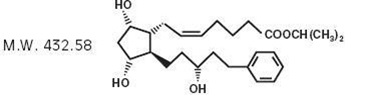
11 DESCRIPTIONLatanoprost is a prostaglandin F2α analogue. Its chemical name is isopropyl-(Z)-7[(1R,2R,3R,5S)3,5 ...
-
12 CLINICAL PHARMACOLOGY12.1 Mechanism of Action - Latanoprost is a prostaglandin F2α analogue that is believed to reduce the IOP by increasing the outflow of aqueous humor. Studies in animals and man suggest that the ...
-
13 NONCLINICAL TOXICOLOGY13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility - Carcinogenesis - Latanoprost was not carcinogenic in either mice or rats when administered by oral gavage at doses of up to 170 ...
-
14 CLINICAL STUDIES14.1 Elevated Baseline IOP - Patients with mean baseline IOP of 24 – 25 mmHg who were treated for 6 months in multi-center, randomized, controlled trials demonstrated 6 – 8 mmHg reductions in ...
-
16 HOW SUPPLIED/STORAGE AND HANDLINGLatanoprost ophthalmic solution is a clear, isotonic, buffered, preserved colorless solution of latanoprost 50 mcg/mL (0.005%) ...
-
17 PATIENT COUNSELING INFORMATIONPotential for Pigmentation - Advise patients about the potential for increased brown pigmentation of the iris, which may be permanent ...
-
PACKAGE LABEL.PRINCIPAL DISPLAY PANELBottle Label: NDC 68462-944-03 Rx Only - Latanoprost - Ophthalmic Solution - 0.005% 125 mcg/2.5 mL - STERILE For eye use only - Carton Label: NDC 68462-944-03 Rx ...
-
INGREDIENTS AND APPEARANCEProduct Information