Label: ALLERGY RELIEF D- loratadine, pseudoephedrine sulfate tablet, film coated, extended release
- NDC Code(s): 56062-555-46, 56062-555-60
- Packager: Publix Super Markets Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated October 19, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
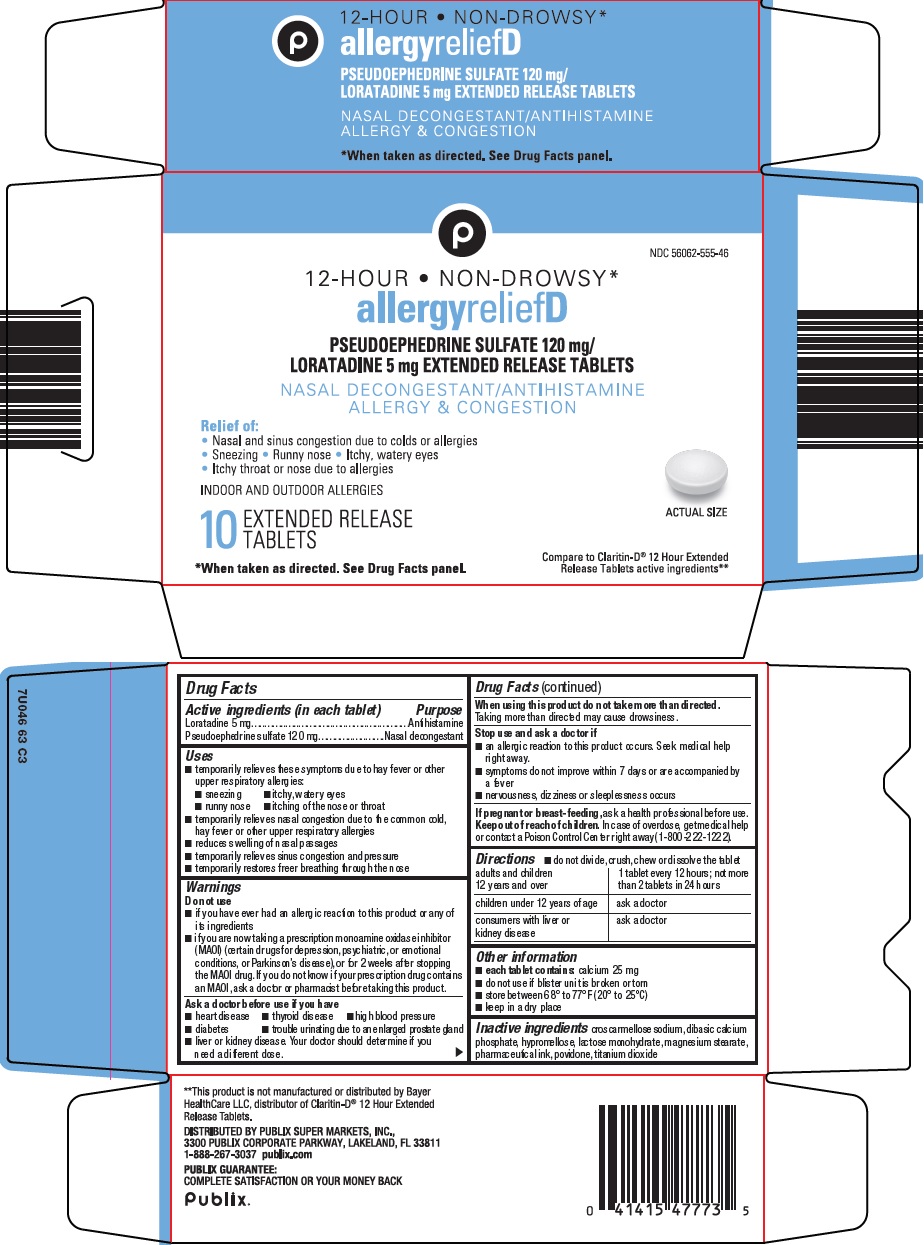
- Active ingredients (in each tablet)
- Purpose
-
Uses
- •
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- •
- sneezing
- •
- itchy, watery eyes
- •
- runny nose
- •
- itching of the nose or throat
- •
- temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies
- •
- reduces swelling of nasal passages
- •
- temporarily relieves sinus congestion and pressure
- •
- temporarily restores freer breathing through the nose
-
Warnings
Do not use
- •
- if you have ever had an allergic reaction to this product or any of its ingredients
- •
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- •
- heart disease
- •
- thyroid disease
- •
- high blood pressure
- •
- diabetes
- •
- trouble urinating due to an enlarged prostate gland
- •
- liver or kidney disease. Your doctor should determine if you need a different dose.
When using this product do not take more than directed.
Taking more than directed may cause drowsiness.
- Directions
- Other information
- Inactive ingredients
-
Package/Label Principal Display Panel
12-HOUR – NON-DROWSY*
allergy relief D
PSEUDOEPHEDRINE SULFATE 120 mg/LORATADINE 5 mg
EXTENDED RELEASE TABLETS
NASAL DECONGESTANT/ANTIHISTIME
ALLERGY & CONGESTION
Relief of:
Nasal and sinus congestion due to colds or allergies
Sneezing - Runny nose - Itchy, watery eyes
Itchy throat or nose due to allergies
INDOOR AND OUTDOOR ALLERGIES
10 EXTENDED RELEASE TABLETS
ACTUAL SIZE
*When taken as directed. See Drug Facts Panel.
Compare to Claritin-D® 12 hour Extended Release Tablets active ingredients
-
INGREDIENTS AND APPEARANCE
ALLERGY RELIEF D
loratadine, pseudoephedrine sulfate tablet, film coated, extended releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:56062-555 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LORATADINE (UNII: 7AJO3BO7QN) (LORATADINE - UNII:7AJO3BO7QN) LORATADINE 5 mg PSEUDOEPHEDRINE SULFATE (UNII: Y9DL7QPE6B) (PSEUDOEPHEDRINE - UNII:7CUC9DDI9F) PSEUDOEPHEDRINE SULFATE 120 mg Inactive Ingredients Ingredient Name Strength CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) ANHYDROUS DIBASIC CALCIUM PHOSPHATE (UNII: L11K75P92J) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color WHITE (to off-white) Score no score Shape ROUND Size 12mm Flavor Imprint Code 7U0 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:56062-555-46 10 in 1 CARTON 04/23/2018 1 1 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC:56062-555-60 20 in 1 CARTON 04/23/2018 2 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA076050 04/23/2018 Labeler - Publix Super Markets Inc (006922009)