Label: EMPLICITI- elotuzumab injection, powder, lyophilized, for solution
- NDC Code(s): 0003-2291-11, 0003-4522-11
- Packager: E.R. Squibb & Sons, L.L.C.
- Category: HUMAN PRESCRIPTION DRUG LABEL
Drug Label Information
Updated March 22, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use EMPLICITI safely and effectively. See full prescribing information for EMPLICITI.
EMPLICITI® (elotuzumab) for injection, for intravenous use
Initial U.S. Approval: 2015INDICATIONS AND USAGE
EMPLICITI is a SLAMF7-directed immunostimulatory antibody indicated in
- •
- combination with lenalidomide and dexamethasone for the treatment of adult patients with multiple myeloma who have received one to three prior therapies. (1)
- •
- combination with pomalidomide and dexamethasone for the treatment of adult patients with multiple myeloma who have received at least two prior therapies including lenalidomide and a proteasome inhibitor. (1)
DOSAGE AND ADMINISTRATION
- •
- With lenalidomide and dexamethasone: 10 mg/kg administered intravenously every week for the first two cycles and every 2 weeks thereafter until disease progression or unacceptable toxicity. (2.1)
- •
- With pomalidomide and dexamethasone: 10 mg/kg administered intravenously every week for the first two cycles and 20 mg/kg every 4 weeks thereafter until disease progression or unacceptable toxicity. (2.2)
- •
- Premedicate with dexamethasone, diphenhydramine, ranitidine and acetaminophen. (2.3)
DOSAGE FORMS AND STRENGTHS
For Injection: 300 mg or 400 mg lyophilized powder in a single-dose vial for reconstitution. (3)
CONTRAINDICATIONS
None (4)
WARNINGS AND PRECAUTIONS
- •
- Infusion reactions: Premedication is required. Interrupt EMPLICITI for Grade 2 or higher and permanently discontinue for severe infusion reaction. (2.3, 2.4, 5.1)
- •
- Infections: Monitor for fever and other signs of infection and treat promptly. (5.2)
- •
- Second Primary Malignancies (SPM): Higher incidences of SPM were observed in a controlled clinical trial of patients with multiple myeloma receiving EMPLICITI. (5.3)
- •
- Hepatotoxicity: Monitor liver function and stop EMPLICITI if hepatotoxicity is suspected. (5.4)
- •
- Interference with determination of complete response: EMPLICITI can interfere with assays used to monitor M-protein. This interference can impact the determination of complete response. (5.5)
ADVERSE REACTIONS
Most common adverse reactions (20% or higher)
- •
- with lenalidomide and dexamethasone are fatigue, diarrhea, pyrexia, constipation, cough, peripheral neuropathy, nasopharyngitis, upper respiratory tract infection, decreased appetite, pneumonia. (6.1)
- •
- with pomalidomide and dexamethasone are constipation and hyperglycemia. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Bristol-Myers Squibb at 1-800-721-5072 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
USE IN SPECIFIC POPULATIONS
Pregnancy: Embryo-fetal toxicity in combination with lenalidomide and dexamethasone or pomalidomide and dexamethasone. (8.1)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 3/2022
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosing when EMPLICITI Is Used in Combination with Lenalidomide and Dexamethasone
2.2 Recommended Dosing when EMPLICITI Is Used in Combination with Pomalidomide and Dexamethasone
2.3 Premedication
2.4 Dose Modifications
2.5 Administration
2.6 Reconstitution and Preparation
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Infusion Reactions
5.2 Infections
5.3 Second Primary Malignancies
5.4 Hepatotoxicity
5.5 Interference with Determination of Complete Response
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Immunogenicity
7 DRUG INTERACTIONS
7.1 Drug Interactions
7.2 Laboratory Test Interference
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
- •
- EMPLICITI is indicated in combination with lenalidomide and dexamethasone for the treatment of adult patients with multiple myeloma who have received one to three prior therapies.
- •
- EMPLICITI is indicated in combination with pomalidomide and dexamethasone for the treatment of adult patients with multiple myeloma who have received at least two prior therapies including lenalidomide and a proteasome inhibitor.
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosing when EMPLICITI Is Used in Combination with Lenalidomide and Dexamethasone
The recommended dosage of EMPLICITI is 10 mg/kg administered intravenously every week for the first two cycles (28-day cycle) and every 2 weeks thereafter in conjunction with the recommended dosing of lenalidomide and low-dose dexamethasone as described below. Continue treatment until disease progression or unacceptable toxicity.
Refer to the dexamethasone and lenalidomide prescribing information for additional information.
Administer premedications before each dose of EMPLICITI [see Dosage and Administration (2.3) and Warnings and Precautions (5.1)].
Administer dexamethasone as follows:
- •
- On days that EMPLICITI is administered, give dexamethasone 28 mg orally between 3 and 24 hours before EMPLICITI plus 8 mg intravenously between 45 and 90 minutes before EMPLICITI.
- •
- On days that EMPLICITI is not administered but a dose of dexamethasone is scheduled (Days 8 and 22 of cycle 3 and all subsequent cycles), give 40 mg orally.
The recommended dosing is presented in Table 1.
Table 1: Recommended Dosing Schedule of EMPLICITI in Combination with Lenalidomide and Dexamethasone Cycle 28-Day Cycles 1 and 2 28-Day Cycles 3+ * Premedicate with the following 45 to 90 minutes prior to EMPLICITI infusion: 8 mg intravenous dexamethasone, H1 blocker: diphenhydramine (25 to 50 mg orally or intravenously) or equivalent; H2 blocker: ranitidine (50 mg intravenously) or equivalent; acetaminophen (650 to 1000 mg orally).
† Oral dexamethasone (28 mg) taken between 3 and 24 hours before EMPLICITI infusion.
** Intravenous dexamethasone 45-90 minutes before EMPLICITI infusion.Day of Cycle
1
8
15
22
1
8
15
22
Premedication*
✓
✓
✓
✓
✓
✓
EMPLICITI (mg/kg) intravenously
10
10
10
10
10
10
Lenalidomide (25 mg) orally
Days 1-21
Days 1-21
Dexamethasone† (mg) orally
28
28
28
28
28
40
28
40
Dexamethasone** (mg) intravenously
8
8
8
8
8
8
Day of Cycle
1
8
15
22
1
8
15
22
2.2 Recommended Dosing when EMPLICITI Is Used in Combination with Pomalidomide and Dexamethasone
The recommended dosage of EMPLICITI is 10 mg/kg administered intravenously every week for the first two cycles (28-day cycle). Starting at cycle 3 (28-day cycle), administer EMPLICITI 20 mg/kg intravenously every 4 weeks. Administer EMPLICITI in conjunction with pomalidomide and low-dose dexamethasone as described below (Table 2). Continue treatment until disease progression or unacceptable toxicity.
Refer to the dexamethasone and pomalidomide prescribing information for additional information.
Administer premedications before each dose of EMPLICITI [see Dosage and Administration (2.3) and Warnings and Precautions (5.1)].
Administer dexamethasone as follows:
- •
- On days that EMPLICITI is administered, for patients 75 years or younger give dexamethasone 28 mg orally between 3 and 24 hours before EMPLICITI plus 8 mg intravenously between 45 and 90 minutes before EMPLICITI and for patients older than 75 years give dexamethasone 8 mg orally between 3 and 24 hours before EMPLICITI plus 8 mg intravenously between 45 and 90 minutes before EMPLICITI.
- •
- On days that EMPLICITI is not administered but a dose of dexamethasone is scheduled (Days 8, 15 and 22 of cycle 3 and all subsequent cycles), give 40 mg orally to patients 75 years or younger and 20 mg orally to patients older than 75 years.
The recommended dosing is presented in Table 2.
Table 2: Recommended Dosing Schedule of EMPLICITI in Combination with Pomalidomide and Dexamethasone Cycle 28-Day Cycles 1 and 2 28-Day Cycles 3+ * Premedicate with the following 45 to 90 minutes prior to EMPLICITI infusion: 8 mg intravenous dexamethasone, H1 blocker: diphenhydramine (25 to 50 mg orally or intravenously) or equivalent; H2 blocker: ranitidine (50 mg intravenously) or equivalent; acetaminophen (650 to 1000 mg orally).
† Oral dexamethasone taken between 3 and 24 hours before EMPLICITI infusion.
** Intravenous dexamethasone 45-90 minutes before EMPLICITI infusion.Day of Cycle
1
8
15
22
1
8
15
22
Premedication*
✓
✓
✓
✓
✓
EMPLICITI (mg/kg) intravenously
10
10
10
10
20
Pomalidomide (4 mg) orally
Days 1-21
Days 1-21
Dexamethasone† (mg) orally ≤75 years old
28
28
28
28
28
40
40
40
Dexamethasone† (mg) orally >75 years old
8
8
8
8
8
20
20
20
Dexamethasone** (mg) intravenously
8
8
8
8
8
2.3 Premedication
Dexamethasone
When EMPLICITI is used in combination with lenalidomide or pomalidomide and dexamethasone, divide dexamethasone into an oral and intravenous dose and administer as shown in Table 1 and Table 2 [see Dosage and Administration (2.1, 2.2)].
Other Medications
In addition to dexamethasone, complete administration of the following medications 45 to 90 minutes prior to EMPLICITI infusion:
- •
- H1 blocker: diphenhydramine (25 to 50 mg orally or intravenously) or equivalent H1 blocker.
- •
- H2 blocker: ranitidine (50 mg intravenously or 150 mg orally) or equivalent H2 blocker.
- •
- Acetaminophen (650 to 1000 mg orally).
2.4 Dose Modifications
If the dose of one drug in the regimen is delayed, interrupted, or discontinued, the treatment with the other drugs may continue as scheduled. However, if dexamethasone is delayed or discontinued, base the decision whether to administer EMPLICITI on clinical judgment (i.e., risk of hypersensitivity).
If a Grade 2 or higher infusion reaction occurs during EMPLICITI administration, interrupt the infusion and institute appropriate medical and supportive measures. Upon resolution to Grade 1 or lower, restart EMPLICITI at 0.5 mL per minute and gradually increase at a rate of 0.5 mL per minute every 30 minutes as tolerated to the rate at which the infusion reaction occurred. Resume the escalation regimen if there is no recurrence of the infusion reaction (see Table 3 and Table 4).
In patients who experience an infusion reaction, monitor vital signs every 30 minutes for 2 hours after the end of the EMPLICITI infusion. If the infusion reaction recurs, stop the EMPLICITI infusion and do not restart on that day [see Warnings and Precautions (5.1)]. Severe infusion reactions may require permanent discontinuation of EMPLICITI therapy and emergency treatment.
Dose delays and modifications for dexamethasone, pomalidomide and lenalidomide should be performed as recommended in their Prescribing Information.
2.5 Administration
Administer the entire EMPLICITI infusion with an infusion set and a sterile, nonpyrogenic, low-protein-binding filter (with a pore size of 0.2 to 1.2 micrometer) using an automated infusion pump.
Initiate EMPLICITI infusion at a rate of 0.5 mL per minute for 10 mg/kg dose. The infusion rate may be increased in a stepwise fashion as described in Table 3 if no infusion reactions develop. The maximum infusion rate should not exceed 5 mL per minute.
Table 3: Infusion Rate for EMPLICITI 10 mg/kg Cycle 1, Dose 1 Cycle 1, Dose 2 Cycle 1, Dose 3 and 4
and All Subsequent CyclesTime Interval
Rate
Time Interval
Rate
Rate
0-30 min
0.5 mL/min
0-30 min
3 mL/min
30-60 min
1 mL/min
30 min or more
4 mL/min
5 mL/min
60 min or more
2 mL/min
-
-
Initiate EMPLICITI infusion rate at 3 mL per minute for 20 mg/kg dose. The infusion rate may be increased in a stepwise fashion as described in Table 4 if no infusion reactions develop. The maximum infusion rate should not exceed 5 mL per minute.
Patients who have escalated to 5 mL/min at 10 mg/kg dose must decrease the rate to 3 mL/min at the first infusion at 20 mg/kg.
Table 4: Infusion Rate for EMPLICITI 20 mg/kg Dose 1
Dose 2 and all subsequent doses
Time Interval
Rate
Rate
0-30 min
3 mL/min
30 min or more
4 mL/min
5 mL/min
Adjust the infusion rate following a Grade 2 or higher infusion reaction [see Dosage and Administration (2.4)].
Do not mix EMPLICITI with, or administer as an infusion with, other medicinal products. No physical or biochemical compatibility studies have been conducted to evaluate the coadministration of EMPLICITI with other agents.
2.6 Reconstitution and Preparation
Calculation of Dose
- •
- Calculate the dose (mg) and determine the number of vials needed for the 10 mg/kg and 20 mg/kg dosage based on patient weight.
- •
- Determine the volume of sterile water for injection (SWFI) needed for reconstitution as shown in Table 5.
Table 5: Reconstitution Instructions for EMPLICITI Strength Amount of Sterile Water for Injection, USP Required for Reconstitution Deliverable Volume of Reconstituted EMPLICITI in the Vial Postreconstitution Concentration * After reconstitution, each vial contains overfill to allow for withdrawal of 12 mL (300 mg) and 16 mL (400 mg), respectively. 300 mg vial
13 mL
12 mL*
25 mg/mL
400 mg vial
17 mL
16 mL*
25 mg/mL
Reconstitution
- •
- Aseptically reconstitute each EMPLICITI vial with a syringe of adequate size and a less than or equal to 18-gauge needle (e.g., 17-gauge). A slight back pressure may be experienced during administration of the Sterile Water for Injection, USP, which is considered normal.
- •
- Hold the vial upright and swirl the solution by rotating the vial to dissolve the lyophilized cake. Invert the vial a few times in order to dissolve any powder that may be present on top of the vial or the stopper. Avoid vigorous agitation. DO NOT SHAKE. The lyophilized powder should dissolve in less than 10 minutes.
- •
- After the remaining solids are completely dissolved, allow the reconstituted solution to stand for 5 to 10 minutes. The reconstituted preparation results in a colorless to slightly yellow, clear to slightly opalescent solution. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Discard the solution if any particulate matter or discoloration is observed.
Dilution
- •
- Once the reconstitution is completed, withdraw the necessary volume for the calculated dose from each vial, up to a maximum of 16 mL from 400 mg vial and 12 mL from 300 mg vial.
- •
- Further dilute with either 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP, into an infusion bag made of polyvinyl chloride or polyolefin. The final infusion concentration should range between 1 mg/mL and 6 mg/mL.
- •
- The volume of 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP should be adjusted so as not to exceed 5 mL/kg of patient weight at any given dose of EMPLICITI.
Complete the EMPLICITI infusion within 24 hours of reconstitution of the EMPLICITI lyophilized powder. If not used immediately, the infusion solution may be stored under refrigeration conditions: 2°C to 8°C (36°F-46°F) and protected from light for up to 24 hours (a maximum of 8 hours of the total 24 hours can be at room temperature, 20°C to 25°C [68°F-77°F], and room light).
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Infusion Reactions
EMPLICITI can cause infusion reactions. Infusion reactions were reported in 10% of patients treated with EMPLICITI in the ELOQUENT-2 trial and 3.3% in the ELOQUENT-3 trial. In the ELOQUENT-2 trial, all reports of infusion reaction were Grade 3 or lower. In the ELOQUENT-2 trial, Grade 3 infusion reactions occurred in 1% of patients. The most common symptoms of an infusion reaction included fever, chills, and hypertension. Bradycardia and hypotension also developed during infusions.
In the ELOQUENT-2 trial, 5% of patients required interruption of the administration of EMPLICITI for a median of 25 minutes due to infusion reactions, and 1% of patients discontinued due to infusion reactions. Of the patients who experienced an infusion reaction, 70% (23/33) had them during the first dose.
In the ELOQUENT-3 trial, the only infusion reaction symptom was chest discomfort (2%), which was Grade 1. All patients in the ELOQUENT-3 trial who experienced an infusion reaction had them during the first treatment cycle.
Administer premedication consisting of dexamethasone, antihistamines (H1 and H2 blockers) and acetaminophen prior to EMPLICITI infusion [see Dosage and Administration (2.3)].
Interrupt EMPLICITI infusion for Grade 2 or higher infusion reactions and institute appropriate medical management [see Dosage and Administration (2.4)].
5.2 Infections
In the ELOQUENT-2 trial (N=635), infections were reported in 81% of patients in EMPLICITI combined with lenalidomide and dexamethasone (E-Ld) arm and 74% in lenalidomide and dexamethasone (Ld). In the ELOQUENT-3 trial (N=115), infections were reported in 65% of patients in both EMPLICITI combined with pomalidomide and dexamethasone (E-Pd) arm and in pomalidomide and dexamethasone (Pd) arm. In the ELOQUENT-2 trial, Grade 3 to 4 infections were noted in 28% and 24% of E-Ld- and Ld-treated patients and in the ELOQUENT-3 trial, 13% and 22% of E-Pd- and Pd-treated patients, respectively. Discontinuations due to infections occurred in 3.5% of E-Ld-treated and 4.1% of Ld-treated patients in the ELOQUENT-2 trial and 7% of E-Pd-treated and 5% of Pd-treated patients in the ELOQUENT-3 trial. Fatal infections were reported in 2.5% and 2.2% of E-Ld- and Ld-treated patients in the ELOQUENT-2 trial and 5% and 3.6% of E-Pd- and Pd-treated patients in the ELOQUENT-3 trial.
Opportunistic infections were reported in 22% of patients in the E-Ld arm and 13% of patients in the Ld arm in the ELOQUENT-2 trial and 10% of patients in the E-Pd arm and 9% of patients in the Pd arm in the ELOQUENT-3 trial. In the ELOQUENT-2 trial, fungal infections occurred in 10% of patients in the E-Ld arm and 5% of patients in the Ld arm. Herpes zoster was reported in 14% of patients treated with E-Ld and 7% of patients treated with Ld in the ELOQUENT-2 trial and 5% of patients treated with E-Pd and 1.8% of patients treated with Pd in the ELOQUENT-3 trial.
Monitor patients for development of infections and treat promptly.
5.3 Second Primary Malignancies
In the ELOQUENT-2 trial (N=635), invasive second primary malignancies (SPM) have been observed in 9% of patients treated with E-Ld and 6% of patients treated with Ld and in the ELOQUENT-3 trial (N=115) in 1.8% of patients treated with Pd and in none of the patients treated with E-Pd. In the ELOQUENT-2 trial, the rate of hematologic malignancies were the same between E-Ld and Ld treatment arms (1.6%). Solid tumors were reported in 3.5% and 2.2% of E-Ld- and Ld-treated patients, respectively. Skin cancer was reported in 4.4% and 2.8% of patients treated with E-Ld and Ld, respectively.
Monitor patients for the development of second primary malignancies.
5.4 Hepatotoxicity
In the ELOQUENT-2 trial (N=635), elevations in liver enzymes (aspartate transaminase/alanine transaminase [AST/ALT] greater than 3 times the upper limit, total bilirubin greater than 2 times the upper limit, and alkaline phosphatase less than 2 times the upper limit) consistent with hepatotoxicity were reported in 2.5% and 0.6% of E-Ld- and Ld-treated patients. Two patients experiencing hepatotoxicity were not able to continue treatment; however, 6 out of 8 patients had resolution and were able to continue treatment. Monitor liver enzymes periodically. Stop EMPLICITI upon Grade 3 or higher elevation of liver enzymes. After return to baseline values, continuation of treatment may be considered.
5.5 Interference with Determination of Complete Response
EMPLICITI is a humanized IgG kappa monoclonal antibody that can be detected on both the serum protein electrophoresis (SPEP) and immunofixation (IFE) assays used for the clinical monitoring of endogenous M-protein [see Drug Interactions (7.2)]. This interference can impact the determination of complete response and possibly relapse from complete response in patients with IgG kappa myeloma protein.
-
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are described in detail in other sections of the label:
- •
- Infusion reaction [see Warnings and Precautions (5.1)].
- •
- Infections [see Warnings and Precautions (5.2)].
- •
- Second Primary Malignancies [see Warnings and Precautions (5.3)].
- •
- Hepatotoxicity [see Warnings and Precautions (5.4)].
- •
- Interference with determination of complete response [see Warnings and Precautions (5.5)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
EMPLICITI in Combination with Lenalidomide and Dexamethasone [ELOQUENT-2]
The safety data described in this section are based on the ELOQUENT-2 study, a randomized, open-label clinical trial in patients with previously treated multiple myeloma. In ELOQUENT-2, EMPLICITI 10 mg/kg was administered with lenalidomide and dexamethasone [see Clinical Studies (14)]. For adverse reaction evaluation, EMPLICITI combined with lenalidomide and dexamethasone was compared with lenalidomide and dexamethasone alone.
The mean age of the population was 66 years and 57% of patients were 65 years of age or older. Sixty percent (60%) of the population were male, 84% were white, 10% were Asian, and 4% were black. The Eastern Cooperative Oncology Group (ECOG) performance status was 0 in 47%, 1 in 44%, and 2 in 9% of patients.
These data reflect exposure of 318 patients to EMPLICITI and 317 to control with a median number of cycles of 19 for EMPLICITI and 14 for control.
Serious adverse reactions were reported in 65% of patients treated on the EMPLICITI arm and 57% for patients treated on the control arm. The most frequent serious adverse reactions in the EMPLICITI arm compared to the control arm were: pneumonia (15% vs. 11%), pyrexia (7% vs. 5%), respiratory tract infection (3.1% vs. 1.3%), anemia (2.8% vs. 1.9%), pulmonary embolism (3.1% vs. 2.5%), and acute renal failure (2.5% vs. 1.9%).
The proportion of patients who discontinued any component of the treatment regimen due to adverse reactions as listed below was similar for both treatment arms; 6.0% for patients treated on the EMPLICITI arm and 6.3% for patients treated on the control.
Adverse reactions occurring at a frequency of 10% or higher in the EMPLICITI arm and 5% or higher than the lenalidomide and dexamethasone arm for the randomized trial in multiple myeloma are presented in Table 6.
Table 6: ELOQUENT-2: Adverse Reactions with a 10% or Higher Incidence for EMPLICITI-Treated Patients and a 5% or Higher Incidence than Lenalidomide and Dexamethasone-Treated Patients [All Grades] EMPLICITI +
Lenalidomide and Dexamethasone
N=318Lenalidomide and Dexamethasone
N=317Primary Term All Grades Grade 3/4 All Grades Grade 3/4 * The term fatigue is a grouping of the following terms: fatigue and asthenia.
† The term cough is a grouping of the following terms: cough, productive cough, and upper airway cough.
‡ The term peripheral neuropathy is a grouping of the following terms: peripheral neuropathy, axonal neuropathy, peripheral motor neuropathy, peripheral sensory neuropathy, and polyneuropathy.
§ The term pneumonia is a grouping of the following terms: pneumonia, atypical pneumonia, bronchopneumonia, lobar pneumonia, bacterial pneumonia, fungal pneumonia, pneumonia influenza, and pneumococcal pneumonia.Fatigue*
62
13
52
12
Diarrhea
47
5.0
36
4.1
Pyrexia
37
2.5
25
2.8
Constipation
36
1.3
27
0.3
Cough†
34
0.3
19
0
Peripheral Neuropathy‡
27
3.8
21
2.2
Nasopharyngitis
25
0
19
0
Upper Respiratory Tract Infection
23
0.6
17
1.3
Decreased Appetite
21
1.6
13
1.3
Pneumonia§
20
14
14
9
Pain in Extremities
16
0.9
10
0.3
Headache
15
0.3
8
0.3
Vomiting
14
0.3
9
0.9
Weight Decreased
14
1.3
6
0
Lymphopenia
13
9
7
3.2
Cataracts
12
6
6
2.8
Oropharyngeal Pain
10
0
4
0
Laboratory abnormalities worsening from baseline and occurring at a frequency of 10% or higher in the EMPLICITI group and 5% or higher than the lenalidomide and dexamethasone group (criteria met for all Grades or Grade 3/4) for ELOQUENT-2 are presented in Table 7.
Table 7: ELOQUENT-2: Laboratory Abnormalities Worsening from Baseline and with a 10% or Higher Incidence for EMPLICITI-Treated Patients and a 5% Higher Incidence than Lenalidomide and Dexamethasone-Treated Patients [Criteria met for All Grades or Grade 3/4] EMPLICITI +
Lenalidomide and Dexamethasone
N=318Lenalidomide and Dexamethasone
N=317Laboratory Parameter All Grades Grade 3/4 All Grades Grade 3/4 Hematology
Lymphopenia
99
77
98
49
Leukopenia
91
32
88
26
Thrombocytopenia
84
19
78
20
Liver and Renal Function Tests
Hypoalbuminemia
73
3.9
66
2.3
Elevated Alkaline Phosphatase
39
1.3
30
0
Chemistry
Hyperglycemia
89
17
85
10
Hypocalcemia
78
11
77
4.7
Low Bicarbonate
63
0.4
45
0
Hyperkalemia
32
7
22
1.6
Vital sign abnormalities were assessed by treatment arm for the randomized trial in multiple myeloma and are presented in Table 8. Percentages are based on patients who had at least one vital sign abnormality any time during the course of study.
Table 8: ELOQUENT-2 Vital Sign Abnormalities EMPLICITI +
Lenalidomide and Dexamethasone
N=318Lenalidomide and Dexamethasone
N=317Vital Sign Parameter % % Systolic Blood Pressure ≥160 mmHg
33
21
Diastolic Blood Pressure ≥100 mmHg
17
12
Systolic Blood Pressure <90 mmHg
29
8
Heart Rate ≥100 bpm
48
30
Heart Rate <60 bpm
66
31
EMPLICITI in Combination with Pomalidomide and Dexamethasone [ELOQUENT-3]
The safety data described in this section are based on ELOQUENT-3, a randomized, open-label clinical trial in patients with previously treated multiple myeloma. In ELOQUENT-3, EMPLICITI 10 mg/kg and 20 mg/kg was administered with pomalidomide and dexamethasone [see Clinical Studies (14)]. For adverse reaction evaluation, EMPLICITI combined with pomalidomide and dexamethasone was compared with pomalidomide and dexamethasone alone.
The mean age of the population was 66 years and 63% of patients were 65 years of age or older. Fifty-seven percent of the population were male, 78% were white, 20% were Asian, and none were black. The ECOG performance status was 0 in 43%, 1 in 46%, and 2 in 10% of patients. These data reflect exposure of 60 patients to EMPLICITI and 55 to control with a median number of cycles of 9 for EMPLICITI and 5 for control.
Serious adverse reactions were reported in 70% of patients treated on the EMPLICITI arm and 60% for patients treated on the control arm. The most frequent serious adverse reactions in the EMPLICITI arm compared to the control arm were: pneumonia (13% vs. 11%) and respiratory tract infection (7% vs. 3.6%). The proportion of patients who discontinued any component of the treatment regimen due to adverse reactions were 5% of the patients in the EMPLICITI arm and 1.8% of the patients in the control arm.
Adverse reactions occurring at a frequency of 10% or higher in the EMPLICITI arm and 5% or higher than the pomalidomide and dexamethasone arm for the randomized trial in multiple myeloma are presented in Table 9.
Table 9: ELOQUENT-3: Adverse Reactions with a 10% or Higher Incidence for EMPLICITI-Treated Patients and a 5% or Higher Incidence than Pomalidomide and Dexamethasone-Treated Patients [All Grades] EMPLICITI +
Pomalidomide and Dexamethasone
N=60Pomalidomide and Dexamethasone
N=55Adverse Reaction All Grades Grade 3/4 All Grades Grade 3/4 * The term pneumonia is grouping of the following terms: pneumonia, atypical pneumonia, lower respiratory tract infection, pneumoccocal sepsis, pneumonia bacterial, pneumonia influenza. Constipation
22
1.7
11
0
Hyperglycemia
20
8
15
7
Pneumonia*
18
10
13
11
Diarrhea
18
0
9
0
Respiratory Tract Infection
17
0
9
1.8
Bone Pain
15
3.3
9
0
Dyspnea
15
3.3
7
1.8
Muscle Spasms
13
0
5
0
Edema Peripheral
13
0
7
0
Lymphopenia
10
8
1.8
1.8
Other clinically important adverse reactions reported in patients treated with EMPLICITI that did not meet the criteria for inclusion in Table 6 and 9 but occurred at a frequency of 5% or greater in the EMPLICITI group and at a frequency at least twice the control rate for the randomized trial in multiple myeloma are listed below:
General disorders and administration site conditions: chest pain, night sweats
Immune system disorders: hypersensitivity
Nervous system disorders: hypoesthesia
Psychiatric disorders: mood altered
Laboratory abnormalities worsening from baseline and occurring at a frequency of 10% or higher in ELOQUENT-3 in the EMPLICITI group and 5% or higher than the pomalidomide and dexamethasone group (criteria met for all Grades or Grade 3/4) for the randomized trial in multiple myeloma are presented in Table 10.
Table 10: ELOQUENT-3: Laboratory Abnormalities Worsening from Baseline and with a 10% or Higher Incidence for EMPLICITI-Treated Patients and a 5% Higher Incidence than Pomalidomide and Dexamethasone-Treated Patients [Criteria met for All Grades or Grade 3/4] EMPLICITI +
Pomalidomide and Dexamethasone
N=60Pomalidomide and Dexamethasone
N=55Laboratory Parameter All Grades Grade 3/4 All Grades Grade 3/4 Hematology
Lymphopenia
98
70
91
35
Leukopenia
80
52
87
35
Thrombocytopenia
78
17
73
20
Liver and Renal Function Tests
Hypoalbuminemia
65
1.7
56
1.8
Chemistry
Hypocalcemia
58
3.3
40
1.8
Hyperglycemia
40
3.3
25
1.8
Hyponatremia
40
5
18
0
Hypokalemia
23
5
16
3.6
Vital sign abnormalities were assessed by treatment arm for the randomized trial in multiple myeloma and are presented in Table 11. Percentages are based on all treated patients
Table 11: ELOQUENT-3: Vital Sign Abnormalities EMPLICITI +
Pomalidomide and Dexamethasone
N=60Pomalidomide and Dexamethasone
N=55Vital Sign Parameter % % Systolic Blood Pressure ≥160 mmHg
18
13
Diastolic Blood Pressure ≥100 mmHg
8
4.0
Systolic Blood Pressure <90 mmHg
7
7
Heart Rate ≥100 bpm
23
24
Heart Rate <60 bpm
43
22
6.2 Immunogenicity
As with all therapeutic proteins, there is a potential for immunogenicity to EMPLICITI. The detection of antibody formation is highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of antibody (including neutralizing antibody) positivity in an assay may be influenced by several factors including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of incidence of antibodies to EMPLICITI in the studies described below with the incidences of antibodies in other studies or to other products may be misleading.
Of 390 patients across four clinical studies including ELOQUENT-2 and in 53 patients in the ELOQUENT-3 trial, who were treated with EMPLICITI and evaluable for the presence of anti-product antibodies, 72 patients (18.5%) in the four clinical trials and 19 patients (36%) in the ELOQUENT-3 trial tested positive for treatment-emergent anti-product antibodies by an electrochemiluminescent (ECL) assay. In 63 (88%) of the 72 patients in the four clinical trials, anti-product antibodies occurred within the first 2 months of the initiation of EMPLICITI treatment and resolved by 2 to 4 months in 49 (78%) patients. In the ELOQUENT-3 trial, in all 19 patients, anti-product antibodies occurred within the first 2 months of the initiation of EMPLICITI treatment and were resolved by 2 to 3 months in 18 (95%) patients.
Neutralizing antibodies post-treatment were detected in 19 of 299 patients in the four clinical trials and 2 of 53 patients in the ELOQUENT-3 trial who were evaluable for the presence of neutralizing antibodies.
-
7 DRUG INTERACTIONS
7.1 Drug Interactions
For important drug interactions involving lenalidomide, pomalidomide and dexamethasone, refer to their respective prescribing information.
7.2 Laboratory Test Interference
EMPLICITI may be detected in the SPEP and serum immunofixation assays of myeloma patients and could interfere with correct response classification. A small peak in the early gamma region on SPEP that is IgGƙ on serum immunofixation may potentially be attributed to EMPLICITI, particularly in patients whose endogenous myeloma protein is IgA, IgM, IgD, or lambda light chain restricted. This interference can impact the determination of complete response and possibly relapse from complete response in patients with IgG kappa myeloma protein [see Warnings and Precautions (5.5)].
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no available data on EMPLICITI use in pregnant women to inform a drug associated risk of major birth defects and miscarriage. Animal reproduction studies have not been conducted with elotuzumab.
EMPLICITI is administered in combination with lenalidomide and dexamethasone or pomalidomide and dexamethasone. Lenalidomide and pomalidomide can cause embryo-fetal harm and are contraindicated for use in pregnancy. Refer to the lenalidomide, pomalidomide and dexamethasone prescribing information for additional information. Lenalidomide and pomalidomide are only available through a REMS program.
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
8.2 Lactation
Risk Summary
There are no data on the presence of EMPLICITI in human milk, the effects on the breastfed child, or the effects on milk production. Because of the potential for serious adverse reactions in the breastfed child from elotuzumab administered in combination with lenalidomide and dexamethasone or pomalidomide and dexamethasone, advise lactating women not to breastfeed during treatment with EMPLICITI. Refer to the lenalidomide, pomalidomide and dexamethasone prescribing information for additional information.
8.3 Females and Males of Reproductive Potential
Pregnancy Testing
Refer to the lenalidomide and pomalidomide labeling for pregnancy testing requirements prior to initiating treatment in females of reproductive potential.
When EMPLICITI is used with lenalidomide or pomalidomide, there is a risk of fetal harm, including severe life-threatening human birth defects associated with lenalidomide and pomalidomide, and the need to follow requirements regarding pregnancy avoidance, including testing.
Contraception
Females
Refer to the lenalidomide and pomalidomide labeling for contraception requirements prior to initiating treatment in females of reproductive potential.
Males
Lenalidomide and pomalidomide are present in the blood and semen of patients receiving the drug. Refer to the lenalidomide and pomalidomide full prescribing information for requirements regarding contraception and the prohibitions against blood and/or sperm donation due to presence and transmission in blood and/or semen and for additional information.
8.5 Geriatric Use
Of the 646 patients across treatment groups in the ELOQUENT-2 randomized trial designed to evaluate the use of EMPLICITI in combination with lenalidomide and low-dose dexamethasone in multiple myeloma, 57% were 65 years of age or older; the number of patients 65 years or older was similar between treatment groups. No overall differences in efficacy or safety were observed between patients 65 years or older and younger patients (less than 65 years of age).
Of the 117 patients across treatment groups in the ELOQUENT-3 randomized trial designed to evaluate the use of EMPLICITI in combination with pomalidomide and low-dose dexamethasone in multiple myeloma, 62% were 65 years of age or older; the number of patients 65 years or older was similar between treatment groups. This study did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
-
10 OVERDOSAGE
The dose of EMPLICITI at which severe toxicity occurs is not known. EMPLICITI does not appear to be removed by dialysis as determined in a study of patients with renal impairment.
In case of overdosage, monitor patients closely for signs or symptoms of adverse reactions and institute appropriate symptomatic treatment.
-
11 DESCRIPTION
Elotuzumab is a humanized recombinant monoclonal antibody directed to SLAMF7, a cell surface glycoprotein. Elotuzumab consists of the complementary determining regions (CDR) of the mouse antibody, MuLuc63, grafted onto human IgG1 heavy and kappa light chain frameworks. Elotuzumab is produced in NS0 cells by recombinant DNA technology. Elotuzumab has a theoretical mass of 148.1 kDa for the intact antibody.
EMPLICITI (elotuzumab) is a sterile, nonpyrogenic, preservative-free lyophilized powder that is white to off-white, whole or fragmented cake in single-dose vials. EMPLICITI for Injection is supplied as 300 mg per vial and 400 mg per vial and requires reconstitution with Sterile Water for Injection, USP (13 mL and 17 mL, respectively) to obtain a solution with a concentration of 25 mg/mL. After reconstitution, each vial contains overfill to allow for withdrawal of 12 mL (300 mg) and 16 mL (400 mg). The reconstituted solution is colorless to slightly yellow, clear to slightly opalescent. Prior to intravenous infusion, the reconstituted solution is diluted with either 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP [see Dosage and Administration (2.6)].
Each 300 mg single-dose vial of EMPLICITI also contains the following inactive ingredients: citric acid monohydrate (2.44 mg), polysorbate 80 (3.4 mg), sodium citrate (16.6 mg), and sucrose (510 mg).
Each 400 mg single-dose vial of EMPLICITI also contains the following inactive ingredients: citric acid monohydrate (3.17 mg), polysorbate 80 (4.4 mg), sodium citrate (21.5 mg), and sucrose (660 mg).
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Elotuzumab is a humanized IgG1 monoclonal antibody that specifically targets the SLAMF7 (Signaling Lymphocytic Activation Molecule Family member 7) protein. SLAMF7 is expressed on myeloma cells independent of cytogenetic abnormalities. SLAMF7 is also expressed on Natural Killer (NK) cells, plasma cells, and at lower levels on specific immune cell subsets of differentiated cells within the hematopoietic lineage.
Elotuzumab directly activates NK cells through the SLAMF7 pathway. Elotuzumab also targets SLAMF7 on myeloma cells and interacts with Fc receptors on effector cells to mediate the killing of myeloma cells through antibody-dependent cellular cytotoxicity (ADCC) and antibody-dependent cellular phagocytosis (ADCP). In nonclinical models, the combination of elotuzumab and lenalidomide resulted in enhanced activation of NK cells that was greater than the effects of either agent alone and increased anti-tumor activity in vitro and in vivo. Elotuzumab has also demonstrated increased anti-tumor activity when combined with pomalidomide in vivo.
12.3 Pharmacokinetics
Elotuzumab exhibits nonlinear pharmacokinetics (PK) resulting in greater than proportional increases in area under the concentration-time curve (AUC) indicative of target-mediated clearance. The administration of the recommended 10 mg/kg EMPLICITI regimen with lenalidomide and dexamethasone is predicted to result in geometric mean (CV%) steady-state trough concentrations of 194 μg/mL (52%). The administration of the recommended EMPLICITI regimen with pomalidomide and dexamethasone is predicted to result in geometric mean (CV%) steady-state trough concentrations of 124 μg/mL (59%).
Elimination
The clearance of elotuzumab in combination with lenalidomide and dexamethasone decreased from a geometric mean (CV%) of 17.5 (21.2%) to 5.8 (31%) mL/day/kg with an increase in dose from 0.5 (i.e., 0.05 times the recommended dosage) to 20 mg/kg (i.e., 2 times the recommended dosage). When elotuzumab is administered with lenalidomide and dexamethasone, approximately 97% of the maximum steady-state concentration is predicted to be eliminated with a geometric mean (CV%) of 82.4 (48%) days. When elotuzumab is administered with pomalidomide and dexamethasone, approximately 97% of the maximum steady-state concentration is predicted to be eliminated with a geometric mean (CV%) of 78 (42%) days.
Specific Populations
Clinically significant differences were not observed in the PK of elotuzumab based on age (37 to 88 years), sex, race, baseline lactate dehydrogenase, albumin, renal impairment (creatinine clearance (CLcr) 15 to 89 mL/min), end-stage renal disease (CLcr <15 mL/min) with or without hemodialysis, mild hepatic impairment (total bilirubin ≤ upper limit of normal (ULN) and aspartate transaminase (AST) > ULN OR total bilirubin 1 to 1.5 times the ULN and AST any value), and coadministration with lenalidomide/dexamethasone or pomalidomide/dexamethasone. The PK of elotuzumab in patients with moderate (total bilirubin > 1.5 to 3 times the ULN and AST any value) to severe (total bilirubin > 3 times the ULN and AST any value) hepatic impairment is unknown.
The clearance of elotuzumab increased with increasing body weight supporting a weight-based dose.
- 13 NONCLINICAL TOXICOLOGY
-
14 CLINICAL STUDIES
ELOQUENT-2 (NCT01239797)
The efficacy and safety of EMPLICITI in combination with lenalidomide and dexamethasone were evaluated in ELOQUENT-2, a randomized, open-label trial in patients with multiple myeloma who had received one to three prior therapies and had documented progression following their most recent therapy.
Eligible patients were randomized in a 1:1 ratio to receive either EMPLICITI in combination with lenalidomide and low-dose dexamethasone or lenalidomide and low-dose dexamethasone. Treatment was administered in 4-week cycles until disease progression or unacceptable toxicity. EMPLICITI 10 mg/kg was administered intravenously each week for the first 2 cycles and every 2 weeks thereafter. Prior to EMPLICITI infusion, dexamethasone was administered as a divided dose: an oral dose of 28 mg and an intravenous dose of 8 mg. In the control group and on weeks without EMPLICITI, dexamethasone 40 mg was administered as a single oral dose weekly. Lenalidomide 25 mg was taken orally once daily for the first 3 weeks of each cycle. Assessment of tumor response was conducted every 4 weeks.
A total of 646 patients were randomized to receive treatment: 321 to EMPLICITI in combination with lenalidomide and low-dose dexamethasone and 325 to lenalidomide and low-dose dexamethasone.
Demographics and baseline disease characteristics were balanced between treatment arms. The median age was 66 years (range, 37-91); 57% of patients were 65 years or older; 60% of patients were male; whites comprised 84% of the study population, Asians 10%, and blacks 4%. The ECOG performance status was 0 in 47%, 1 in 44%, and 2 in 9% of patients, and ISS Stage was I in 43%, II in 32%, and III in 21% of patients. The cytogenetic categories of del 17p and t(4;14) were present in 32% and 9% of patients, respectively. The median number of prior therapies was 2. Thirty-five percent (35%) of patients were refractory (progression during or within 60 days of last therapy) and 65% were relapsed (progression after 60 days of last therapy). Prior therapies included stem cell transplant (55%), bortezomib (70%), melphalan (65%), thalidomide (48%), and lenalidomide (6%).
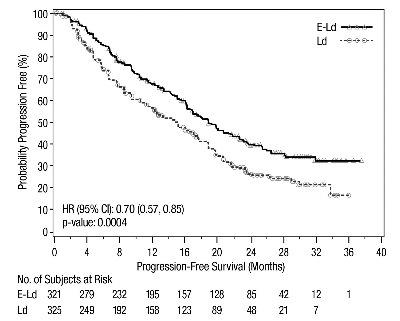
The efficacy of EMPLICITI was evaluated by progression-free survival (PFS) as assessed by hazard ratio, and overall response rate (ORR) as determined by a blinded Independent Review Committee using the European Group for Blood and Marrow Transplantation (EBMT) response criteria. Efficacy results are shown in Table 12 and Figure 1. The median number of treatment cycles was 19 for the EMPLICITI group and 14 for the comparator arm with a minimum follow-up of two years.
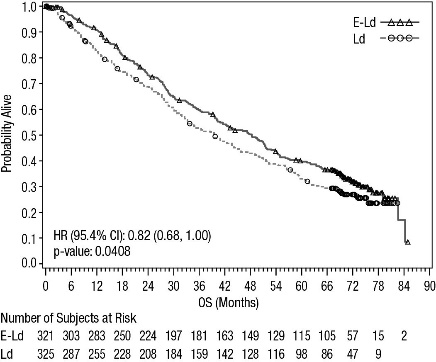
A pre-planned final overall survival (OS) analysis was performed after at least 427 deaths occurred. The minimum follow-up was 70.6 months. The OS results at final analysis reached statistical significance. A significantly longer OS was observed in patients in the E-Ld arm compared to patients in Ld arm, representing an 18% reduction in the risk of death. Efficacy results are presented in Table 12 and Figure 2.
Table 12: ELOQUENT-2 Efficacy Results EMPLICITI +
Lenalidomide/
Dexamethasone
N=321Lenalidomide/
Dexamethasone
N=325PFS * p-value based on the log-rank test stratified by ß2 microglobulins (<3.5 mg/L vs ≥3.5 mg/L), number of prior lines of therapy (1 vs 2 or 3), and prior immunomodulatory therapy (no vs prior thalidomide only vs other).
† European Group for Blood and Marrow Transplantation (EBMT) criteria.
‡ p-value based on the Cochran-Mantel-Haenszel chi-square test stratified by ß2 microglobulins (<3.5 mg/L vs ≥3.5 mg/L), number of prior lines of therapy (1 vs 2 or 3), and prior immunomodulatory therapy (no vs prior thalidomide only vs other).
§ Complete response (CR) + stringent complete response (sCR).
¶ EMPLICITI’s interference with the assessment of myeloma protein with immunofixation and serum protein electrophoresis assay may interfere with correct response classification [see Drug Interactions (7)].
Hazard Ratio [95% CI]
0.70 [0.57, 0.85]
Stratified log-rank test p-value*
0.0004
Median PFS in months [95% CI]
19.4 [16.6, 22.2]
14.9 [12.1, 17.2]
Response
Overall Response (ORR)† n (%)
[95% CI]252 (78.5)
[73.6, 82.9]213 (65.5)
[60.1, 70.7]p-value‡
0.0002
Complete Response (CR + sCR)†,§ n (%)
14 (4.4)¶
24 (7.4)
Very Good Partial Response (VGPR)† n (%)
91 (28.3)
67 (20.6)
Partial Response (PR)† n (%)
147 (45.8)
122 (37.5)
Overall Survival
Hazard Ratio [95.4% CI]
0.82 [0.68, 1.00]
Stratified log-rank test p-value*
0.0408
Median OS in months [95% CI]
48.3 [40.3, 51.9]
39.6 [33.3, 45.3]
Figure 1: ELOQUENT-2 Progression-Free Survival
The 1- and 2-year rates of PFS for EMPLICITI in combination with lenalidomide and dexamethasone treatment were 68% and 41%, respectively, compared with 57% and 27%, respectively, for lenalidomide and dexamethasone treatment.
Figure 2: ELOQUENT-2 Overall Survival
ELOQUENT-3 (NCT02654132)
The efficacy and safety of EMPLICITI in combination with pomalidomide and dexamethasone were evaluated in ELOQUENT-3, a randomized, open-label trial in patients with relapsed or refractory multiple myeloma.
Eligible patients were randomized in a 1:1 ratio to receive either EMPLICITI in combination with pomalidomide and low-dose dexamethasone or pomalidomide and low-dose dexamethasone. Treatment was administered in 4-week cycles until disease progression or unacceptable toxicity. EMPLICITI 10 mg/kg was administered intravenously each week for the first 2 cycles and 20 mg/kg every 4 weeks thereafter.
Prior to EMPLICITI infusion, dexamethasone was administered. Dexamethasone was administered on day 1, 8, 15 and 22 of each cycle. On weeks with EMPLICITI infusion, dexamethasone was administered as a divided dose: subjects 75 years or younger, an oral dose of 28 mg and an intravenous dose of 8 mg, and in subjects older than 75 years an oral dose of 8 mg and an intravenous dose of 8 mg. On weeks without an EMPLICITI infusion and in the control group, dexamethasone was administered in subjects 75 years or younger as an oral dose of 40 mg and in subjects older than 75 years as an oral dose of 20 mg dexamethasone was administered orally. Assessment of tumor response was conducted every 4 weeks.
A total of 117 patients were randomized to receive treatment: 60 to EMPLICITI in combination with pomalidomide and low-dose dexamethasone and 57 to pomalidomide and low-dose dexamethasone.
Demographics and baseline disease characteristics were balanced between treatment arms. The median age was 67 years (range, 36-81); 62% of patients were 65 years or older; 57% of patients were male; whites comprised 77% of the study population, Asians 21%, and blacks 1%. The ECOG performance status was 0 in 44%, 1 in 46%, and 2 in 10% of patients, and ISS Stage was I in 50%, II in 38%, and III in 12% of patients. The chromosomal lab abnormalities as determined by FISH of del 17p and t(4;14) were present in 5% and 11% of patients, respectively. The median number of prior therapies was 3. Eighty-seven percent (87%) of patients were refractory to lenalidomide, 80% refractory to a proteasome inhibitor, 70% were refractory to both lenalidomide and a proteasome inhibitor. Prior therapies included stem cell transplant (55%), bortezomib (100%), lenalidomide (99%), cyclophosphamide (66%), melphalan (63%), carfilzomib (21%), and daratumumab (3%).
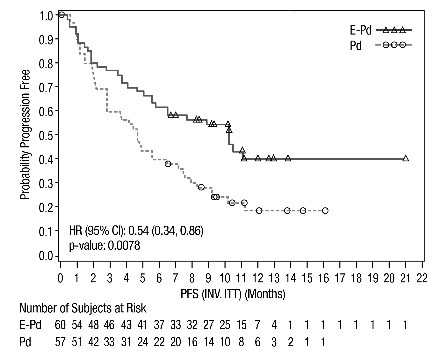
The efficacy of EMPLICITI was evaluated by progression-free survival (PFS) and overall response rate (ORR) as determined by the investigator. Efficacy results are shown in Table 13 and Figure 3. The median number of treatment cycles was 9 for the EMPLICITI group and 5 for the comparator arm with a minimum follow-up of 9.1 months.
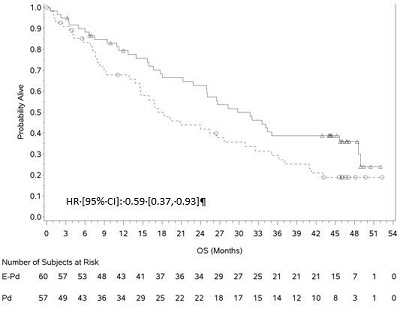
A pre-planned final OS analysis was performed after at least 78 deaths occurred. The minimum follow-up was 45.0 months. A longer OS was observed in patients in the E-Pd arm compared to patients in the Pd arm. Efficacy results are presented in Table 13 and Figure 4.
Table 13: ELOQUENT-3 Efficacy Results EMPLICITI +
Pomalidomide/
Dexamethasone
N=60Pomalidomide/
Dexamethasone
N=57* p-value based on the log-rank test stratified by stage of disease at study entry (International Staging System I-II vs III) and number of prior lines of therapy (2-3 vs >=4) at randomization.
† International Myeloma Working Group (IMWG) criteria.
‡ p-value based on the Cochran-Mantel-Haenszel chi-square test stratified by stage of disease at study entry (International Staging System I-II vs III) and number of prior lines of therapy (2-3 vs >=4) at randomization.
§ Complete response (CR) + stringent complete response (sCR).
¶ EMPLICITI’s interference with the assessment of myeloma protein with immunofixation and serum protein electrophoresis assay may interfere with correct response classification [see Drug Interactions (7)].
NE=Non-EstimablePFS
Hazard Ratio [95% CI]
0.54 [0.34, 0.86]
Stratified log-rank test p-value*
0.0078
Median PFS in months [95% CI]
10.25 [5.59, NE]
4.67 [2.83, 7.16]
Response
Overall Response (ORR)† n (%)
[95% CI]32 (53.3)
[40.0, 66.3]15 (26.3)
[15.5, 39.7]p-value‡
0.0029
Complete Response (CR + sCR)†,§ n (%)
5 (8.3)¶
1 (1.8)
Very Good Partial Response (VGPR)† n (%)
7 (11.7)
4 (7.0)
Partial Response (PR)† n (%)
20 (33.3)
10 (17.5)
Overall Survival
Number of death events
37 (61.7)
41 (74.5)
Hazard Ratio [95% CI]
0.59 [0.37, 0.93]
Median OS in months [95% CI]
29.80 [22.87, 45.67]
17.41 [13.83, 27.70]
Figure 3: ELOQUENT-3 Progression-Free Survival
Figure 4: ELOQUENT-3 Overall Survival
-
16 HOW SUPPLIED/STORAGE AND HANDLING
EMPLICITI (elotuzumab) is white to off-white lyophilized powder available as follows:
Carton Content
NDC
One 300 mg single-dose vial
0003-2291-11
One 400 mg single-dose vial
0003-4522-11
Store EMPLICITI under refrigeration at 2°C to 8°C (36°F-46°F). Protect EMPLICITI from light by storing in the original package until time of use. Do not freeze or shake.
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Infusion Reactions
- •
- EMPLICITI may cause infusion reactions. Advise patients to contact their healthcare provider if they experience signs and symptoms of infusion reactions, including fever, chills, rash, or breathing problems within 24 hours of infusion [see Warnings and Precautions (5.1)].
- •
- Advise patients that they will be required to take the following oral medications prior to EMPLICITI dosing to reduce the risk of infusion reaction [see Dosage and Administration (2.3)]:
- •
- Dexamethasone orally as prescribed
- •
- H1 blocker: diphenhydramine or equivalent (if oral)
- •
- H2 blocker: ranitidine or equivalent (if oral)
- •
- Acetaminophen (650 to 1000 mg orally)
Pregnancy
- •
- Advise patients that lenalidomide and pomalidomide have the potential to cause fetal harm and has specific requirements regarding contraception, pregnancy testing, blood and sperm donation, and transmission in sperm. Lenalidomide and pomalidomide are only available through a REMS program [see Use in Specific Populations (8.1)].
Infections
- •
- Inform patients of the risk of developing infections during treatment with EMPLICITI, and to report any symptoms of infection [see Warnings and Precautions (5.2)].
Second Primary Malignancies
- •
- Inform patients of the risk of developing SPM during treatment with EMPLICITI [see Warnings and Precautions (5.3)].
Hepatotoxicity
- •
- Inform patients of the risk of hepatotoxicity during treatment with EMPLICITI and to report any signs and symptoms associated with this event to their healthcare provider for evaluation [see Warnings and Precautions (5.4)].
- Manufactured by:
Bristol-Myers Squibb Company
Princeton, NJ 08543 USA
U.S. License No. 1713
-
PATIENT PACKAGE INSERT
Patient Information
EMPLICITI® (em-plis-city)
(elotuzumab)
for injection
EMPLICITI is used with other prescription medicines called REVLIMID® (lenalidomide) and dexamethasone or POMALYST® (pomalidomide) and dexamethasone. Read the Medication Guide that comes with REVLIMID if used with REVLIMID and POMALYST if used with POMALYST. You can ask your healthcare provider or pharmacist for information about dexamethasone.
What is EMPLICITI?
EMPLICITI is a prescription medicine used to treat:
- •
- multiple myeloma in combination with the medicines REVLIMID (lenalidomide) and dexamethasone in adults who have received one to three prior treatments for their multiple myeloma.
- •
- multiple myeloma in combination with the medicines POMALYST (pomalidomide) and dexamethasone in adults who have received at least two prior treatments including REVLIMID (lenalidomide) and a proteasome inhibitor.
It is not known if EMPLICITI is safe and effective in children.
Before you receive EMPLICITI, tell your healthcare provider about all of your medical conditions, including if you:
- •
- have an infection
- •
- are pregnant or plan to become pregnant. It is not known if EMPLICITI may harm your unborn baby. However, REVLIMID and POMALYST may cause birth defects or death of an unborn baby.
- o
- Before receiving EMPLICITI with REVLIMID and dexamethasone, or EMPLICITI with POMALYST and dexamethasone, females and males must agree to the instructions in the REVLIMID REMS® program for the use of EMPLICITI in combination with REVLIMID and dexamethasone and the POMALYST REMS® program for the use of EMPLICITI in combination with POMALYST and dexamethasone.
- o
- The REVLIMID REMS and POMALYST REMS programs have specific requirements about birth control (contraception), pregnancy testing, blood donation, and sperm donation that you need to know. Talk to your healthcare provider to learn more about REVLIMID or POMALYST.
- •
- are breastfeeding or plan to breastfeed. It is not known if EMPLICITI passes into breast milk. You should not breastfeed during treatment with EMPLICITI and REVLIMID and dexamethasone or EMPLICITI and POMALYST and dexamethasone.
- •
- Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
How will I receive EMPLICITI?
- •
- EMPLICITI will be given to you by intravenous (IV) infusion into your vein.
- •
- Your EMPLICITI treatment schedule is divided into cycles that are 28 days (4 weeks) long. A cycle includes the number of days you are on treatment and also the time you spend resting in between treatments.
- •
-
EMPLICITI with REVLIMID and dexamethasone is usually given as follows:
- o
- Cycles 1 and 2 (28 days per cycle), you will receive EMPLICITI one time every week.
- o
- Cycles 3 and up (28 days per cycle), you will receive EMPLICITI one time every 2 weeks.
- •
-
EMPLICITI with POMALYST and dexamethasone is usually given as follows:
- o
- Cycle 1 and 2 (28 days per cycle), you will receive EMPLICITI one time every week.
- o
- Cycle 3 and up (28 days per cycle), you will receive EMPLICITI one time every 4 weeks.
- •
- Your healthcare provider will decide how many treatments you will receive.
- •
- Before every EMPLICITI infusion, you will receive medicines to help reduce the risk of infusion reactions.
- •
- If you miss any appointments call your healthcare provider as soon as possible.
What are the possible side effects of EMPLICITI?
EMPLICITI may cause serious side effects, including:
- •
- Infusion reactions. Infusion reactions can happen during your infusion or within 24 hours after your infusion of EMPLICITI. Your healthcare provider will give you medicines before each infusion of EMPLICITI to help reduce the risk of an infusion reaction.
- If you have an infusion reaction while receiving EMPLICITI, your healthcare provider will slow or stop your infusion and treat your reaction. If you have a severe infusion reaction, your healthcare provider may stop your treatment completely. Tell your healthcare provider or get medical help right away if you have any of these symptoms after your infusion with EMPLICITI:
- o
- fever
- o
- chills
- o
- rash
- o
- chest pain
- o
- trouble breathing
- o
- dizziness
- o
- light-headedness
- •
- Infections. People with multiple myeloma who receive EMPLICITI with REVLIMID and dexamethasone or EMPLICITI with POMALYST and dexamethasone may develop infections that can be serious. Tell your healthcare provider right away if you have any signs and symptoms of an infection, including:
- o
- fever
- o
- flu-like symptoms
- o
- cough
- o
- shortness of breath
- o
- burning with urination
- o
- a painful skin rash
- •
- Risk of new cancers (malignancies). People with multiple myeloma who receive EMPLICITI with REVLIMID and dexamethasone or EMPLICITI with POMALYST and dexamethasone have a risk of developing new cancers. Talk with your healthcare provider about your risk of developing new cancers if you receive EMPLICITI. Your healthcare provider will check you for new cancers during your treatment with EMPLICITI.
- •
- Liver problems. EMPLICITI may cause liver problems. Your healthcare provider will do blood tests to check your liver during treatment with EMPLICITI. Tell your healthcare provider if you have signs and symptoms of liver problems, including: tiredness, weakness, loss of appetite, yellowing of your skin or eyes, color changes in your stools, confusion, or swelling of the stomach area.
The most common side effects of EMPLICITI when used with REVLIMID and dexamethasone include:
- •
- fatigue
- •
- diarrhea
- •
- fever
- •
- constipation
- •
- cough
- •
- numbness, weakness, tingling, or burning pain in your arms or legs
- •
- sore throat or runny nose
- •
- upper respiratory tract infection
- •
- decreased appetite
- •
- pneumonia
The most common side effects of EMPLICITI when used with POMALYST and dexamethasone include:
- •
- constipation
- •
- high blood sugar
These are not all of the possible side effects of EMPLICITI.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
You may also report side effects to Bristol-Myers Squibb at 1-800-721-5072.
General information about the safe and effective use of EMPLICITI
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet.
You can ask your pharmacist or healthcare provider for information about EMPLICITI that is written for health professionals.
What are the ingredients of EMPLICITI?
Active ingredient: elotuzumab
Inactive ingredients: citric acid monohydrate, polysorbate 80, sodium citrate, sucrose
For more information, call 1-844-EMPLICITI (844-367-5424) or visit EMPLICITI.com.
EMPLICITI® is a trademark of Bristol-Myers Squibb Company. All other trademarks are the property of their respective owners.
Manufactured by: Bristol-Myers Squibb Company, Princeton, NJ 08543 USA
U.S. License No. 1713
This Patient Information has been approved by the U.S. Food and Drug Administration.
Revised: 3/2022
- EMPLICITI 300 mg Representative Packaging
- EMPLICITI 400 mg Representative Packaging
-
INGREDIENTS AND APPEARANCE
EMPLICITI
elotuzumab injection, powder, lyophilized, for solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0003-2291 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ELOTUZUMAB (UNII: 1351PE5UGS) (ELOTUZUMAB - UNII:1351PE5UGS) ELOTUZUMAB 300 mg Inactive Ingredients Ingredient Name Strength CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) 2.44 mg POLYSORBATE 80 (UNII: 6OZP39ZG8H) 3.4 mg SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) 16.6 mg SUCROSE (UNII: C151H8M554) 510 mg Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0003-2291-11 1 in 1 CARTON 11/30/2015 1 1 in 1 VIAL, SINGLE-USE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA761035 11/30/2015 EMPLICITI
elotuzumab injection, powder, lyophilized, for solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0003-4522 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ELOTUZUMAB (UNII: 1351PE5UGS) (ELOTUZUMAB - UNII:1351PE5UGS) ELOTUZUMAB 400 mg Inactive Ingredients Ingredient Name Strength CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) 3.17 mg POLYSORBATE 80 (UNII: 6OZP39ZG8H) 4.4 mg SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) 21.5 mg SUCROSE (UNII: C151H8M554) 660 mg Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0003-4522-11 1 in 1 CARTON 11/30/2015 1 1 in 1 VIAL, SINGLE-USE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA761035 11/30/2015 Labeler - E.R. Squibb & Sons, L.L.C. (011550092)