Label: 4151 FIRST AID KIT kit
- NDC Code(s): 0498-0100-01, 0498-0730-01, 0498-2001-01, 0498-4151-01
- Packager: Honeywell Safety Products USA, INC
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated January 22, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
-
Warnings
For external use only- Obtain immediate medical treatment for all open wounds in or near the eyes. To avoid contamination, do not touch tip of container to any surface. Do not reuse. Once opened, discard.
Do not use
- if solution changes color or becomes cloudy
- if you have open wounds in or near the eyes, get medical help right away
- Directions
- Inactive Ingredients
- Questions?
- First Aid Burn Cream Active ingredient
- First Aid Burn Cream Purpose
- First Aid Burn Cream Uses
- First Aid Burn Cream Warnings
- First Aid Burn Cream Directions
- Other information
- Inactive ingredients
- Questions
- BZK Antiseptic Wipe Active ingredient
- BZK Purpose
- BZK Uses
-
BZK
Warnings
For external use only
BZK
Do not use- in the eyes or over large areas of the body
- on mucous membranes
- on irritated skin
- in case of deep puncture wounds, animal bites or serious burns, consult a doctor
- longer than 1 week unless directed by a doctor
- BZK Directions
- BZK Other information
- BZK Inactive ingredients
- BZK Questions
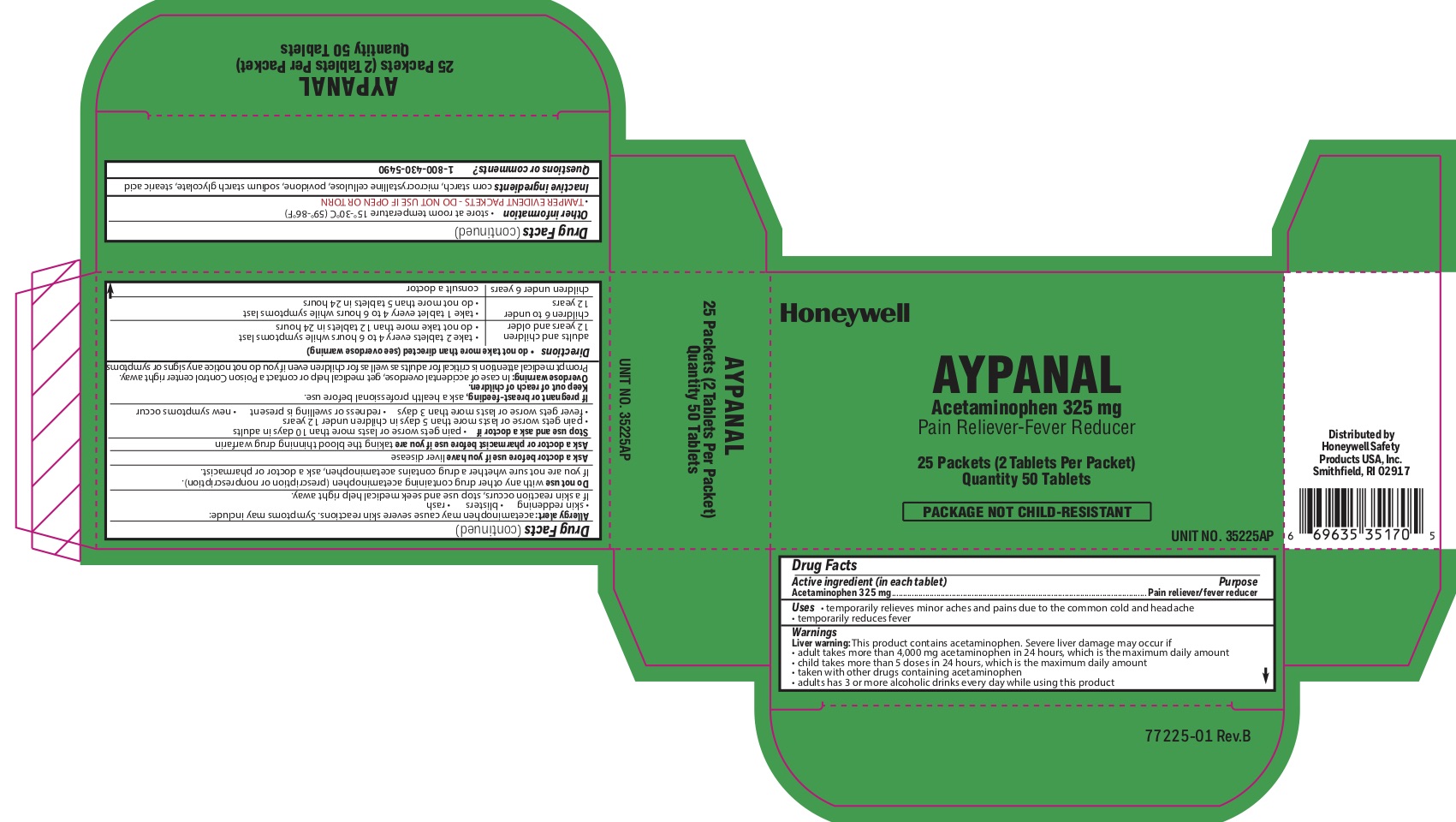
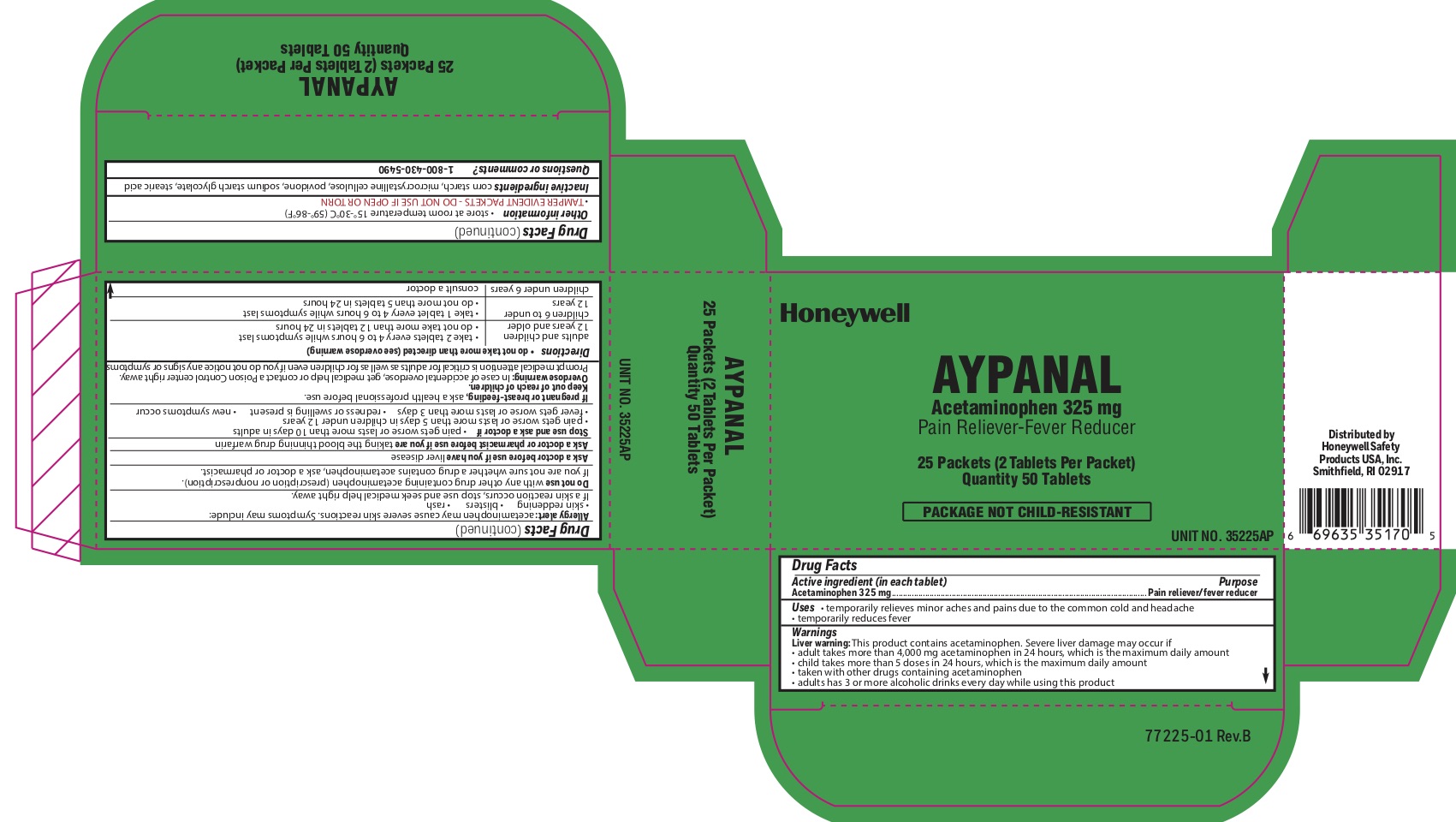
- Aypanal Active igredient
- Aypanal Purpose
- Aypanal Uses
-
Aypanal
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take: more than 4,000 mg in 24 hours, which is the maximum daily amount - child takes
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
skin reddening
blisters
rash -
Aypanal
Directions
- do not take more than directed (see overdose warning) adults and children 12 years of age or older
- take two tablets every 4-6 hours while symptoms last
- do not take more than directed (see overdose warning)
adults and children 12 years of age or older
- take two tablets every 4-6 hours while symptoms last
- do not take more than 12 tablets in 24 hours
- children 6 to under 12 years of age
- take 1 tablet every 4-6 hours while symptoms last
- do not take more than 5 tablets in 24 hours
- children under 6 years
- consult a doctor
- Aypanal Other information
- Aypanal Inactive ingredients
- Aypanal Questions or Comments?
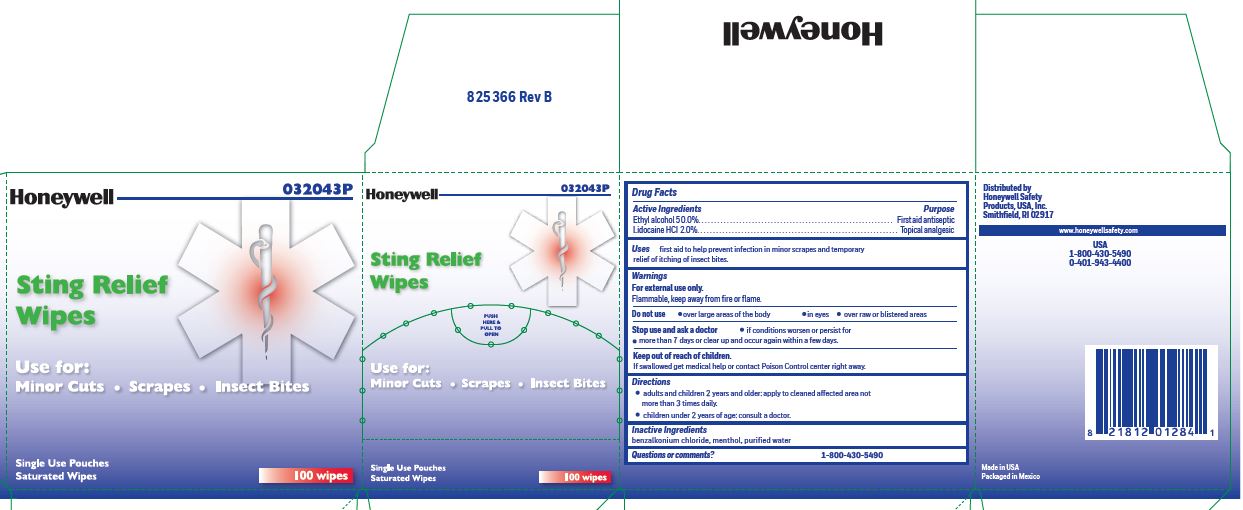
- Sting Relief Active ingredient
- Sting Relief Purpose
- Sting Relief Uses
- Sting Relief Warnings
- Sting Relief Directions
- Sting Relief Inactive ingredients
- Sting relief Questions or Comments
- Neomycin Antibiotic Ointment Active ingredient
- Neomycin Antibiotic Ointment Purpose
- Neomycin Antibiotic Ointment Uses
- Neomycin Antibiotic Ointment Warnings
- Neomycin Antibiotic Ointment Directions
- Neomycin Antibiotic Ointment Other information
- Neomycin Antibiotic Ointment Inactive ingredient
- Neomycin Antibiotic Ointment Questions
- Hand Sanitizer Active ingredient
- Hand Sanitizer Purpose
- Hand Sanitizer Uses
- Hand Sanitizer Warnings
- Hand Sanitizer Directions
- Hand Santitizer Other information
- Hand Sanitizer Inactive ingredients
- Hand Sanitizer Questions or Comments
-
4151
SF00004420 Kit Contents
1 EYE DRESS PKT W/4 ADH STRIPS
1 TWEEZER PLASTICS 4"
1 FIRST AID GUIDE ASHI
10 HAND SANITIZER 0.9G WJ BULK
2 GAUZE CLEAN-WRAP BDGE N/S 2"
1 GAUZE CLEAN-WRAP BDGE N/S 3"
2 ABD COMBINE PAD 5" X 9"
1 CPR FILTERSHIELD 77-100
1 BAGGED COMP MISC
1 1 OZ, BUFF EYEWASH
1 SCISSOR BDGE 4" RED PLS HDL
LBL STOCK 6-3/8"X4"
LBL STOCK 4"X2-7/8"
1 LBL STOCK 3"x1-7/8"
2 PR LRG NITRILE GLVES ZIP BAG
2 1" X 3" PLASTIC BANDS 16/BAG
2 TAPE ADHESIVE 1/2 X 2.5 125133
1 WATER-JEL BURN DRESSING 4 X 4
1 ADH BNDG PLASTIC EX-LG 4"X 2"
1 KIT, PP 16 UNIT FA
1 LBL CONTENTS ANSI 2015 CL A
1 TRI BNDG NON WOVEN 40"X40"X56"
1 COLD PACK UNIT 4"X6" BULK
4 GAUZE PADS 3"X3" 12PLY
3 WOVEN FINGERTIP BANDAGE 2"
2 WOVEN KNUCKLE BANDAGE
- Eye Wash Package label
- First Aid Burn Cream Principal Display Panel
- BZK Principal Display Panel
- Aypanal Principal Display Panel
- Sting Relief Principal Display Panel
- Neomycin Antibiotic Ointment Principal Display Panel
- Hand Sanitizer Principal Display Panel
- 4151 Kit Label SF00004420
-
INGREDIENTS AND APPEARANCE
4151 FIRST AID KIT
4151 first aid kit kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0498-4151 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-4151-01 1 in 1 KIT; Type 0: Not a Combination Product 10/18/2018 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BOTTLE 30 mL Part 2 3 PACKET 6 Part 3 6 POUCH 2.4 mL Part 4 10 PACKET 9 g Part 5 10 PACKET 9 g Part 6 10 PACKET 14 mL Part 7 10 PACKET 13 mL Part 1 of 7 EYESALINE EMERGENCY EYEWASH
purified water liquidProduct Information Item Code (Source) NDC:0498-0100 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength WATER (UNII: 059QF0KO0R) (WATER - UNII:059QF0KO0R) WATER 98.6 mL in 100 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM PHOSPHATE, DIBASIC (UNII: GR686LBA74) SODIUM PHOSPHATE, MONOBASIC, MONOHYDRATE (UNII: 593YOG76RN) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-0100-01 30 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M018 12/18/2018 Part 2 of 7 AYPANAL NON-ASPIRIN
acetaminophen tabletProduct Information Item Code (Source) NDC:0498-2001 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 325 mg Inactive Ingredients Ingredient Name Strength STEARIC ACID (UNII: 4ELV7Z65AP) SODIUM STARCH GLYCOLATE TYPE A CORN (UNII: AG9B65PV6B) STARCH, CORN (UNII: O8232NY3SJ) POVIDONE (UNII: FZ989GH94E) Product Characteristics Color white Score 2 pieces Shape ROUND Size 10mm Flavor Imprint Code circle;U Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-2001-01 2 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 04/10/2012 Part 3 of 7 STING RELIEF PAD
ethyl alcohol, lidocaine swabProduct Information Item Code (Source) NDC:0498-0733 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE HYDROCHLORIDE (UNII: V13007Z41A) (LIDOCAINE - UNII:98PI200987) LIDOCAINE HYDROCHLORIDE ANHYDROUS 20 mg in 1 mL ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 0.5 mL in 1 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) MENTHOL (UNII: L7T10EIP3A) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 0.4 mL in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 12/23/2017 Part 4 of 7 FIRST AID BURN
benzalkonium chloride, lidocaine hydrochloride creamProduct Information Item Code (Source) NDC:0498-0903 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 0.13 g in 100 g LIDOCAINE HYDROCHLORIDE (UNII: V13007Z41A) (LIDOCAINE - UNII:98PI200987) LIDOCAINE HYDROCHLORIDE 0.5 g in 100 g Inactive Ingredients Ingredient Name Strength PROPYLENE GLYCOL (UNII: 6DC9Q167V3) ALOE VERA LEAF (UNII: ZY81Z83H0X) WATER (UNII: 059QF0KO0R) STEARIC ACID (UNII: 4ELV7Z65AP) METHYLPARABEN (UNII: A2I8C7HI9T) CETYL ALCOHOL (UNII: 936JST6JCN) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) PEG-100 STEARATE (UNII: YD01N1999R) LIGHT MINERAL OIL (UNII: N6K5787QVP) EDETATE DISODIUM (UNII: 7FLD91C86K) TROLAMINE (UNII: 9O3K93S3TK) GLYCERIN (UNII: PDC6A3C0OX) PROPYLPARABEN (UNII: Z8IX2SC1OH) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 0.9 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 12/20/2017 Part 5 of 7 NEOMYCIN
antibiotic ointmentProduct Information Item Code (Source) NDC:0498-0730 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NEOMYCIN SULFATE (UNII: 057Y626693) (NEOMYCIN - UNII:I16QD7X297) NEOMYCIN SULFATE 3.5 mg in 1 g Inactive Ingredients Ingredient Name Strength PETROLATUM (UNII: 4T6H12BN9U) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-0730-01 0.9 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 03/31/2010 Part 6 of 7 ANTISEPTIC TOWELETTE
benzalkonium chloride liquidProduct Information Item Code (Source) NDC:0498-0501 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 1.3 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 1.4 mL in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 12/21/2017 Part 7 of 7 INSTANT HAND SANITIZER
alcohol liquidProduct Information Item Code (Source) NDC:59898-420 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 62 mL in 100 mL Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) TRIISOPROPANOLAMINE (UNII: W9EN9DLM98) CARBOMER COPOLYMER TYPE A (UNII: 71DD5V995L) WATER (UNII: 059QF0KO0R) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 1.3 mL in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 04/15/2011 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 10/18/2018 Labeler - Honeywell Safety Products USA, INC (118768815)