BASIC CARE DAYTIME NIGHTTIME SEVERE COLD AND FLU- acetaminophen, dextromethorphan hbr, doxylamine succinate, guaifenesin, phenylephrine hcl
L. Perrigo Company
----------
Amazon daytime nighttime severe cold & flu Drug Facts
Active ingredients (in each caplet) - Nighttime
Acetaminophen 325 mg
Dextromethorphan HBr 10 mg
Doxylamine succinate 6.25 mg
Phenylephrine HCl 5 mg
Active ingredients (in each caplet) - Daytime
Acetaminophen 325 mg
Dextromethorphan HBr 10 mg
Guaifenesin 200 mg
Phenylephrine HCl 5 mg
Uses - Nighttime
- •
- temporarily relieves common cold/flu symptoms:
- •
- nasal congestion
- •
- sinus congestion and pressure
- •
- cough due to minor throat and bronchial irritation
- •
- cough to help you sleep
- •
- minor aches and pains
- •
- headache
- •
- fever
- •
- sore throat
- •
- runny nose and sneezing
- •
- reduces swelling of nasal passages
- •
- temporarily restores freer breathing through the nose
- •
- promotes nasal and/or sinus drainage
Uses - Daytime
- •
- temporarily relieves common cold/flu symptoms:
- •
- nasal congestion
- •
- sinus congestion and pressure
- •
- cough due to minor throat and bronchial irritation
- •
- minor aches and pains
- •
- headache
- •
- fever
- •
- sore throat
- •
- reduces swelling of nasal passages
- •
- temporarily restores freer breathing through the nose
- •
- promotes nasal and/or sinus drainage
- •
- helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
- •
- more than 4,000 mg of acetaminophen in 24 hours
- •
- with other drugs containing acetaminophen
- •
- 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- •
- skin reddening
- •
- blisters
- •
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Sore throat warning: If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
Do not use
- •
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- •
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- •
- if you have ever had an allergic reaction to this product or any of its ingredients
Ask a doctor before use if you have - Nighttime
- •
- liver disease
- •
- heart disease
- •
- high blood pressure
- •
- thyroid disease
- •
- diabetes
- •
- glaucoma
- •
- cough that occurs with too much phlegm (mucus)
- •
- a breathing problem such as emphysema or chronic bronchitis
- •
- persistent or chronic cough such as occurs with smoking, asthma, or emphysema
- •
- trouble urinating due to an enlarged prostate gland
Ask a doctor before use if you have - Daytime
- •
- liver disease
- •
- heart disease
- •
- high blood pressure
- •
- thyroid disease
- •
- diabetes
- •
- trouble urinating due to an enlarged prostate gland
- •
- cough that occurs with too much phlegm (mucus)
- •
- persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
Ask a doctor or pharmacist before use if you are - Nighttime
- •
- taking sedatives or tranquilizers
- •
- taking the blood thinning drug warfarin
When using this product - Nighttime
- •
- do not use more than directed
- •
- excitability may occur, especially in children
- •
- marked drowsiness may occur
- •
- avoid alcoholic drinks
- •
- be careful when driving a motor vehicle or operating machinery
- •
- alcohol, sedatives, and tranquilizers may increase drowsiness
Stop use and ask a doctor if - Nighttime
- •
- you get nervous, dizzy or sleepless
- •
- pain, nasal congestion, or cough gets worse or lasts more than 7 days
- •
- fever gets worse or lasts more than 3 days
- •
- redness or swelling is present
- •
- new symptoms occur
- •
- cough comes back or occurs with rash or headache that lasts.
These could be signs of a serious condition.
Stop use and ask a doctor if - Daytime
- •
- you get nervous, dizzy or sleepless
- •
- pain, nasal congestion, or cough gets worse or lasts more than 7 days
- •
- fever gets worse or lasts more than 3 days
- •
- redness or swelling is present
- •
- new symptoms occur
- •
- cough comes back or occurs with rash or headache that lasts.
These could be signs of a serious condition.
Keep out of reach of children.
Overdose warning: In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222). Quick medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
Directions – Nighttime and Daytime
- •
- take only as directed - see overdose warning
- •
- do not exceed 4 doses per 24 hrs
|
adults & children 12 yrs & over |
2 caplets with water every 4 hrs |
|
children 4 to under 12 yrs |
ask a doctor |
|
children under 4 yrs |
do not use |
Inactive ingredients - Nighttime
crospovidone, D&C yellow no. 10 aluminum lake, FD&C blue no. 1 aluminum lake, FD&C blue no. 2 aluminum lake, FD&C yellow no. 6 aluminum lake, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, povidone, pregelatinized starch, silicon dioxide, stearic acid, talc, titanium dioxide
Inactive ingredients - Daytime
croscarmellose sodium, crospovidone, FD&C blue #2 aluminum lake, FD&C red #40 aluminum lake, maltodextrin, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, povidone, pregelatinized starch, silicon dioxide, stearic acid, talc, titanium dioxide
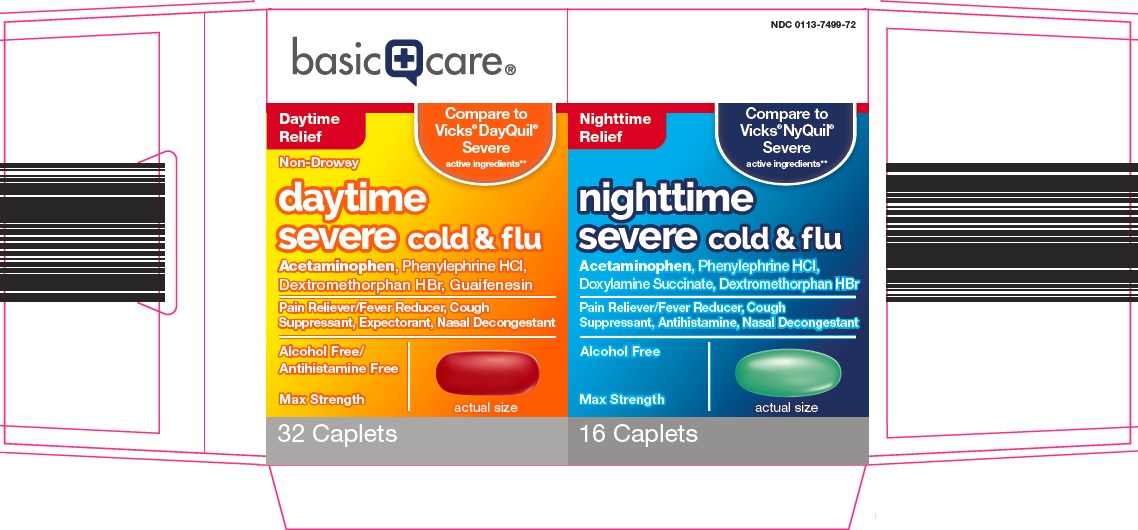
Package/Label Principal Display Panel
Daytime Relief
Compare to Vicks® DayQuil® Severe active ingredients
Non-Drowsy
daytime
severe cold & flu
Acetaminophen, Phenylephrine HCl,
Dextromethorphan HBr, Guaifenesin
Pain Reliever/Fever Reducer, Cough Suppressant, Expectorant, Nasal Decongestant
Alcohol Free/Antihistamine Free
Max Strength
actual size
32 Caplets
Nighttime Relief
Compare to Vicks® NyQuil® Severe active ingredients
nighttime
severe cold & flu
Acetaminophen, Phenylephrine HCl,
Doxylamine Succinate, Dextromethorphan HBr
Pain Reliever/Fever Reducer, Cough Suppressant, Antihistamine, Nasal Decongestant
Alcohol Free
Max Strength
actual size
16 Caplets
| BASIC CARE DAYTIME NIGHTTIME SEVERE COLD AND FLU
acetaminophen, dextromethorphan hbr, doxylamine succinate, guaifenesin, phenylephrine hcl kit |
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
| Labeler - L. Perrigo Company (006013346) |