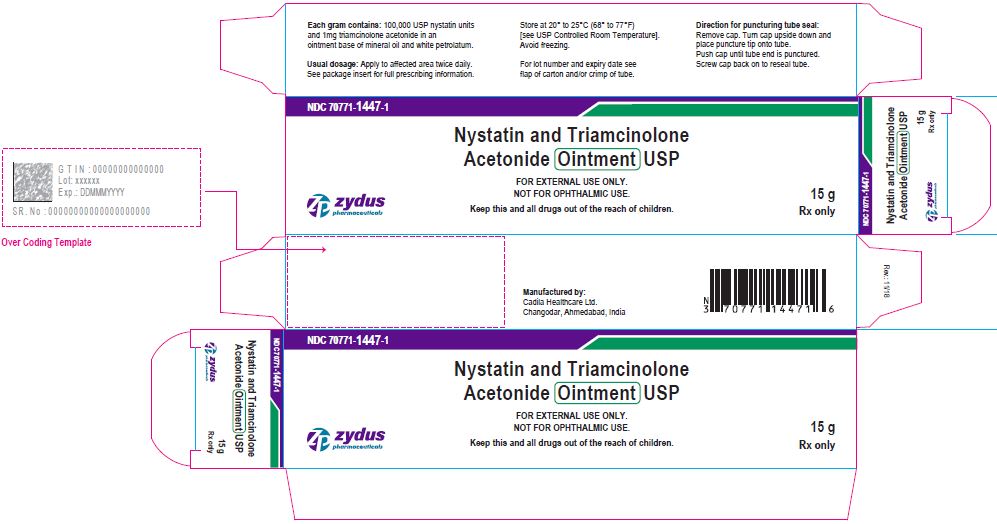
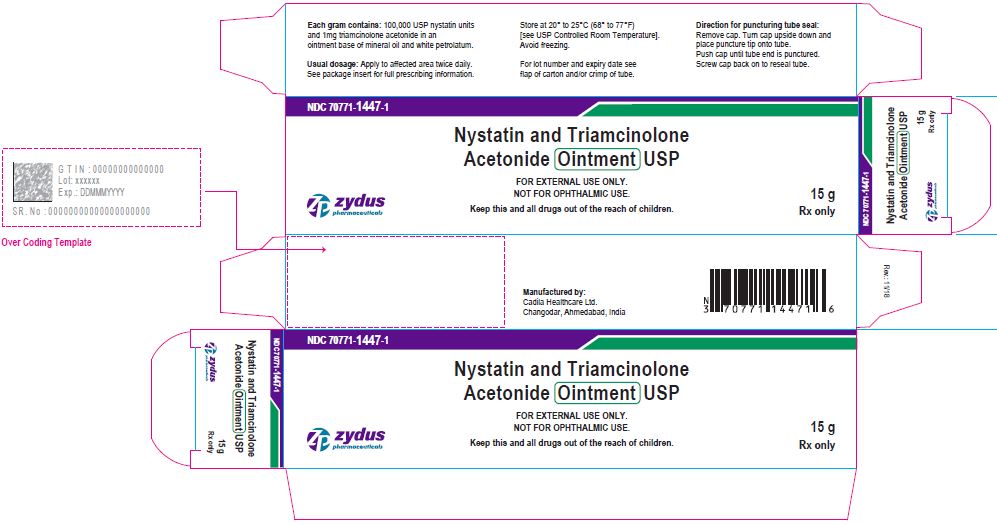
Label: NYSTATIN AND TRIAMCINOLONE ACETONIDE ointment
- NDC Code(s): 70771-1447-1, 70771-1447-2, 70771-1447-3
- Packager: Zydus Lifesciences Limited
- Category: HUMAN PRESCRIPTION DRUG LABEL
Drug Label Information
Updated November 5, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
NYSTATIN AND TRIAMCINOLONE ACETONIDE
nystatin and triamcinolone acetonide ointmentProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70771-1447 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NYSTATIN (UNII: BDF1O1C72E) (NYSTATIN - UNII:BDF1O1C72E) NYSTATIN 100000 [USP'U] in 1 g TRIAMCINOLONE ACETONIDE (UNII: F446C597KA) (TRIAMCINOLONE ACETONIDE - UNII:F446C597KA) TRIAMCINOLONE ACETONIDE 1 mg in 1 g Inactive Ingredients Ingredient Name Strength MINERAL OIL (UNII: T5L8T28FGP) PETROLATUM (UNII: 4T6H12BN9U) Product Characteristics Color YELLOW (YELLOW) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70771-1447-1 1 in 1 CARTON 11/20/2018 1 15 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:70771-1447-2 1 in 1 CARTON 11/20/2018 2 30 g in 1 TUBE; Type 0: Not a Combination Product 3 NDC:70771-1447-3 1 in 1 CARTON 11/20/2018 3 60 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA207764 11/20/2018 Labeler - Zydus Lifesciences Limited (918596198) Registrant - Zydus Lifesciences Limited (650650802) Establishment Name Address ID/FEI Business Operations Zydus Lifesciences Limited 650650802 ANALYSIS(70771-1447) , MANUFACTURE(70771-1447)