PHENOXYBENZAMINE HYDROCHLORIDE- phenoxybenzamine hydrochloride capsule
Hikma Pharmaceuticals USA Inc.
----------
Phenoxybenzamine Hydrochloride Capsules, USP
DESCRIPTION
Each Phenoxybenzamine Hydrochloride Capsule, USP contains 10 mg of phenoxybenzamine hydrochloride, USP. Inactive ingredients consist of anhydrous lactose, black monogramming ink, colloidal silicon dioxide and sodium lauryl sulfate. The black monogramming ink contains ammonium hydroxide, iron oxide black, isopropyl alcohol, N-butyl alcohol, propylene glycol, and shellac. The capsule shells also contain FD&C Red No. 40, gelatin, purified water and titanium dioxide.
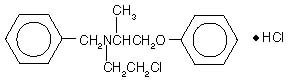
Phenoxybenzamine is N-(2-Chloroethyl)-N-(1-methyl-2-phenoxyethyl)benzylamine hydrochloride:
Phenoxybenzamine hydrochloride is a white to almost white powder with a molecular weight of 340.3, which melts between 136° and 141°C. It is soluble in alcohol and chloroform; insoluble in ether and water.
CLINICAL PHARMACOLOGY
Phenoxybenzamine hydrochloride is a long-acting, adrenergic, alpha-receptor blocking agent, which can produce and maintain “chemical sympathectomy” by oral administration. It increases blood flow to the skin, mucosa and abdominal viscera, and lowers both supine and erect blood pressures. It has no effect on the parasympathetic system.
Twenty to 30 percent of orally administered phenoxybenzamine appears to be absorbed in the active form.1
The half-life of orally administered phenoxybenzamine hydrochloride is not known; however, the half-life of intravenously administered drug is approximately 24 hours. Demonstrable effects with intravenous administration persist for at least 3 to 4 days, and the effects of daily administration are cumulative for nearly a week.1
INDICATIONS AND USAGE
Phenoxybenzamine hydrochloride capsules are indicated in the treatment of pheochromocytoma, to control episodes of hypertension and sweating. If tachycardia is excessive, it may be necessary to use a beta-blocking agent concomitantly.
CONTRAINDICATIONS
Conditions where a fall in blood pressure may be undesirable; hypersensitivity to the drug or any of its components.
WARNINGS
Phenoxybenzamine-induced alpha-adrenergic blockade leaves beta-adrenergic receptors unopposed. Compounds that stimulate both types of receptors may, therefore, produce an exaggerated hypotensive response and tachycardia.
PRECAUTIONS
General
Administer with caution in patients with marked cerebral or coronary arteriosclerosis or renal damage. Adrenergic blocking effect may aggravate symptoms of respiratory infections.
Drug Interactions2
Phenoxybenzamine hydrochloride may interact with compounds that stimulate both alpha-and beta-adrenergic receptors (i.e., epinephrine) to produce an exaggerated hypotensive response and tachycardia. (See WARNINGS.)
Phenoxybenzamine blocks hyperthermia production by levarterenol, and blocks hypothermia production by reserpine.
Carcinogenesis and Mutagenesis
Case reports of carcinoma in humans after long-term treatment with phenoxybenzamine have been reported. Hence long-term use of phenoxybenzamine is not recommended.3, 4 Carefully weigh the benefits and risks before prescribing this drug.
Phenoxybenzamine hydrochloride showed in vitro mutagenic activity in the Ames test and mouse lymphoma assay; it did not show mutagenic activity in vivo in the micronucleus test in mice. In rats and mice, repeated intraperitoneal administration of phenoxybenzamine hydrochloride (three times per week for up to 52 weeks) resulted in peritoneal sarcomas. Chronic oral dosing in rats (for up to 2 years) produced malignant tumors of the small intestine and non-glandular stomach, as well as ulcerative and/or erosive gastritis of the glandular stomach. Whereas squamous cell carcinomas of the non-glandular stomach were observed at all tested doses of phenoxybenzamine hydrochloride, there was a no-observed-effect-level of 10 mg/kg for tumors (carcinomas and sarcomas) of the small intestine. This dose is, on a body surface area basis, about twice the maximum recommended human dosage of 20 mg b.i.d.
Pregnancy
Teratogenic Effects:
Pregnancy Category C:
Adequate reproductive studies in animals have not been performed with phenoxybenzamine hydrochloride. It is also not known whether phenoxybenzamine can cause fetal harm when administered to a pregnant woman. Phenoxybenzamine should be given to a pregnant woman only if clearly needed.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, and because of the potential for serious adverse reactions from phenoxybenzamine hydrochloride, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
ADVERSE REACTIONS
The following adverse reactions have been observed, but there are insufficient data to support an estimate of their frequency.
Autonomic Nervous System*: Postural hypotension, tachycardia, inhibition of ejaculation, nasal congestion, miosis.
*These so-called “side effects” are actually evidence of adrenergic blockade and vary according to the degree of blockade.
Miscellaneous: Gastrointestinal irritation, drowsiness, fatigue.
OVERDOSAGE
SYMPTOMS - These are largely the result of blocking of the sympathetic nervous system and of the circulating epinephrine. They may include postural hypotension, resulting in dizziness or fainting; tachycardia, particularly postural; vomiting; lethargy; shock.
TREATMENT
When symptoms and signs of overdosage exist, discontinue the drug. Treatment of circulatory failure, if present, is a prime consideration. In cases of mild overdosage, recumbent position with legs elevated usually restores cerebral circulation. In the more severe cases, the usual measures to combat shock should be instituted. Usual pressor agents are not effective. Epinephrine is contraindicated because it stimulates both alpha- and beta- receptors; since alpha- receptors are blocked, the net effect of epinephrine administration is vasodilation and a further drop in blood pressure (epinephrine reversal).
The patient may have to be kept flat for 24 hours or more in the case of overdose, as the effect of the drug is prolonged. Leg bandages and an abdominal binder may shorten the period of disability.
I.V. Infusion of levarterenol bitartrate** may be used to combat severe hypotensive reactions, because it stimulates alpha- receptors primarily. Although phenoxybenzamine hydrochloride is an alpha-adrenergic blocking agent, a sufficient dose of levarterenol bitartrate will overcome this effect.
The oral LD50 for phenoxybenzamine hydrochloride is approximately 2000 mg/kg in rats and approximately 500 mg/kg in guinea pigs.
DOSAGE AND ADMINISTRATION
The dosage should be adjusted to fit the needs of each patient. Small initial doses should be slowly increased until the desired effect is obtained or the side effects from blockade become troublesome. After each increase, the patient should be observed on that level before instituting another increase. The dosage should be carried to a point where symptomatic relief and/or objective improvement are obtained, but not so high that the side effects from blockade become troublesome.
Initially, 10 mg of phenoxybenzamine hydrochloride twice a day. Dosage should be increased every other day, usually to 20 to 40 mg 2 or 3 times a day, until an optimal dosage is obtained, as judged by blood pressure control.
Long-term use of phenoxybenzamine is not recommended (see PRECAUTIONS: Carcinogenesis and Mutagenesis).
HOW SUPPLIED
Phenoxybenzamine Hydrochloride Capsules, USP
10 mg capsule is supplied as a red opaque/red opaque capsule with “54 036” printed in black on the cap and “54 036” printed in black ink on the capsule body, containing a white to off-white powder.
NDC 0054-0349-25: Bottle of 100 Capsules
REFERENCES
- 1.
- Weiner, N.: Drugs That Inhibit Adrenergic Nerves and Block Adrenergic Receptors, in Goodman, L., and Gilman, A., The Pharmacological Basis of Therapeutics, ed. 6, New York, Macmillan Publishing Co., 1980, p. 179; p. 182.
- 2.
- Martin, E.W.: Drug Interactions Index 1978/1979, Philadelphia, J.B. Lippincott Co., 1978, pp. 209-210.
- 3.
- Nettesheim O, Hoffken G, Gahr M, Breidert M: Haematemesis and dysphagia in a 20-year-old woman with congenital spine malformation and situs inversus partialis [German]. Zeitschrift fur Gastroenterologie. 2003;41(4):319-24. 165
- 4.
- Vaidyanathan S, Mansour P, Soni BM, Hughes PL, Singh G: Chronic lymphocytic leukaemia, synchronous small cell carcinoma and squamous neoplasia of the urinary bladder in a paraplegic man following long-term phenoxybenzamine therapy. Spinal Cord. 2006;44(3):188-91.
- ** Available as Levophed® Bitartrate (brand of norepinephrine bitartrate) from Abbott Laboratories.
- Distr. by: Hikma
- Pharmaceuticals USA Inc.
- Berkeley Heights, NJ 07922
C50000639/01
Revised September 2020
| PHENOXYBENZAMINE HYDROCHLORIDE
phenoxybenzamine hydrochloride capsule |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Hikma Pharmaceuticals USA Inc. (080189610) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| West-Ward Columbus Inc. | 058839929 | MANUFACTURE(0054-0349) | |