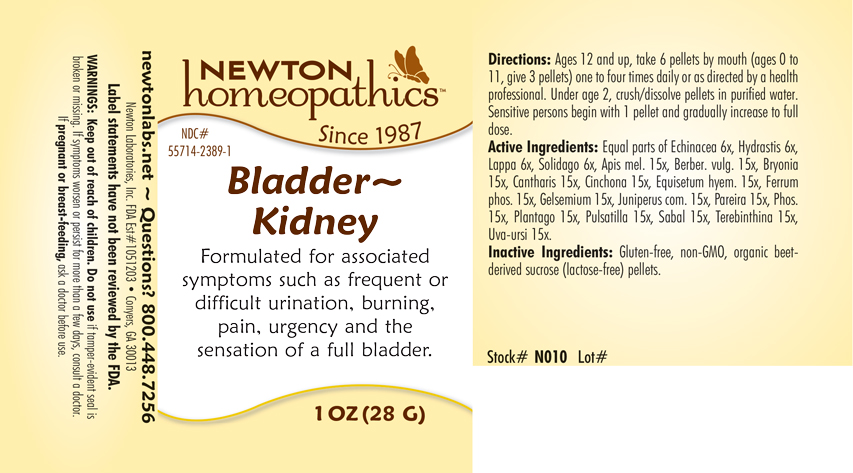
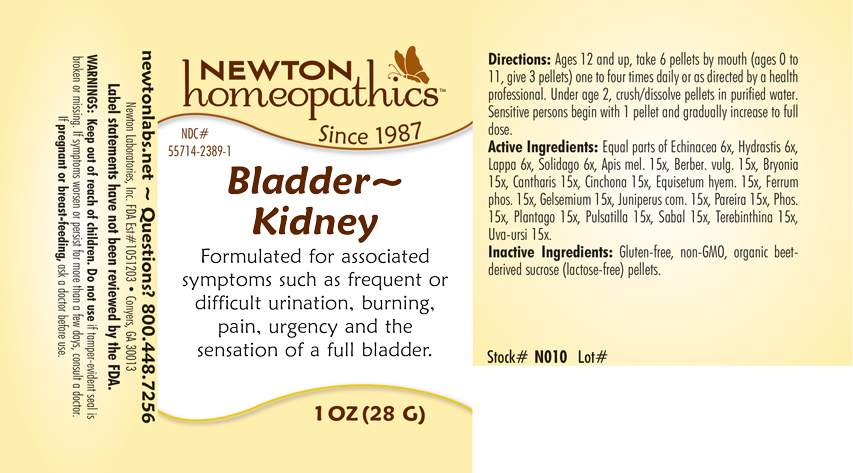
Label: BLADDER - KIDNEY- apis mel., berber. vulg., bryonia, cantharis, cinchona, equisetum hyemale, ferrum phosphoricum, gelsemium, juniperus com., pareira, phosphorus, plantago, pulsatilla, sabal, terebinthina, uva-ursi, echinacea, hydrastis, lappa, solidago pellet
- NDC Code(s): 55714-2389-1
- Packager: Newton Laboratories, Inc.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated November 24, 2020
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- OTC - KEEP OUT OF REACH OF CHILDREN SECTION
- OTC - PREGNANCY OR BREAST FEEDING SECTION
- WARNINGS SECTION
-
QUESTIONS SECTION
newtonlabs.net ~ Questions? 800.448.7256
Newton Laboratories, Inc. FDA Est # 1051203 - Conyers, GA 30013
- INACTIVE INGREDIENT SECTION
- OTC - PURPOSE SECTION
-
OTC - ACTIVE INGREDIENT SECTION
Equal parts of Echinacea 6x, Hydrastis 6x, Lappa 6x, Solidago 6x, Apis mel. 15x, Berber. vulg. 15x, Bryonia 15x, Cantharis 15x, Cinchona 15x, Equisetum hyem. 15x, Ferrum phos. 15x, Gelsemium 15x, Juniperus com. 15x, Pareira 15x, Phos. 15x, Plantago 15x, Pulsatilla 15x, Sabal 15x, Terebinthina 15x, Uva-ursi 15x.
- DOSAGE & ADMINISTRATION SECTION
- INDICATIONS & USAGE SECTION
- PACKAGE LABEL
-
INGREDIENTS AND APPEARANCE
BLADDER - KIDNEY
apis mel., berber. vulg., bryonia, cantharis, cinchona, equisetum hyemale, ferrum phosphoricum, gelsemium, juniperus com., pareira, phosphorus, plantago, pulsatilla, sabal, terebinthina, uva-ursi, echinacea, hydrastis, lappa, solidago pelletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:55714-2389 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength APIS MELLIFERA (UNII: 7S82P3R43Z) (APIS MELLIFERA - UNII:7S82P3R43Z) APIS MELLIFERA 15 [hp_X] in 1 g BERBERIS VULGARIS ROOT BARK (UNII: 1TH8Q20J0U) (BERBERIS VULGARIS ROOT BARK - UNII:1TH8Q20J0U) BERBERIS VULGARIS ROOT BARK 15 [hp_X] in 1 g BRYONIA ALBA ROOT (UNII: T7J046YI2B) (BRYONIA ALBA ROOT - UNII:T7J046YI2B) BRYONIA ALBA ROOT 15 [hp_X] in 1 g LYTTA VESICATORIA (UNII: 3Q034RO3BT) (LYTTA VESICATORIA - UNII:3Q034RO3BT) LYTTA VESICATORIA 15 [hp_X] in 1 g CINCHONA OFFICINALIS BARK (UNII: S003A158SB) (CINCHONA OFFICINALIS BARK - UNII:S003A158SB) CINCHONA OFFICINALIS BARK 15 [hp_X] in 1 g EQUISETUM HYEMALE (UNII: 59677RXH25) (EQUISETUM HYEMALE - UNII:59677RXH25) EQUISETUM HYEMALE 15 [hp_X] in 1 g FERROSOFERRIC PHOSPHATE (UNII: 91GQH8I5F7) (FERROSOFERRIC PHOSPHATE - UNII:91GQH8I5F7) FERROSOFERRIC PHOSPHATE 15 [hp_X] in 1 g GELSEMIUM SEMPERVIRENS ROOT (UNII: 639KR60Q1Q) (GELSEMIUM SEMPERVIRENS ROOT - UNII:639KR60Q1Q) GELSEMIUM SEMPERVIRENS ROOT 15 [hp_X] in 1 g JUNIPER BERRY (UNII: O84B5194RL) (JUNIPER BERRY - UNII:O84B5194RL) JUNIPER BERRY 15 [hp_X] in 1 g CHONDRODENDRON TOMENTOSUM ROOT (UNII: 395A3P448Z) (CHONDRODENDRON TOMENTOSUM ROOT - UNII:395A3P448Z) CHONDRODENDRON TOMENTOSUM ROOT 15 [hp_X] in 1 g PHOSPHORUS (UNII: 27YLU75U4W) (PHOSPHORUS - UNII:27YLU75U4W) PHOSPHORUS 15 [hp_X] in 1 g PLANTAGO MAJOR (UNII: W2469WNO6U) (PLANTAGO MAJOR - UNII:W2469WNO6U) PLANTAGO MAJOR 15 [hp_X] in 1 g PULSATILLA VULGARIS (UNII: I76KB35JEV) (PULSATILLA VULGARIS - UNII:I76KB35JEV) PULSATILLA VULGARIS 15 [hp_X] in 1 g SAW PALMETTO (UNII: J7WWH9M8QS) (SAW PALMETTO - UNII:J7WWH9M8QS) SAW PALMETTO 15 [hp_X] in 1 g TURPENTINE (UNII: XJ6RUH0O4G) (TURPENTINE - UNII:XJ6RUH0O4G) TURPENTINE 15 [hp_X] in 1 g ARCTOSTAPHYLOS UVA-URSI LEAF (UNII: 3M5V3D1X36) (ARCTOSTAPHYLOS UVA-URSI LEAF - UNII:3M5V3D1X36) ARCTOSTAPHYLOS UVA-URSI LEAF 15 [hp_X] in 1 g ECHINACEA, UNSPECIFIED (UNII: 4N9P6CC1DX) (ECHINACEA, UNSPECIFIED - UNII:4N9P6CC1DX) ECHINACEA, UNSPECIFIED 6 [hp_X] in 1 g GOLDENSEAL (UNII: ZW3Z11D0JV) (GOLDENSEAL - UNII:ZW3Z11D0JV) GOLDENSEAL 6 [hp_X] in 1 g ARCTIUM LAPPA ROOT (UNII: 597E9BI3Z3) (ARCTIUM LAPPA ROOT - UNII:597E9BI3Z3) ARCTIUM LAPPA ROOT 6 [hp_X] in 1 g SOLIDAGO VIRGAUREA FLOWERING TOP (UNII: 5405K23S50) (SOLIDAGO VIRGAUREA FLOWERING TOP - UNII:5405K23S50) SOLIDAGO VIRGAUREA FLOWERING TOP 6 [hp_X] in 1 g Inactive Ingredients Ingredient Name Strength SUCROSE (UNII: C151H8M554) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55714-2389-1 28 g in 1 BOTTLE, GLASS; Type 0: Not a Combination Product 10/30/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 10/30/2018 Labeler - Newton Laboratories, Inc. (788793610) Registrant - Newton Laboratories, Inc. (788793610) Establishment Name Address ID/FEI Business Operations Newton Laboratories, Inc. 788793610 manufacture(55714-2389)