VANTAS- histrelin acetate implant
Endo Pharmaceuticals Inc.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use VANTAS® safely and effectively. See full prescribing information for VANTAS®.
VANTAS® (histrelin acetate) subcutaneous implant Initial U.S. Approval: 2004 RECENT MAJOR CHANGESINDICATIONS AND USAGEVANTAS is a gonadotropin releasing hormone (GnRH) agonist indicated for the palliative treatment of advanced prostate cancer. (1). DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHSSubcutaneous implant: 50 mg histrelin acetate (3). CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
ADVERSE REACTIONS
To report SUSPECTED ADVERSE REACTIONS, contact Endo Pharmaceuticals Solutions Inc. at 1-800-462-3636 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling. Revised: 12/2020 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
VANTAS is indicated for the palliative treatment of advanced prostate cancer.
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dose
The recommended dose of VANTAS is one implant for 12 months.
Each implant contains 50 mg histrelin acetate to deliver 41 mg histrelin.
The implant is inserted subcutaneously in the inner aspect of the upper arm and provides continuous release of histrelin (50 mcg/day) for 12 months of hormonal therapy.
VANTAS should be removed after 12 months of therapy (the implant has been designed to allow for a few additional weeks of histrelin release, in order to allow flexibility of medical appointments). At the time an implant is removed, another implant may be inserted to continue therapy.
In patients treated with gonadotropin releasing hormone (GnRH) analogs for prostate cancer, treatment is usually continued upon development of non-metastatic and metastatic castration-resistant prostate cancer.
2.2 Recommended Procedure for Implant Insertion and Removal
This procedure section is intended to provide guidance for the insertion and removal of VANTAS.
Insertion of a new implant can proceed using the following Insertion Procedure. If a previous VANTAS implant must first be removed, please see the Removal Procedure instructions below.
Insertion Procedure
Many of the supplies necessary to insert the implant, including the Insertion Tool and local anesthetic, are provided in a separate Implantation Kit that is shipped along with the implant. Please note that the implant, in its sealed vial, pouch, and carton, must be kept refrigerated (2°C to 8°C) until needed for the procedure. Once removed from refrigeration, the vial containing the implant (still in its unopened pouch and carton) may remain at room temperature for up to 7 days, if necessary, before being used. If not used in that time, the packaged implant may again be properly refrigerated until the expiration date on the carton.
NOTE: The Implantation Kit is to be stored at 20°C to 25°C (68°F to 77°F) [See USP Controlled Room Temperature] only.
Insertion of the VANTAS implant is a surgical procedure. Sterile gloves and aseptic technique must be used to minimize any chance of infection.
1. Setting up the Sterile Field
Using proper aseptic technique, the sterilized components of the Implantation Kit needed for the insertion procedure, including the Insertion Tool, are to be carefully dispensed from their packaging onto the Sterile Field drape (non-fenestrated) provided. NOTE THAT THE KIT BOX AND ALL PACKAGING, INCLUDING THE SURFACE OF THE VIALS, ARE NOT STERILE and should be kept off of the Sterile Field drape.
The implant vial should not be opened until just before the time of insertion. Open the vial by removing the metal band and carefully pour the sterile contents (implant and sterile saline) onto the Sterile Field drape. The implant can then be handled with sterile gloves or with the sterile mosquito clamp provided. AVOID bending or pinching the implant.
2. Preparing the Patient and the Insertion Site
The patient should be on his back, ideally with the arm least used (e.g., left arm for a right-handed person) positioned either bent or extended so that the physician has ready access to the inner aspect of the upper arm. Propping the arm with pillows may help the patient more easily hold the position. The suggested optimum site for subcutaneous insertion is approximately half-way between the shoulder and the elbow, in line with the crease between the biceps and triceps muscles.
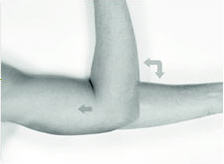
Antiseptic
Swab the insertion area with topical antiseptic, then overlay with the fenestrated Sterile Field drape provided, so that the opening is over the insertion site (for clarity of illustration, the following images do not show the drape).

Anesthetic
The method of anesthesia utilized (i.e., local, conscious sedation, general) is at the discretion of the healthcare provider.
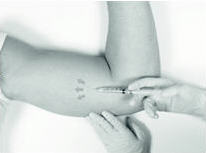
If local anesthesia is selected: a vial of a local anesthetic (e.g., lidocaine HCl 1% with epinephrine or lidocaine HCl 1%)(note that the exterior of the vial is not sterile) has been provided along with a sterile hypodermic needle for injection. After determining the absence of known allergies to the anesthetic agent, inject anesthetic into the subcutaneous tissue, starting at the planned incision site, then infiltrating along the intended subcutaneous insertion path, up to the length of the implant (a little more than one inch).
3. Loading the Insertion Tool
The sterile Insertion Tool is comprised of a fixed handle attached to a retractable, bevel-tipped cannula, into the chamber of which the implant is placed for subcutaneous insertion. Inside the fully extended cannula, up to the level of the black marking, is a fixed piston upon which the implant rests. During the final step of the insertion procedure, the cannula will be retracted into the handle using the slide mechanism (green button), thereby exposing and leaving the implant to remain in the subcutaneous tissue.

When first grasping the sterile Insertion Tool, confirm that the cannula is fully extended. Verify this by inspecting the position of the green retraction button. The button should be locked in position all the way forward, towards the cannula, farthest from the handle.

The implant can be picked up using sterile gloves or with the sterile mosquito clamp provided. Avoid bending or pinching the implant. Note that the implant may come out of its vial slightly curved and/or partially flattened after refrigerated storage.
To help make the implant more symmetrical prior to loading into the Tool, you can roll the implant a few times (using a sterile glove) between the fingers and thumb.
Insert the implant into the cannula of the Insertion Tool manually or using the mosquito clamp. When inserting the implant into the cannula, DO NOT FORCE the implant. If resistance is felt, the implant should be removed and manually manipulated or rolled as needed, and re-inserted into the cannula.

or
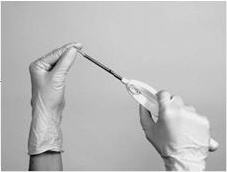
When fully inserted, the implant rests inside the cannula so that just the tip of the implant is visible at the beveled end of the cannula.
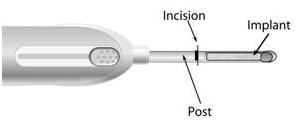
4. Making the Incision
Using the sterile scalpel provided, make an incision transverse to the long axis of the arm, and of a size adequate to allow the bore of the cannula to be inserted into the subcutaneous tissue.
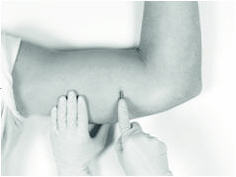
5. Inserting the Implant
The insertion may be easier if a “pocket” for the implant is first created by blunt dissection through the incision, subcutaneously along the path of the anesthetic, using the cannula of the loaded Insertion Tool, or using a sterile hemostatic clamp or equivalent surgical tool.
Be sure to VISIBLY RAISE THE SKIN (known as tenting) at all times during the pocket-making and insertion procedures to ensure correct subcutaneous placement (“just under the skin”) of the implant. Note that the cannula of the Insertion Tool, or whatever tool is being used to create the pocket, SHOULD NOT ENTER MUSCLE TISSUE. Deep insertion of the implant will not affect the performance of VANTAS, but may cause difficulty in the later removal of the implant.
If using the cannula of the loaded Insertion Tool to create the pocket, carefully insert the tip of the cannula into the incision and advance through the subcutaneous tissue, while visibly raising the skin along the length of the cannula up to, but no farther than, the inscribed black line on the cannula. DO NOT DEPRESS THE GREEN RETRACTION BUTTON ON THE TOOL WHILE INSERTING OR ADVANCING THE TOOL INTO THE INCISION.
Pull the Tool back, almost to the beveled tip of the cannula, and advance the Tool forward again, so that the cannula re-enters the pocket completely, but no farther than the inscribed black line. Be sure to keep the insertion path just immediately subcutaneous.
If another tool was used to create the pocket, now insert the loaded cannula of the Insertion Tool containing the implant through the incision, up to the inscribed black line.
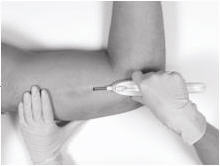
Hold the Insertion Tool in place with the base against the patient’s arm as you carefully move your thumb to the green retraction button. Depress the button to release the locking mechanism, then slide the button back toward the handle until it stops, all the while holding the body of the Insertion Tool in place.
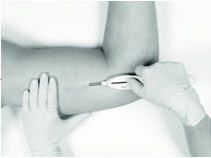
Retracting the button causes the cannula to withdraw from the incision, leaving the implant in the subcutaneous tissue. DO NOT FURTHER ADVANCE THE CANNULA ONCE THE RETRACTION PROCESS HAS STARTED. Likewise, do not withdraw the Insertion Tool until the button is fully retracted or the implant may be pulled partially out of the incision. Once the retraction is complete, the Tool can be fully withdrawn.
NOTE: It is helpful during the process of retraction and withdrawal of the cannula to apply pressure to the skin over the implant, to help ensure that the implant remains in the subcutaneous pocket.
If there is a need to re-start the process at any time during the insertion procedure, withdraw the Insertion Tool, carefully extract the implant from the cannula and reset the retraction button on the Tool to its forward-most position. Examine the implant before reloading the implant into the Insertion Tool, and start again.
Placement of the implant should be confirmed by palpation. Note that the tip of a properly-placed implant may not be visible through the incision.
After implantation, briefly cover the site with a sterile gauze pad and apply pressure to ensure hemostasis.
6. Closing the Incision
To close the incision, you can use the absorbable sutures and/or the sterile adhesive surgical strips provided. To improve adhesion of the strips, you can apply benzoin tincture antiseptic (provided) to the skin, and let it dry, before applying the adhesive strips.
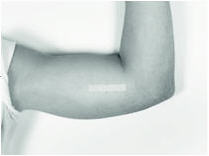
Once closed, cover the incision site with sterile gauze pads and secure the dressing with the bandage provided.
Please provide the patient with a Patient Information Leaflet, which includes information about the implant and instructions on proper care of the insertion site.
Removal Procedure
VANTAS should be removed after 12 months of therapy. Most of the supplies necessary to remove the implant, including the local anesthetic and the sterile mosquito clamp, are provided in the Implantation Kit that is shipped along with a new VANTAS implant. Note that the Implantation Kit is to be stored at 20°C to 25°C (68°F to 77°F) [See USP Controlled Room Temperature] only. See the Insertion Procedure above for further instructions.
Removal of the VANTAS implant is a surgical procedure. Sterile gloves and aseptic technique must be used to minimize any chance of infection.
1. Setting up the Sterile Field
Using proper aseptic technique, the sterilized components of the Implantation Kit needed for the implant removal procedure are to be carefully dispensed from their packaging onto the Sterile Field drape (non-fenestrated) provided. NOTE THAT THE KIT BOX AND ALL PACKAGING, INCLUDING THE SURFACE OF THE VIALS, ARE NOT STERILE and should be kept off of the Sterile Field drape.
2. Preparing the Patient and the Site
The patient should be on his back, with the arm containing the implant positioned, either bent or extended, so that the physician has ready access to the inner aspect of the upper arm. Propping the arm with pillows may help the patient more easily hold the position.
The implant to be removed should first be located by palpating the inner aspect of the upper arm, near the incision from the prior year.
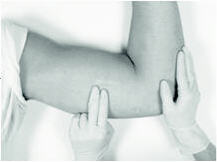
Generally, the previous implant is readily palpated. In the event the implant is difficult to locate, ultrasound may be used. If ultrasound fails to locate the implant, other imaging techniques such as CT or MRI may be used to locate it (plain films are not recommended as the implant is not radiopaque).
Antiseptic
Swab the area above and around the previous implant with topical antiseptic. Overlay the area with the fenestrated Sterile Field drape provided, so that the hole is over the previous insertion site (for clarity of illustration, the following images do not show the drape).
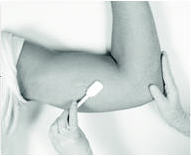
Anesthetic
The method of anesthesia utilized (i.e., local, conscious sedation, general) is at the discretion of the healthcare provider.
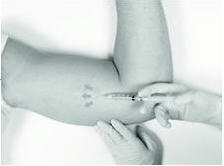
If local anesthesia is selected: a vial of a local anesthetic (e.g., lidocaine HCl 1% with epinephrine or lidocaine HCl 1%) (note that the exterior of the vial is not sterile) has been provided along with a sterile hypodermic needle for injection. After determining the absence of known allergies to the anesthetic agent, inject anesthetic into the subcutaneous tissue at and around the site of the intended incision (the site of the previous implant).
3. Making the Incision and Removing the Implant
Using the sterile scalpel provided, make an incision of a size adequate to allow the implant to be easily removed and, if a new implant will be inserted, large enough for the bore of the cannula of the Insertion Tool provided.
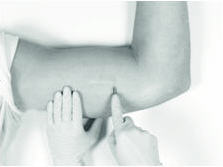
Generally, the tip of the implant will be visible through the incision, possibly covered by a pseudocapsule of tissue. In order to facilitate the removal of the implant, it may be necessary to palpate the head of the implant through the incision using your smallest finger, especially if the head of the implant is not readily visible. In addition, you may need to push down on the distal end of the implant and “massage it forward” toward the incision.
Carefully nick the pseudocapsule to reveal the polymer tip of the implant. It may be beneficial to insert the sterile mosquito clamp provided into the hole created in the pseudocapsule and expand by opening the clamp. Widening the opening of the pseudocapsule may ease the extraction of the implant.
Gently but securely grasp the implant with the sterile mosquito clamp and extract the implant.
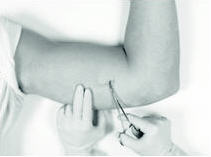
Dispose of the implant in a proper manner, treating it like any other biowaste.
Briefly cover the site with a sterile gauze pad and apply pressure to ensure hemostasis.
If inserting a new implant, see the Insertion Procedure instructions provided above. Note that you can insert the new implant into the same “pocket” as the removed implant, or make a new incision at a different site in the same arm or in the contralateral arm.
If a new implant is not to be inserted, proceed to close the incision.
4. Closing the Incision
To close the incision, you can use the absorbable sutures and/or the sterile adhesive surgical strips provided. To improve adhesion of the strips, you can apply benzoin tincture antiseptic (provided) to the skin, and let it dry, before applying the adhesive strips.
Once closed, cover the incision site with sterile gauze pads and secure the dressing with the bandage provided.
3 DOSAGE FORMS AND STRENGTHS
Subcutaneous Implant: 50 mg histrelin acetate, delivering 50 mcg histrelin acetate per day (equivalent to 41 mcg histrelin per day) over 12 months in a small thin flexible tube with histrelin acetate drug core inside a partially to completely full cylindrical hydrogel polymer reservoir. The implant may vary in color from off-white to light brown and may be uneven within the drug core.
4 CONTRAINDICATIONS
VANTAS is contraindicated in patients who are hypersensitive to gonadotropin releasing hormone (GnRH) or GnRH agonist analogs.
5 WARNINGS AND PRECAUTIONS
5.1 Transient Increase in Serum Testosterone
VANTAS causes a transient increase in serum concentrations of testosterone during the first week of treatment. Patients may experience worsening of symptoms or onset of new symptoms, including bone pain, neuropathy, hematuria, or ureteral or bladder outlet obstruction.
5.2 Spinal Cord Compression and Urinary Tract Obstruction
Cases of spinal cord compression, which may result in paralysis, and ureteral obstruction, which may cause renal impairment, have been reported with GnRH agonists. Patients with metastatic vertebral lesions and/or with urinary tract obstruction should be closely observed during the first few weeks of therapy.
5.3 Difficulty Locating or Removing Implant
In all clinical trials combined, an implant was not recovered in 8 patients. For two of these [see Clinical Studies (14)], serum testosterone rose above castrate level and the implant was neither palpable nor visualized with ultrasound. These two implants were believed to have been extruded without appreciation by the patients. In the other six, serum testosterone remained below the castrate level, but the implant was not palpable. No further diagnostic tests were conducted. One of these patients underwent in-clinic surgical exploration that did not locate the implant.
Implant insertion is a surgical procedure. Careful adherence to the Recommended Procedure for Implant Insertion and Removal [see Dosage and Administration (2.2)] is advised to minimize the potential for complications and for implant expulsion. In addition, patients should be instructed to refrain from wetting the arm for 24 hours and from heavy lifting or strenuous exertion of the inserted arm for 7 days after implant insertion.
5.4 Hyperglycemia and Diabetes
Hyperglycemia and an increased risk of developing diabetes have been reported in men receiving GnRH agonists. Hyperglycemia may represent development of diabetes mellitus or worsening of glycemic control in patients with diabetes. Monitor blood glucose and/or glycosylated hemoglobin (HbA1c) periodically in patients receiving a GnRH agonist and manage with current practice for treatment of hyperglycemia or diabetes.
5.5 Cardiovascular Disease
Increased risk of developing myocardial infarction, sudden cardiac death and stroke has been reported in association with use of GnRH agonists in men. The risk appears low based on the reported odds ratios, and should be evaluated carefully along with cardiovascular risk factors when determining a treatment for patients with prostate cancer. Patients receiving a GnRH agonist should be monitored for symptoms and signs suggestive of development of cardiovascular disease and be managed according to current clinical practice.
5.6 Laboratory Tests
Response to VANTAS should be monitored by measuring serum concentrations of testosterone and prostate-specific antigen periodically, especially if the anticipated clinical or biochemical response to treatment has not been achieved.
Results of testosterone determinations are dependent on assay methodology. It is advisable to be aware of the type and precision of the assay methodology to make appropriate clinical and therapeutic decisions.
5.7 Effect on QT/QTc Interval
Androgen deprivation therapy may prolong the QT/QTc interval. Providers should consider whether the benefits of androgen deprivation therapy outweigh the potential risks in patients with congenital long QT syndrome, congestive heart failure, frequent electrolyte abnormalities, and in patients taking drugs known to prolong the QT interval. Electrolyte abnormalities should be corrected. Consider periodic monitoring of electrocardiograms and electrolytes.
5.8 Embryo-Fetal Toxicity
The safety and efficacy of VANTAS have not been established in females. Based on findings from animal studies and its mechanism of action, VANTAS can cause fetal harm when administered to a pregnant woman [see Clinical Pharmacology (12.1)]. In animal reproduction studies, administration of VANTAS to pregnant rats and rabbits during the period of organogenesis caused an increase in fetal loss at clinically relevant exposures. Advise pregnant patients and females of reproductive potential of the potential risk to the fetus [see Use in Specific Populations (8.1)].
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of VANTAS was evaluated in 171 patients with prostate cancer treated for up to 36 months in two clinical trials. The pivotal study (Study 1) consisted of 138 patients, while a separate supportive study (Study 2) consisted of 33 patients.
VANTAS, like other GnRH analogs, caused a transient increase in serum testosterone concentrations during the first week of treatment. Therefore, potential exacerbations of signs and symptoms of the disease during the first few weeks of treatment are of concern in patients with vertebral metastases and/or urinary obstruction or hematuria. If these conditions are aggravated, it may lead to neurological problems such as weakness and/or paresthesia of the lower limbs or worsening of urinary symptoms [see Warnings and Precautions (5.1)].
In the first 12 months after initial insertion of the implant(s), an implant extruded through the incision site in eight of 171 patients in the clinical trials (see the Recommended Procedure for correct implant placement).
In the pivotal study (Study 1) a detailed evaluation for implant site reactions was conducted. Out of the 138 patients in the study, 19 patients (13.8%) experienced local or insertion site reactions. All these local site reactions were reported as mild in severity. The majority were associated with initial insertion or removal and insertion of a new implant, and began and resolved within the first two weeks following implant insertion. Reactions persisted in 4 (2.8%) patients. An additional 4 (2.8%) patients developed application-site reactions after the first two weeks following insertion.
Local reactions after implant insertion included bruising (7.2% of patients) and pain/soreness/tenderness (3.6% of patients). Other, less frequently reported, reactions included erythema (2.8% of patients) and swelling (0.7% of patients). In this study, two patients had events described as local infections/inflammations, one that resolved after treatment with oral antibiotics and the other without treatment.
Local reactions following insertion of a subsequent implant were comparable to those seen after initial insertion.
The following possibly or probably related systemic adverse events occurred during clinical trials of up to 24 months of treatment with VANTAS, and were reported in ≥ 2% of patients (Table 1).
| * Expected pharmacological consequences of testosterone suppression. ** 5 of the 8 patients had a single occurrence of mild renal impairment (defined as creatinine clearance ≥30 <60 mL/min), which returned to a normal range by the next visit. |
||
| Body System | Adverse Event | Number (%) |
|---|---|---|
| Vascular Disorders | Hot flashes* | 112 (65.5%) |
| General Disorders | Fatigue Weight increased | 17 (9.9%) 4 (2.3%) |
| Skin and Appendage Disorders | Implant site reaction | 10 (5.8%) |
| Reproductive System and Breast Disorders | Erectile dysfunction* Gynecomastia* Testicular atrophy* | 6 (3.5%) 7 (4.1%) 9 (5.3%) |
| Psychiatric Disorders | Insomnia Libido decreased* | 5 (2.9%) 4 (2.3%) |
| Renal and Urinary Disorders | Renal impairment** | 8 (4.7%) |
| Gastrointestinal Disorders | Constipation | 6 (3.5%) |
| Nervous System Disorders | Headache | 5 (2.9%) |
Hot flashes were the most common adverse event reported (65.5% of patients). In terms of severity, 2.3% of patients reported severe hot flashes, 25.4% of patients reported moderate hot flashes and 37.7% reported mild hot flashes. In addition, the following possibly or probably related systemic adverse events were reported by < 2% of patients using VANTAS in clinical studies.
-
Blood and Lymphatic System Disorders: Anemia
-
Cardiac Disorders: Palpitations, ventricular extrasystoles
-
Gastrointestinal Disorders: Abdominal discomfort, nausea
-
General Disorders: Feeling cold, lethargy, malaise, edema peripheral, pain, pain exacerbated, weakness, weight decreased
-
Hepatobiliary Disorders: Hepatic disorder
-
Injury, Poisoning and Procedural Complications: Stent occlusion
-
Laboratory Investigations: Aspartate aminotransferase increased, blood glucose increased, blood lactate dehydrogenase increased, blood testosterone increased, creatinine clearance decreased, prostatic acid phosphatase increased
-
Metabolism and Nutrition Disorders: Appetite increased, fluid retention, food craving, hypercalcaemia, hypercholesterolemia
-
Musculoskeletal and Connective Tissue Disorders: Arthralgia, back pain, back pain aggravated, bone pain, muscle twitching, myalgia, neck pain, pain in limb
-
Nervous System Disorders: Dizziness, tremor
-
Psychiatric Disorders: Depression, irritability
-
Renal and Urinary Disorders: Calculus renal, dysuria, hematuria aggravated, renal failure aggravated, urinary frequency, urinary frequency aggravated, urinary retention
-
Reproductive System and Breast Disorders: Breast pain, breast tenderness, genital pruritus male, gynecomastia aggravated, sexual dysfunction
-
Respiratory, Thoracic and Mediastinal Disorders: Dyspnea exertional
-
Skin and Subcutaneous Tissue Disorders: Contusion, hypotrichosis, night sweats, pruritus, sweating increased
-
Vascular Disorders: Flushing, hematoma
- Changes in Bone Density: Decreased bone density has been reported in the medical literature in men who have had orchiectomy or who have been treated with a GnRH agonist analog. It can be anticipated that long periods of medical castration in men will have effects on bone density.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of VANTAS. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Pituitary Apoplexy: Cases of pituitary apoplexy (a clinical syndrome secondary to infarction of the pituitary gland) have been reported after the administration of gonadotropin-releasing hormone agonists. In a majority of these cases, a pituitary adenoma was diagnosed with a majority of pituitary apoplexy cases occurring within 2 weeks of the final dose, and some within the first hour. In these cases, pituitary apoplexy has presented as sudden headache, vomiting, visual changes, opthalmoplegia, altered mental status, and sometimes cardiovascular collapse. Immediate medical attention has been required.
Drug-Induced Liver Injury: Severe liver injury has been reported in association with VANTAS. The toxicity was reversible with the removal of the VANTAS implant.
Nervous System Disorders: Convulsions
7 DRUG INTERACTIONS
Overview: No pharmacokinetic-based drug-drug interaction studies were conducted with VANTAS [see Clinical Pharmacology (12.3)].
Drug-Laboratory Test Interactions: Therapy with histrelin results in suppression of the pituitary-gonadal system. Results of diagnostic tests of pituitary gonadotropic and gonadal functions conducted during and after histrelin therapy may be affected.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
The safety and efficacy of VANTAS have not been established in females. Based on findings in animal studies and its mechanism of action, VANTAS can cause fetal harm when administered to a pregnant woman [see Clinical Pharmacology (12.1)]. Expected hormonal changes that occur with VANTAS treatment increase the risk for pregnancy loss. The limited data with histrelin use in pregnant women are insufficient to determine a drug-associated risk for major birth defects or adverse developmental outcomes. In animal reproduction studies, administration of histrelin to pregnant rats and rabbits during the period of organogenesis caused an increase in fetal loss at clinically relevant exposures (see Data). Advise pregnant patients and females of reproductive potential of the potential risk to the fetus.
Data
Animal Data
Histrelin acetate administered to pregnant rats during the period of organogenesis increased fetal mortality and post-implantation loss at doses of 1, 3, 5 or 15 mcg/kg/day (approximately 0.2- to 3-times clinical exposure based on body surface area). These dosages also reduced maternal body weight gain, stimulated ovarian follicular development, increased placental weight and caused abnormal morphology and an increase in fetal size. Histrelin acetate administered to pregnant rabbits during the period of organogenesis increased fetal mortality and abortion/early termination at the two highest doses and caused total litter loss at all doses of 20, 50 or 80 mcg/kg/day (approximately 8- to 31-times clinical exposures based on body surface area).
8.2 Lactation
The safety and efficacy of VANTAS have not been established in females. There are no data on the presence of VANTAS in human or animal milk, the effects on the breastfed child, or the effects on milk production. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in a breastfed child, the developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for VANTAS and any potential adverse effects on the breastfed child from VANTAS or from the underlying maternal condition.
8.3 Females and Males of Reproductive Potential
Infertility
Male
Based on findings in animals and mechanism of action, VANTAS may impair fertility in males of reproductive potential [see Nonclinical Toxicology (13.1)].
10 OVERDOSAGE
Histrelin acetate injection of up to 200 mcg/kg (rats, rabbits), or 2000 mcg/kg (mice) resulted in no systemic toxicity. This represents 20 to 200 times the maximal recommended human dose of 10 mcg/kg/day. Adverse event profiles were similar in patients receiving one, two or four VANTAS implants.
11 DESCRIPTION
VANTAS® (histrelin acetate) implant is a sterile, non-biodegradable, diffusion-controlled hydrogel polymer reservoir containing histrelin acetate, a synthetic nonapeptide analog of the naturally occurring gonadotropin releasing hormone (GnRH). VANTAS is designed to deliver approximately 50 mcg histrelin acetate per day (equivalent to approximately 41 mcg histrelin per day) over 12 months.
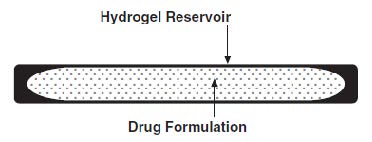
The sterile VANTAS implant looks like a small thin flexible tube and consists of a 50-mg histrelin acetate drug core inside a 3.5 cm by 3 mm, cylindrical hydrogel polymer reservoir (Figure A). The implant may appear partially to completely full with variation in color from off-white to light brown. The color may be uneven within the core.
Figure A. VANTAS Implant diagram (not to scale)
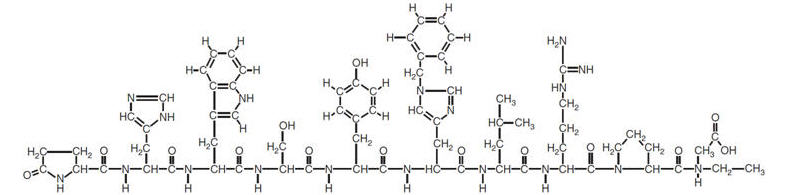
Histrelin acetate is chemically described as: 5-oxo-L-prolyl-L-histidyl-L-tryptophyl-L-seryl-L-tyrosyl-Ntbenzyl-D-histidyl-L-leucyl-L-arginyl-N-ethyl-L-prolinamide acetate (salt) [C66H86N18O12 (1.7-2.8 moles) CH3COOH, (0.6-7.0 moles) H2O], with the molecular weight of 1443.70 (or 1323.50 as histrelin base).
Histrelin acetate has the following structural formula:
The drug core also contains the inactive ingredient stearic acid NF. The hydrogel polymer reservoir is a hydrophilic cartridge composed of 2-hydroxyethyl methacrylate, 2-hydroxypropyl methacrylate, trimethylolpropane trimethacrylate, benzoin methyl ether, Perkadox-16, and Triton X-100. Each implant is packaged hydrated in a glass vial containing 2 mL of sterile 1.8% sodium chloride solution. The implant is primed for immediate release of the drug upon insertion.
A single use, sterile Insertion Tool is provided along with the implant that may be used for the placement of the implant into the subcutaneous tissue of the inner aspect of the upper arm. The Insertion Tool is enclosed in a sterile bag and is provided separately from the implant in the Implantation Kit [see Dosage and Administration (2.2)].
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Histrelin acetate is a gonadotropin releasing hormone (GnRH) agonist that acts as a potent inhibitor of gonadotropin secretion when given continuously in therapeutic doses. Both animal and human studies indicate that following an initial stimulatory phase, chronic, subcutaneous administration of histrelin acetate desensitizes responsiveness of the pituitary gonadotropin which, in turn, causes a reduction in testicular steroidogenesis.
In humans, administration of histrelin acetate results in an initial increase in circulating levels of luteinizing hormone (LH) and follicle-stimulating hormone (FSH), leading to a transient increase in concentration of gonadal steroids (testosterone and dihydrotestosterone in males); however, continuous administration of histrelin acetate results in decreased levels of LH and FSH due to a reversible down-regulation of the GnRH receptors in the pituitary gland and desensitization of the pituitary gonadotropes. In males, testosterone is reduced to castration levels. These decreases occur within 2 to 4 weeks after initiation of treatment.
Histrelin acetate is not active when given orally.
12.3 Pharmacokinetics
Absorption: Following subcutaneous insertion of one VANTAS implant in advanced prostate cancer patients (n = 17), peak serum concentrations of 1.10 ± 0.375 ng/mL (mean ± SD) occurred at a median of 12 hours. Continuous subcutaneous release was evident, as serum levels were sustained throughout the 52 week dosing period (see Figure 1). The mean serum histrelin concentration at the end of the 52 week treatment duration was 0.13 ± 0.065 ng/mL. When histrelin serum concentrations were measured following a second implant inserted after 52 weeks, the observed serum concentrations over 8 weeks following the second implant were comparable to the same period following the first implant. The average rate of subcutaneous drug release from 41 implants assayed for residual drug content was 56.7 ± 7.71 mcg/day, over the 52 week dosing period. The relative bioavailability for the VANTAS implant in prostate cancer patients with normal renal and hepatic function compared to a subcutaneous bolus dose in healthy male volunteers was 92%. Serum histrelin concentrations were proportional to dose after one, two or four 50 mg VANTAS implants (50, 100 or 200 mg as histrelin acetate) in 42 prostate cancer patients.
Figure 1: Mean Serum Histrelin Concentration versus Time Profile for 17 Patients Following Insertion of First and Second VANTAS Implants. (Note that only four patients underwent intensive pharmacokinetic sampling during the first 96 hours following the second implant.)
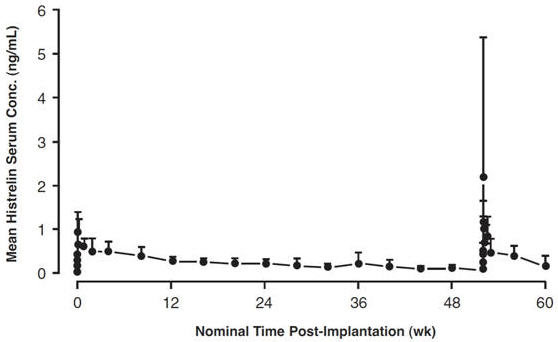
Distribution: The apparent volume of distribution of histrelin following a subcutaneous bolus dose (500 mcg) in healthy volunteers was 58.4 ± 7.86 L. The fraction of drug unbound in plasma measured in vitro was 29.5% ± 8.9% (mean ± SD).
Metabolism: An in vitro drug metabolism study using human hepatocytes identified a single histrelin metabolite resulting from C-terminal dealkylation. Peptide fragments resulting from hydrolysis are also likely metabolites. Following a subcutaneous bolus dose in healthy volunteers the apparent clearance of histrelin was 179 ± 37.8 mL/min (mean ± SD) and the terminal half-life was 3.92 ± 1.01 hr (mean ± SD). The apparent clearance following a 50 mg (as histrelin acetate) VANTAS implant in 17 prostate cancer patients was 174 ± 56.5 mL/min (mean ± SD).
Excretion: No drug excretion study was conducted with VANTAS implants.
Special Populations:
Geriatrics: The majority (89.9%) of the 138 patients studied in the pivotal clinical trial were age 65 and over.
Pediatrics: VANTAS is not indicated for use in pediatric patients. [see Use in Specific Populations (8.4)].
Race: When serum histrelin concentrations were compared for 7 Hispanic, 30 Black and 77 Caucasian patients, average serum histrelin concentrations were similar.
Renal Insufficiency: When average serum histrelin concentrations were compared between 42 prostate cancer patients with mild to severe renal impairment (CLcr: 15-60 mL/min) and 92 patients with no renal or hepatic impairment, levels were approximately 50% higher in those patients with renal impairment (0.392 ng/mL versus 0.264 ng/mL). These changes in exposure as a result of renal impairment are not considered to be clinically relevant. Therefore, no changes in drug dosing are warranted for these patient subpopulations.
Hepatic insufficiency: The influence of hepatic insufficiency on histrelin pharmacokinetics has not been adequately studied.
Drug-Drug Interactions: No pharmacokinetic-based drug-drug interaction studies were conducted with VANTAS.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies were conducted in rats for 2 years at doses of 5, 25 or 150 mcg/kg/day (up to 22 times human exposures using body surface area comparisons, based on a 65 mcg/day dose in adults) and in mice for 18 months at doses of 20, 200, or 2000 mcg/kg/day (up to 150 times human exposure using body surface area comparisons, based on a 65 mcg/day dose in adults). As seen with other GnRH agonists, histrelin acetate injection administration was associated with an increase in tumors of hormonally responsive tissues. There was a significant increase in pituitary adenomas in rats. There was an increase in pancreatic islet-cell adenomas in treated female rats and a non-dose-related increase in testicular Leydig-cell tumors (highest incidence in the low-dose group). In mice, there was significant increase in mammary-gland adenocarcinomas in all treated females. In addition, there were increases in stomach papillomas in male rats given high doses, and an increase in histiocytic sarcomas in female mice at the highest dose.
Mutagenicity studies have not been performed with histrelin acetate. Saline extracts of implants with and without histrelin were negative in a battery of genotoxicity studies. Studies examining fertility following withdrawal of histrelin acetate have been conducted in rats and monkeys given subcutaneous daily doses of histrelin acetate for 6 months, at doses of up to 180 mcg/kg/day (up to 27-times [rat] and 54-times [monkey] adult clinical exposures using body surface area comparisons, based on a 65 mcg/day dose in humans). Full reversibility of fertility suppression was demonstrated. The development and reproductive performance of offspring from parents treated with histrelin acetate has not been investigated.
14 CLINICAL STUDIES
In one open-label, multicenter, Phase 3 study (Study 1), 138 patients with prostate cancer were treated with a single VANTAS implant and were evaluated for at least 60 weeks. Of these, 37 patients had Jewett stage C disease, 29 had stage D disease, and the remaining 72 patients had an elevated or rising serum PSA after definitive therapy for localized disease. Serum testosterone levels were assessed as the primary efficacy endpoint to evaluate both achievement and maintenance of castrate testosterone suppression, with treatment success being defined as a serum testosterone level ≤ 50 ng/dL. At Week 52, the study included the option for removal and insertion of a new implant, with evaluation for an additional 52 weeks (the “extension phase”). A total of 120 patients completed the initial 52-week treatment period. Reasons for discontinuation were: death (n=6), disease progression (n=5), implant expulsion (n=3), hospice placement (n=2), and patient request/no specific reason given (n=2). Of the 120 patients who successfully completed 52 weeks of treatment, 111 were evaluable for efficacy. A total of 113 patients underwent removal of the first implant and insertion of a second implant for another year of therapy.
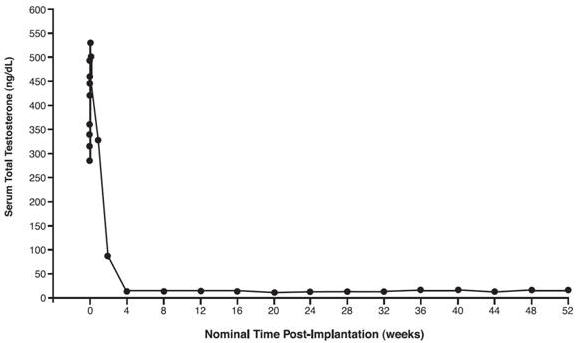
In a subset of 17 patients, serum testosterone concentrations were measured within the first week following initial implantation. In these 17 patients, mean serum testosterone concentrations increased from 376.4 ng/dL at Baseline to 530.5 ng/dL on Day 2, then decreased to below baseline by Week 2, and to below the 50 ng/dL castrate threshold by Week 4 (see Figure 2). Serum testosterone concentrations remained below the castrate level in this subset for the entire treatment period.
Figure 2: Mean Serum Total Testosterone Concentrations for all PK Patients, n=17.
(Note that in this group, sampling began minutes after insertion of VANTAS.)
In the overall treatment group (n=138), mean serum testosterone was 388.3 ng/dL at Baseline. At the time of first assessment of testosterone (at the end of Week 1), the mean serum testosterone concentration was 382.8 ng/dL. At Week 2, mean serum testosterone was 92.2 ng/dL. At Week 4 it was 15 ng/dL. At Week 52, the final mean testosterone concentration was 14.3 ng/dL (see Figure 3).
Figure 3. Mean Serum Total Testosterone Concentrations (+SD) for All Patients (n=138) Who Received One VANTAS Implant. (Note that in this group, sampling began at the end of Week 1.)
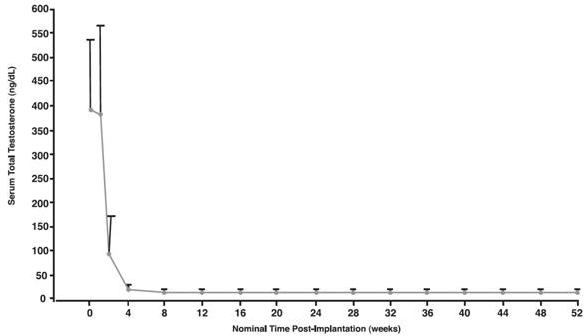
Of 138 patients who received an implant, one discontinued prior to Day 28 when the implant was expelled on Day 15. Three others did not have an efficacy measurement for the Day 28 visit. Otherwise serum testosterone was suppressed to below the castrate level (≤ 50 ng/dL) in all 134 evaluable patients (100%) on Day 28. All three patients with missing values at Day 28 were castrate by the time of their next visit (Day 56).
Once serum testosterone concentrations at or below castrate level (≤ 50 ng/dL) were achieved, a total of 4 patients (3%) demonstrated breakthrough during the study. In one patient, a serum testosterone of 63 ng/dL was reported at Week 44. In another patient, a serum testosterone of 3340 ng/dL was reported at Week 40. This aberrant value was possibly related to lab error. In two patients, serum testosterone rose above castrate level and the implant could neither be palpated nor visualized with ultrasound. In the first patient, serum testosterone was 669 ng/dL at Week 8 and 311 ng/dL at Week 12. This patient reported strenuous exertion after insertion of the implant and a large scab formed at the insertion site. The implant may have been expelled without the patient’s appreciation of the event. The other patient developed erythema at the insertion site at Week 22 and was treated with oral antibiotics. At Week 26, the implant was not palpable and was not visualized with ultrasound. At Week 34, the serum testosterone rose to 135 ng/dL. The implant may have been expelled without the patient’s appreciation of the event. A new implant was inserted.
Of 120 patients who completed 52 weeks of treatment, a total of 115 patients had a serum testosterone measurement at Week 52. Of these, all had serum testosterone ≤ 50 ng/dL. In patients without a Week 52 value, castrate levels were achieved by Day 28, were maintained up to Week 52, and remained below the castrate threshold after Week 52.
In all 18 patients who prematurely discontinued prior to Week 52 – except one (implant expulsion on Day 15) – castrate levels of serum testosterone were achieved by Day 28 and were maintained up to and including the time of withdrawal.
A total of 113 patients had a new implant inserted for a second year of therapy following removal of the first implant. Of this group, 68 patients had measurement of serum testosterone on Day 2 or Day 3 and on Day 7 after insertion of the second implant in order to assess for the “acute-on-chronic” phenomenon. No acute increase in serum testosterone was seen in any patient in this group following insertion of the new implant.
Serum prostate specific antigen (PSA) was monitored as a secondary endpoint. Serum PSA decreased from baseline in all patients after they began treatment with VANTAS. Serum PSA decreased to within normal limits by Week 24 in 103 of 111 evaluable patients (93%).
Prior to conducting the pivotal study, a Phase 2, dose-ranging study was performed in 42 patients with advanced prostate cancer. Efficacy was assessed by serum testosterone levels as the primary efficacy endpoint. Patients received 1, 2 or 4 implants. The use of 2 or 4 implants did not confer any additional benefit in suppression of testosterone beyond that produced by the single implant.
16 HOW SUPPLIED/STORAGE AND HANDLING
VANTAS (NDC 67979-500-01) is supplied in a carton containing 2 inner cartons, one for the VANTAS implant and one for the VANTAS Implantation Kit:
The VANTAS implant contains 50 mg of histrelin acetate. The VANTAS implant carton contains a cold pack for refrigerated shipment and a small carton containing an amber plastic pouch. Inside the pouch is a glass vial with a Teflon-coated stopper and an aluminum seal, containing the implant in 2 mL of sterile 1.8% sodium chloride solution. (Note: The 3.5 mL vial is not completely filled with saline.)
Upon receipt, refrigerate the small carton containing the amber plastic pouch and glass vial (with the implant inside) until the day of insertion. The implant vial should not be opened until just before the time of insertion.
Store the implant refrigerated, 2°C to 8°C (36°F to 46°F), in the unopened glass vial with the sterile 1.8% sodium chloride solution, overwrapped in the amber plastic pouch and carton, until the expiration date provided. Excursion permitted to 25°C (77°F) for 7 days. Protect from light. Do not freeze.
Store the VANTAS Implantation Kit at room temperature only.
The VANTAS Implantation Kit carton contains one each of the following (individually wrapped in sterile packaging): implant insertion tool, #15 disposable scalpel, syringe with 18 gauge needle, 25 gauge 1.5” needle, SS mosquito clamp, benzoin tincture antiseptic, alcohol swabs (2 packages), fenestrated drape, non-fenestrated drape, skin antiseptic swab, gauze sponges, surgical closure strips, coated absorbable sutures, cohesive bandage, and a local anesthetic (e.g., lidocaine HCl 1% with epinephrine or lidocaine HCl 1%).
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Hypersensitivity
Inform patients that if they have experienced hypersensitivity with other GnRH agonists like VANTAS, VANTAS is contraindicated [see Contraindications (4)].
Transient Increase in Serum Testosterone
Inform patients that VANTAS can cause worsening of symptoms or onset of new symptoms, including bone pain, neuropathy, hematuria, or ureteral or bladder outlet obstruction [see Warnings and Precautions (5.1)]
Spinal Cord Compression and Urinary Tract Obstruction
Inform patients that cases of spinal cord compression which may result in paralysis or ureteral obstruction have been reported with GnRH agonists like VANTAS. Advise patients to report new or worsening signs or symptoms of spinal cord compression or urinary tract obstruction to their healthcare provider [see Warnings and Precautions (5.2)]
Difficulty Locating or Removing Implant
Instruct patients to refrain from wetting the arm for 24 hours and from heavy lifting or strenuous exertion of the inserted arm for 7 days after implant insertion [see Warnings and Precautions (5.3)].
Hyperglycemia and Diabetes
Advise patients that there is an increased risk of hyperglycemia and diabetes with VANTAS therapy. Inform patients that periodic monitoring for hyperglycemia and diabetes is required when being treated with VANTAS [see Warnings and Precautions (5.4)].
Cardiovascular Disease
Inform patients that there is an increased risk of myocardial infarction, sudden cardiac death, and stroke with VANTAS treatment. Advise patients to immediately report signs and symptoms associated with these events to their healthcare provider for evaluation [see Warnings and Precautions (5.5)].
Implant Site Reactions
Inform patients that injection site related adverse reactions may occur such as transient bruising, pain/soreness/tenderness, erythema, and swelling. Advise patients to contact their healthcare provider if they experience severe implant site reactions or implant extrusion through the incision site [see Adverse Reactions (6.1)].
Urogenital Disorders
Advise patients that VANTAS may cause sexual dysfunction [see Adverse Reactions (6.1)].
Infertility
Advise males of reproductive potential that VANTAS may impair fertility [see Use in Specific Populations (8.3)].
Continuation of VANTAS Treatment
Inform patients that VANTAS is usually continued, often with additional medication, after the development of non-metastatic and metastatic castration-resistant prostate cancer [see Dosage and Administration (2)].
Distributed by:
Endo Pharmaceuticals Inc.
Malvern, PA 19355
VANTAS® is a registered trademark of Endo Pharmaceuticals Inc. or one of its affiliates.
©2020 Endo Pharmaceuticals Inc. All rights reserved.
Revised: 12/2020
PATIENT INFORMATION
VANTAS® (Van-tas)
(histrelin acetate)
subcutaneous implant
What is VANTAS?
VANTAS is an implant that contains the medicine histrelin and is placed under the skin. VANTAS may help relieve the symptoms of advanced prostate cancer.
It is not known if VANTAS is safe or effective in females.
It is not known if VANTAS is safe or effective in children.
Do not use VANTAS if you are allergic to gonadotropin releasing hormone (GnRH) or GnRH agonists medicines like VANTAS. See the end of this leaflet for a complete list of ingredients in VANTAS.
Before receiving VANTAS, tell your healthcare provider about all of your medical conditions, including if you:
- have cancer that has spread to your spine
- have trouble urinating
- have diabetes
- have heart problems, including a condition called long QT syndrome
- have problems with body salt (electrolytes) levels, such as sodium, potassium, calcium or magnesium
- have an allergy to numbing medicines (local anesthetics). These medicines may be used when the implant is placed into and removed from your arm.
- are pregnant or plan to become pregnant. VANTAS can harm your unborn baby.
- are breastfeeding or plan to breastfeed. It is not known if VANTAS passes into your breast milk.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins and herbal supplements.
How will I receive VANTAS?
- VANTAS is placed under the skin of the inside of your upper arm by your healthcare provider. Your healthcare provider will numb your arm, make a small cut, and then place VANTAS under the skin. The cut will be closed with stitches and special surgical tape and covered with a bandage.
- Keep your arm clean and dry for 24 hours after VANTAS is placed.
- Avoid heavy lifting and exercise for 7 days with the arm that VANTAS was placed in. Avoid bumping the site for a few days. Your healthcare provider will give you complete instructions.
- After VANTAS is placed under the skin, the medicine is released into your body for 12 months.
- After 12 months, VANTAS must be removed. Your healthcare provider may place a new VANTAS at this time to continue your treatment.
- If the VANTAS implant cannot be felt under your skin at the time for removal, your healthcare provider may order special tests, such as ultrasound or computed tomography (CT) scan, in order to locate it for removal.
- Your healthcare provider will do blood tests to check your response to treatment with VANTAS.
What are the possible side effects of VANTAS?
VANTAS may cause serious side effects, including:
-
Increase in blood testosterone levels. VANTAS can cause a temporary increase in blood testosterone levels during the first week after it is inserted. Your symptoms may get worse or you may get new symptoms. Call your healthcare provider right away if you get new or worsening:
- bone pain
- weakness, numbness, tingling, or burning sensation in your legs
- blood in your urine
- trouble urinating or cannot urinate
-
Pressure on the spinal cord and blockage of the urinary tract have happened in some people taking GnRH agonists like VANTAS. Pressure on the spinal cord may cause paralysis and blockage of the urinary tract. Tell your healthcare provider if you get new or worsening signs or symptoms, including:
- loss of ability to move (paralysis)
- pain or stiffness in the neck, back, or lower back
- numbness, cramping, or weakness in the arms, hands, or legs
- urinating less than usual or unable to urinate
- loss of control of the bowels or bladder
- Problems with finding or removing the VANTAS implant.
- High blood sugar (hyperglycemia) and diabetes. There is an increased risk of high blood sugar and diabetes in people taking GnRH agonists like VANTAS. Your healthcare provider should monitor your blood sugar levels during treatment with VANTAS and treat you if needed.
- Heart problems. There is an increased risk for developing heart attacks, sudden death due to heart problems and stroke in people taking GnRH agonists like VANTAS. Your healthcare provider should monitor you for heart problems during treatment with VANTAS and treat you if needed. Tell your healthcare provider or get emergency medical help right away if you get signs and symptoms of heart problems, including:
- chest pain that may spread to the arms, neck, jaw, back, or stomach area (abdomen)
- feeling sweaty
- shortness of breath
- feeling sick or vomiting
- lightheadedness or dizziness
- feel like your heart is racing or fluttering
- sudden numbness or weakness, especially on one side of the body
- severe headache or confusion
- problems with vision, speech, or balance
- Changes in the electrical activity of your heart (QT prolongation). Your healthcare provider may check the electrical activity of your heart during treatment with VANTAS.
The most common side effects of VANTAS include:
- hot flashes
- tiredness
- skin reactions at the place where the implant is inserted. These reactions may include temporary bruising, pain, soreness, tenderness, redness, and swelling. Tell your healthcare provider if you get severe reactions where the implant is inserted or if the implant becomes exposed through the skin.
- testicles become smaller
Other side effects include sexual dysfunction and fertility problems in males, which may affect your ability to father children. Talk to your healthcare provider if this is a concern for you.
These are not all of the possible side effects of VANTAS.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of VANTAS.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. You can ask your pharmacist or healthcare provider for information about VANTAS that is written for health professionals.
What are the ingredients in VANTAS?
Active ingredient: histrelin acetate.
Inactive ingredients: stearic acid NF, hydrogel polymer reservoir composed of 2-hydroxyethyl methacrylate, 2-hydroxypropyl methacrylate, trimethylolpropane trimethacrylate, benzoin methyl ether, Perkadox-16, and Triton X-100.
Distributed by: Endo Pharmaceuticals Inc., Malvern, PA 19355
VANTAS® is a registered trademark of Endo Pharmaceuticals Inc. or one of its affiliates.
©2020 Endo Pharmaceuticals Inc. All rights reserved.
This Patient Information has been approved by the U.S. Food and Drug Administration.
Revised: 12/2020
| VANTAS
histrelin acetate implant |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Endo Pharmaceuticals Inc. (178074951) |

