LONHALA MAGNAIR- glycopyrrolate solution
Sunovion Pharmaceuticals Inc.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use LONHALA® MAGNAIR® safely and effectively. See full prescribing information for LONHALA MAGNAIR.
LONHALA MAGNAIR (glycopyrrolate) inhalation solution, for oral inhalation use Initial U.S. Approval: 1961 INDICATIONS AND USAGELONHALA MAGNAIR is an anticholinergic indicated for the long-term, maintenance treatment of airflow obstruction in patients with chronic obstructive pulmonary disease (COPD). (1) DOSAGE AND ADMINISTRATIONDOSAGE FORMS AND STRENGTHSLONHALA Inhalation Solution is supplied as a sterile solution for inhalation in a unit-dose single-use low-density polyethylene (LDPE) vial. Each 1 mL vial contains 25 mcg of glycopyrrolate. (3) CONTRAINDICATIONSLONHALA MAGNAIR is contraindicated in patients with a hypersensitivity to glycopyrrolate or any of the ingredients. (4) WARNINGS AND PRECAUTIONS
ADVERSE REACTIONSMost common adverse reactions (incidence greater than or equal to 2.0% and higher than placebo) are dyspnea and urinary tract infection. (6.1) To report SUSPECTED ADVERSE REACTIONS, contact Sunovion Pharmaceuticals Inc. at 1-877-737-7226 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. DRUG INTERACTIONSAnticholinergics: May interact additively with concomitantly used anticholinergic medications. Avoid administration of LONHALA MAGNAIR with other anticholinergic-containing drugs. (7.2) USE IN SPECIFIC POPULATIONSUse in patients with severe renal impairment should be considered if the potential benefit of the treatment outweighs the risk. (8.6) See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling. Revised: 8/2020 |
FULL PRESCRIBING INFORMATION
1. INDICATIONS AND USAGE
LONHALA MAGNAIR is indicated for the long-term maintenance treatment of airflow obstruction in patients with chronic obstructive pulmonary disease (COPD), including chronic bronchitis and/or emphysema.
2. DOSAGE AND ADMINISTRATION
For oral inhalation only. Do not swallow LONHALA solution. LONHALA vials should only be used with MAGNAIR [see Overdosage (10)].
The recommended dose of LONHALA is the inhalation of the contents of one LONHALA vial twice-daily using MAGNAIR. LONHALA vials should only be administered with MAGNAIR. Patients should be instructed on the correct use of this drug product and device.
LONHALA MAGNAIR should be administered at the same time of the day, (1 vial in the morning and 1 vial in the evening), every day. More frequent administration or a greater number of inhalations (more than 1 vial twice daily) of LONHALA MAGNAIR is not recommended.
Store LONHALA vials in the foil pouch, and only remove IMMEDIATELY BEFORE USE with MAGNAIR.
No dosage adjustment is required for geriatric patients, patients with hepatic impairment, or patients with mild to moderate renal impairment.
3. DOSAGE FORMS AND STRENGTHS
LONHALA Inhalation Solution is supplied as a sterile, clear, colorless, aqueous solution for inhalation in a unit-dose single-use low-density polyethylene (LDPE) vial. Each 1 mL vial contains 25 mcg of glycopyrrolate.
4. CONTRAINDICATIONS
LONHALA MAGNAIR is contraindicated in patients with a hypersensitivity to glycopyrrolate or any of the ingredients [see Warnings and Precautions (5.3)].
5. WARNINGS AND PRECAUTIONS
5.1. Deterioration of Disease and Acute Episodes
LONHALA MAGNAIR should not be initiated in patients during acutely deteriorating or potentially life-threatening episodes of COPD. LONHALA MAGNAIR has not been studied in subjects with acutely deteriorating COPD. The initiation of LONHALA MAGNAIR in this setting is not appropriate.
LONHALA MAGNAIR should not be used as rescue therapy for the treatment of acute episodes of bronchospasm. LONHALA MAGNAIR has not been studied in the relief of acute symptoms and extra doses should not be used for that purpose. Acute symptoms should be treated with an inhaled, short-acting beta2-agonist.
COPD may deteriorate acutely over a period of hours or chronically over several days or longer. If LONHALA MAGNAIR no longer controls symptoms of bronchoconstriction the patient's inhaled, short-acting beta2-agonist becomes less effective; or the patient needs more inhalations of a short-acting beta2-agonist than usual, these may be markers of deterioration of disease. In this setting, a re-evaluation of the patient and the COPD treatment regimen should be undertaken at once. Increasing the daily dose of LONHALA MAGNAIR beyond the recommended dose is not appropriate in this situation.
5.2. Paradoxical Bronchospasm
As with other inhaled medicines, LONHALA MAGNAIR can produce paradoxical bronchospasm that may be life-threatening. If paradoxical bronchospasm occurs following dosing with LONHALA MAGNAIR, it should be treated immediately with an inhaled, short-acting bronchodilator; LONHALA MAGNAIR should be discontinued immediately, and alternative therapy instituted.
5.3. Immediate Hypersensitivity Reactions
Immediate hypersensitivity reactions may occur after administration of LONHALA MAGNAIR. If signs suggesting allergic reactions occur, in particular, angioedema (including difficulties in breathing or swallowing, swelling of the tongue, lips, and face), urticaria, or skin rash, LONHALA MAGNAIR should be discontinued immediately and alternative therapy instituted.
5.4. Worsening of Narrow-Angle Glaucoma
LONHALA MAGNAIR should be used with caution in patients with narrow-angle glaucoma. Prescribers and patients should be alert for signs and symptoms of acute narrow-angle glaucoma (e.g., eye pain or discomfort, blurred vision, visual halos or colored images in association with red eyes from conjunctival congestion and corneal edema). Instruct patients to consult a physician immediately should any of these signs or symptoms develop.
5.5. Worsening of Urinary Retention
LONHALA MAGNAIR should be used with caution in patients with urinary retention. Prescribers and patients should be alert for signs and symptoms of urinary retention (e.g., difficulty passing urine, painful urination), especially in patients with prostatic hyperplasia or bladder-neck obstruction. Instruct patients to consult a physician immediately should any of these signs or symptoms develop.
6. ADVERSE REACTIONS
The following adverse reactions are described in greater detail in other sections:
- Paradoxical bronchospasm [see Warnings and Precautions (5.2)]
- Immediate hypersensitivity reactions [see Warnings and Precautions (5.3)]
- Worsening of narrow-angle glaucoma [see Warnings and Precautions (5.4)]
- Worsening of urinary retention [see Warnings and Precautions (5.5)]
6.1. Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The LONHALA MAGNAIR safety database included 2379 subjects with COPD in two 12-week efficacy studies and one 48-week long-term safety study. A total of 431 subjects received treatment with LONHALA MAGNAIR 25 mcg twice-daily (BID). The safety data described below are based on the two 12-week trials and the one 48-week trial.
12-Week Trials
LONHALA MAGNAIR was studied in two 12-week placebo-controlled trials in subjects with COPD. In these trials, 431 subjects were treated with LONHALA MAGNAIR at the recommended dose of 25 mcg twice daily. The population had a mean age of 63 years (ranging from 40 to 87 years), with 56% males, 90% Caucasian, and a mean post-bronchodilator forced expiratory volume in one second (FEV1) percent predicted of 52% of predicted normal value (20%-80%) at study entry. The study population also included subjects with pre-existing cardiovascular disease as well as subjects with continued use of stable long-acting bronchodilator (LABA) +/- inhaled corticosteroid (ICS) and ipratropium bromide background therapy. Subjects with unstable cardiac disease, narrow-angle glaucoma, or symptomatic prostatic hypertrophy or bladder outlet obstruction were excluded from these studies.
Table 1 shows the most common adverse reactions incidence greater than or equal to 2.0% in the LONHALA MAGNAIR group and higher than placebo in the two 12-week placebo-controlled trials.
The proportion of subjects who discontinued treatment due to adverse reactions was 5% for the LONHALA MAGNAIR-treated subjects and 9% for placebo-treated subjects.
| Placebo
(N=430) N (%) | LONHALA MAGNAIR 25 mcg BID
(N=431) N (%) |
|
| Dyspnea | 13 (3.0) | 21 (4.9) |
| Urinary Tract Infection | 6 (1.4) | 9 (2.1) |
Other adverse reactions defined as events with an incidence of ≥1.0% but less than 2.0% with LONHALA MAGNAIR but more common than with placebo included the following: wheezing, upper respiratory tract infection, nasopharyngitis, oedema peripheral, and fatigue.
48-Week Trial
In a long-term open-label safety trial, 1086 subjects were treated for up to 48-weeks with LONHALA MAGNAIR 50 mcg twice-daily (N=620) or tiotropium (N=466). The demographic and baseline characteristics of the long-term safety trial were similar to those of the placebo-controlled efficacy studies described above. The adverse reactions reported in the long-term safety trial were consistent with those observed in the placebo-controlled studies of 12-weeks. Adverse reactions that occurred at a frequency greater than that seen in either active treatment dose in the pooled 12-week placebo controlled studies and ≥ 2.0% were: diarrhea, edema peripheral, bronchitis, nasopharyngitis, pneumonia, sinusitis, upper respiratory tract infection, urinary tract infection, back pain, headache, Chronic Obstructive Pulmonary Disease, cough, dyspnea, oropharyngeal pain, and hypertension.
7. DRUG INTERACTIONS
7.1. Sympathomimetics and Steroids
In clinical studies, concurrent administration of glycopyrrolate and other drugs commonly used in the treatment of COPD including sympathomimetics (long and short-acting beta2 agonists), anticholinergics (short-acting anti-muscarinic antagonists) and oral and inhaled steroids showed no increases in adverse drug reactions.
7.2. Anticholinergics
There is a potential for an additive interaction with concomitantly used anticholinergic medications. Therefore, avoid unnecessary co-administration of LONHALA MAGNAIR with other anticholinergic-containing drugs as this may lead to an increase in anticholinergic effects [see Warnings and Precautions (5.4, 5.5) and Adverse Reactions (6)].
8. USE IN SPECIFIC POPULATIONS
8.1. Pregnancy
Risk Summary
There are no adequate and well-controlled studies in pregnant women. LONHALA MAGNAIR should only be used during pregnancy if the expected benefit to the patient outweighs the potential risk to the fetus. Women should be advised to contact their physician if they become pregnant while taking LONHALA MAGNAIR. In animal reproduction studies, there were no teratogenic effects in Wistar rats and New Zealand White rabbits at inhaled doses approximating 1521 and 580 times, respectively, the maximum recommended human daily inhalation dose (MRHDID) based on an AUC comparison [see Data].
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Labor or Delivery
The potential effect of LONHALA MAGNAIR on labor and delivery is unknown. LONHALA MAGNAIR should be used during labor and delivery only if the potential benefit to the patient justifies the potential risk to the fetus.
Data
Animal Data
Developmental studies in Wistar rats and New Zealand White rabbits in which glycopyrrolate was administered by inhalation during the period of organogenesis did not result in evidence of teratogenicity at exposures approximately 1521 and 580 times, respectively, the MRHDID of LONHALA MAGNAIR based on a comparison of plasma AUC levels (maternal doses up to 3.8 mg/kg/day in rats and 4.4 mg/kg/day in rabbits).
Glycopyrrolate had no effects on peri-natal and post-natal development in rats following subcutaneous exposure of approximately 1137 times the MRHDID of LONHALA MAGNAIR based on an AUC comparison (at a maternal dose of up to 1.885 mg/kg/day).
8.2. Lactation
Risk Summary
There are no data on the presence of glycopyrrolate or its metabolites in human milk, the effects on the breastfed infant, or the effects on milk production. However, in a study of lactating rats, glycopyrrolate was present in the milk [see Data]. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for LONHALA MAGNAIR and any potential adverse effects on the breastfed infant from LONHALA MAGNAIR or from the underlying maternal condition.
8.4. Pediatric Use
LONHALA MAGNAIR is not indicated for use in children. The safety and efficacy of LONHALA MAGNAIR in pediatric patients have not been established.
8.5. Geriatric Use
Based on available data, no adjustment of the dosage of LONHALA MAGNAIR in geriatric patients is warranted. LONHALA MAGNAIR can be used at the recommended dose in elderly patients 75 years of age and older.
Of the total number of subjects in clinical studies of LONHALA MAGNAIR, 41% were aged 65 and older, while 8% were aged 75 and older. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
8.6. Renal Impairment
No dose adjustment is required for patients with mild and moderate renal impairment. The effects of renal impairment on the pharmacokinetics of glycopyrrolate have not been studied [see Clinical Pharmacology (12.3)].
8.7. Hepatic Impairment
No dose adjustment is required for patients with hepatic impairment. The effects of hepatic impairment on the pharmacokinetics of glycopyrrolate have not been studied [see Clinical Pharmacology (12.3)].
10. OVERDOSAGE
An overdose of glycopyrrolate may lead to anticholinergic signs and symptoms such as nausea, vomiting, dizziness, lightheadedness, blurred vision, increased intraocular pressure (causing pain, vision disturbances, or reddening of the eye), obstipation or difficulties in voiding.
In COPD patients, orally inhaled administration of LONHALA MAGNAIR at a total daily dose of 200 mcg for 28 consecutive days (maximum of 1 mg) was well tolerated. Pharmacokinetic results from several studies conducted in COPD patients showed that a single, well tolerated dose of 1000 mcg had a Cmax of 1534 pg/mL and AUC0-inf of 5271 pg*hr/mL. These values are approximately 44 fold and 21 fold higher, respectively, than the estimated daily Cmax of 34.5 pg/mL and AUC0-inf of 255 pg*hr/mL for a 25 mcg BID dose regimen at steady-state.
11. DESCRIPTION
LONHALA MAGNAIR consists of LONHALA vials and a MAGNAIR nebulization system. LONHALA (glycopyrrolate) Inhalation Solution is a sterile, clear, colorless, aqueous solution for oral inhalation.
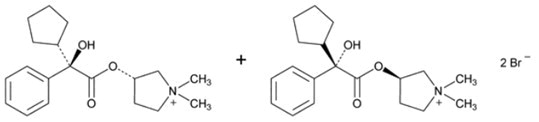
Glycopyrrolate USP, the active component of LONHALA Inhalation Solution, is chemically described as (3RS)-3-[(2SR)-(2-cyclopentyl-2-hydroxy-2-penylacetyl)oxy]-1,1-dimethlypyrrolidinium bromide. Glycopyrrolate is a synthetic quaternary ammonium compound that acts as a competitive antagonist at muscarinic acetylcholine receptors, also referred to as an anticholinergic. Glycopyrrolate, C19H28BrNO3, is a white, odorless, crystalline powder that is soluble in water and in alcohol. It has a molecular mass of 398.33. The structural formula is:
The inactive ingredients in LONHALA are: citric acid monohydrate, sodium chloride, sodium hydroxide and water for injection.
LONHALA Inhalation Solution is supplied in low-density polyethylene (LDPE) unit dose vials, each containing 1.0 mL of the solution. Each unit-dose vial contains 25 mcg of glycopyrrolate in a sterile, isotonic saline solution, pH-adjusted to 4.0 with citric acid and sodium hydroxide.
Like all other nebulized treatments, the amount delivered to the lungs will depend upon patient factors. Under standardized in vitro testing per USP<1601> adult breathing pattern (500 mL tidal volume, 15 breaths per minute, and inhalation: exhalation ratio of 1:1), the mean delivered dose from the mouthpiece was approximately 14.2 mcg of glycopyrrolate (equivalent to 11.4 mcg glycopyrronium and 56.8% label claim). The mass median aerodynamic diameter (MMAD) of the nebulized aerosol particles/droplets is 3.71 μm 95% CI (2.92 - 4.49 μm) as determined using the Next Generation Impactor (NGI) method. The mean nebulization time was approximately 2 to 3 minutes.
12. CLINICAL PHARMACOLOGY
12.1. Mechanism of Action
Glycopyrrolate is a long-acting muscarinic antagonist, which is often referred to as an anticholinergic. It has similar affinity to the subtypes of muscarinic receptors M1 to M5. In the airways, it exhibits pharmacological effects through inhibition of M3 receptor at the smooth muscle leading to bronchodilation. The competitive and reversible nature of antagonism was shown with human and animal origin receptors and isolated organ preparations. In preclinical in vitro as well as in vivo studies, prevention of methacholine and acetylcholine induced bronchoconstrictive effects was dose-dependent and lasted longer than 24 hours. The clinical relevance of these findings is unknown. The bronchodilation following inhalation of glycopyrrolate is predominantly a site-specific effect.
12.2. Pharmacodynamics
Cardiac Electrophysiology:
In the dose ranging and confirmatory clinical studies, the administration of LONHALA MAGNAIR did not demonstrate any clinically relevant changes in cardiac function including: vital signs (heart rate, blood pressure), electrocardiograms (including QTc) and Holter monitoring. In addition, no major adverse cardiovascular events (MACE) were reported following the administration of LONHALA MAGNAIR 25 mcg in any clinical study.
12.3. Pharmacokinetics
Absorption
Following oral inhalation using MAGNAIR, glycopyrrolate was rapidly absorbed and reached peak plasma levels <20 minutes post dose.
In patients with COPD, pharmacokinetic steady-state plasma levels of glycopyrrolate were reached within one week of the start of treatment. A twice daily dose regimen leads to approximately 2-3 fold accumulation of systemic glycopyrrolate exposure at steady-state.
Distribution
The in vitro human plasma protein binding of glycopyrrolate was 38% to 41% at concentrations of 1 to 10 ng/mL.
Metabolism
In vitro metabolism studies show glycopyrrolate hydroxylation resulting in a variety of mono- and bis-hydroxylated metabolites and direct hydrolysis resulting in the formation of a carboxylic acid derivative (M9). Further in vitro investigations showed that multiple CYP isoenzymes contribute to the oxidative biotransformation of glycopyrrolate and the hydrolysis to M9 is likely to be catalyzed by members from the cholinesterase family pre-systemically and/or via first pass metabolism from the swallowed dose fraction of orally inhaled glycopyrrolate.
Elimination
After intravenous administration of [3H]-labelled glycopyrrolate to humans, the mean urinary excretion of radioactivity in 48 hours amounted to 85% of the dose. A further 5% of the dose was found in the bile.
Renal elimination of parent drug accounts for about 60 to 70% of total clearance of systemically available glycopyrrolate whereas non-renal clearance processes account for about 30 to 40%. Biliary clearance contributes to the non-renal clearance, but the majority of non-renal clearance is thought to be due to metabolism.
Drug Interactions
In vitro inhibition studies demonstrated that glycopyrrolate has no relevant capacity to inhibit CYP1A2, CYP2A6, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, CYP2E1 or CYP3A4/5, the efflux transporters MDR1, MRP2 or MXR, and the uptake transporters OATP1B1, OATP1B3, OAT1, OAT3, OCT1 or OCT2.
In vitro enzyme induction studies did not indicate a clinically relevant induction by glycopyrrolate for cytochrome P450 isoenzymes, or for UGT1A1 and the transporters MDR1 and MRP2.
There is potential for additive interaction with concomitantly used anticholinergic medications. Therefore, avoid coadministration of LONHALA MAGNAIR with other anticholinergic-containing drugs as this may lead to an increase in anticholinergic effects [see Warnings and Precautions (5.4, 5.5) and Adverse Reactions (6)].
Specific Populations
A population pharmacokinetic analysis of data in COPD patients indicated no clinically relevant effect of age (41 to 80 years) or body weight (40.1 to 154.8 kg) on systemic exposure to glycopyrrolate. In addition, there was no evidence of clinically significant ethnic/race effect.
Renal Impairment
The effects of renal impairment on the pharmacokinetics of glycopyrrolate have not been studied [see Use in Specific Populations (8.6)].
Hepatic Impairment
The effects of hepatic impairment on the pharmacokinetics of glycopyrrolate have not been studied. Glycopyrrolate is cleared predominantly from systemic circulation by renal excretion [see Use in Specific Populations (8.7)].
13. NONCLINICAL TOXICOLOGY
13.1. Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies of glycopyrrolate did not result in an increase in the incidence of tumors in a 2-year inhalation study of glycopyrrolate in Wistar rats at doses up to 0.56 mg/kg/day, approximately 143 times the MRHDID of LONHALA MAGNAIR in adults on an AUC basis. Also, no evidence of tumorigenicity occurred in a 26-week oral (gavage) study in male and female TgrasH2 mice that received glycopyrrolate at doses up to 93.8 and 125.1 mg/kg/day, respectively, approximately 66 times the MRHDID of LONHALA MAGNAIR.
Glycopyrrolate was not mutagenic in the following genotoxicity assays: the in vitro Ames assay, in vitro human lymphocyte chromosomal aberration assay, and in vivo rat bone marrow micronucleus assay.
Impairment of fertility was observed in male and female Wistar rats at a subcutaneous glycopyrrolate dose of 1.88 mg/kg/day (approximately and 2035 and 1136 times, respectively, the MRHD of LONHALA MAGNAIR on an AUC basis) based upon findings of decreased implantation sites and corresponding reduction of live fetuses. No effects on fertility and reproductive performance occurred in male and female rats at a subcutaneous glycopyrrolate dose of 0.63 mg/kg/day, approximately 384 times the MRHD of LONHALA MAGNAIR on an AUC basis).
14. CLINICAL STUDIES
The safety and efficacy of LONHALA MAGNAIR were evaluated in 2 dose-ranging studies, 2 placebo-controlled confirmatory studies (12-week studies), and a 48-week long-term safety study. The efficacy of LONHALA MAGNAIR is based primarily on the dose-ranging studies in 378 subjects with COPD and the 2 placebo-controlled confirmatory studies in 1293 subjects with COPD.
14.1. Dose Ranging Studies
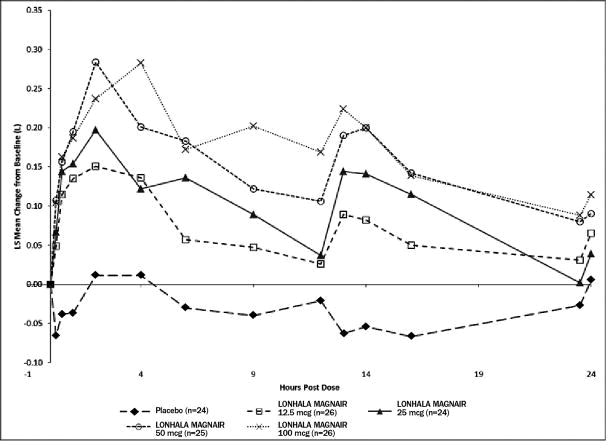
Dose selection for the confirmatory COPD studies for LONHALA MAGNAIR was supported by two studies. Study A was a randomized, double-blind, placebo-controlled, parallel arm study with a 28-day treatment period. The study included LONHALA MAGNAIR doses of placebo, 12.5 mcg, 25 mcg, 50 mcg, and 100 mcg twice daily. The Study demonstrated a dose-response effect on peak and trough FEV1 over 24-hour dosing period in subjects treated with LONHALA MAGNAIR twice daily [Figure 1 (Day 1) and Figure 2 (Day 28)]. The LS mean differences in trough FEV1 from baseline after 28 days compared to placebo for the 12.5 mcg, 25 mcg, 50 mcg, and 100 mcg twice daily doses were 0.117 L (95% CI: 0.037, 0.197); 0.128 L (95% CI: 0.048, 0.209), 0.146 L (95% CI: 0.067, 0.226), and 0.177 L (95% CI: 0.099, 0.255), respectively. In Study A, all subjects in each treatment group (N=282) had FEV1 AUC0-12h serial spirometry assessments while a subset of subjects (N=125; shown in Figure 1 and Figure 2 below) had extended FEV1 AUC12-24h assessments on Days 1 and 28.
Study B was a randomized, six-way, crossover study with 7-day treatment periods separated by 5-7-day washout periods. Study B included LONHALA MAGNAIR doses of placebo, 3 mcg, 6.25 mcg, 12.5 mcg, and 50 mcg twice daily with aclidinium bromide 400 mcg BID as an active control.
The dose-ranging results from Study A and Study B supported the evaluation of LONHALA MAGNAIR 25 mcg and 50 mcg twice-daily in the confirmatory COPD trials. The results of Study A are reported in Figure 1 below.
Figure 1: LS Mean Change from Baseline in FEV1 (L) Over Time on Day 1 (Study A)
Figure 2: LS Mean Change from Baseline in FEV1 (L) Over Time on Day 28 (Study A)
14.2. Confirmatory Studies
There were 2 confirmatory studies (Study 1 and Study 2) for LONHALA MAGNAIR. Both studies were randomized, double-blind, placebo-controlled, parallel-group 12-week studies in subjects with COPD designed to evaluate the efficacy of LONHALA MAGNAIR on lung function. These studies treated subjects who had a clinical diagnosis of COPD, were 40 years of age or older, had a history of smoking greater than or equal to 10 pack-years, a post –bronchodilator FEV1 less than or equal to 80% of predicted, and an FEV1/FVC ratio less than 0.7. Subjects also had pre-existing or concurrent cardiovascular disease and stable, background LABA ± ICS and SAMA use were permitted. Subjects in Study 1 and Study 2 had a mean age of 63 years, were primarily male (56%), Caucasian (90%), and current smokers (53%) with an average smoking history of 52 pack-years. At screening, the mean post-bronchodilator percent predicted FEV1 was 52% (range: 20% to 80%), the mean post-bronchodilator percent FEV1/FVC was 54% (range: 20% to 71%), and the mean percent reversibility was 18% (range: -33% to 86%).
Study 1 and Study 2 evaluated LONHALA MAGNAIR (glycopyrrolate) 25 mcg and 50 mcg twice-daily and placebo twice-daily. The primary endpoint was the change from baseline in trough FEV1 at Day 84 compared with placebo. LONHALA MAGNAIR twice-daily demonstrated a larger increase in LS mean change from baseline in trough FEV1 compared to placebo. Compared to LONHALA MAGNAIR 25 mcg twice daily, LONHALA MAGNAIR 50 mcg twice daily did not provide sufficient additional benefit on a variety of endpoints, including FEV1, to support use of higher doses. Table 2 presents the results from Studies 1 and 2 for LONHALA MAGNAIR 25 mcg twice daily.
|
*Study results are from a treatment policy strategy which analyzes all collected data, including data for some patients who discontinued study treatment prior to Week 12 and may have received other COPD treatment but were followed. Analyses of efficacy data measured only while on randomized blinded study treatment showed similar results. |
|||||
| Treatment | N | Change from baseline LS Mean (SE) | Comparison | Treatment Difference LS Mean (SE) | 95% CI |
| Study 1 | |||||
| LONHALA MAGNAIR 25 mcg BID | 217 | 0.089 (0.014) | LONHALA MAGNAIR - Placebo | 0.096 (0.019) | 0.059, 0.133 |
| Placebo | 218 | -0.008 (0.014) | |||
| Study 2 | |||||
| LONHALA MAGNAIR 25 mcg BID | 214 | 0.092 (0.014) | LONHALA MAGNAIR - Placebo | 0.081 (0.020) | 0.042, 0.120 |
| Placebo | 212 | 0.011 (0.015) | |||
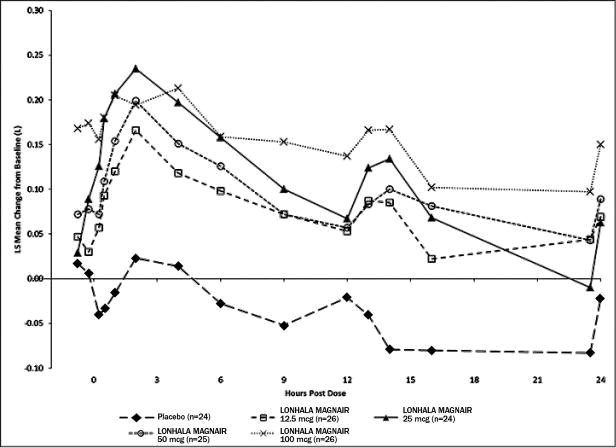
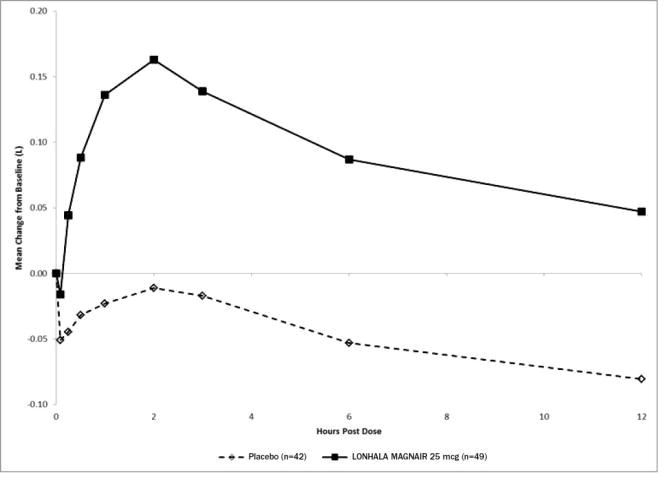
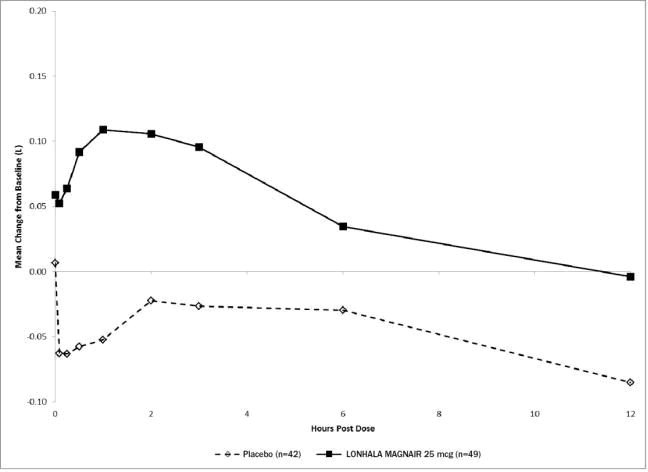
In Study 1, serial spirometric evaluations throughout the 12-hour dosing interval were performed in a subset of subjects on Day 1 and Day 84. The spirometric curves from Study 1 on Day 1 and Day 84 are displayed in Figure 3 and Figure 4.
Figure 3: Mean Change from Baseline in FEV1 (L) Over Time on Day 1 (Substudy Population)
Figure 4: Mean Change from Baseline in FEV1 (L) Over Time on Day 84 (Substudy Population)
The peak FEV1 was defined as the highest postdose FEV1 within the first 12 hours after morning dosing for each subject on Days 1 and 84, respectively, for the substudy population.
The mean peak FEV1 improvement from baseline for LONHALA MAGNAIR on Day 1 and on Day 84 in the subset of subjects was 0.228 L and 0.214 L (Study 1) respectively.
The St. George's Respiratory Questionnaire (SGRQ) was assessed in Studies 1 and 2. In Study 1, the SGRQ responder rate (defined as an improvement in score of 4 or more as threshold) for the LONHALA MAGNAIR 25 mcg treatment arm was 51% compared to 40% for placebo [Odds Ratio:1.55; 95% CI: 1.03, 2.33]. In Study 2, the SGRQ responder rate for the LONHALA MAGNAIR 25 mcg treatment arm was 41% compared to 29% for placebo [Odds Ratio: 1.72; 95% CI: 1.11, 2.67].
16. HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
LONHALA MAGNAIR is supplied as a 1 mL sterile, clear, colorless, aqueous solution in low-density polyethylene (LDPE) unit-dose vials overwrapped in foil. LONHALA MAGNAIR is available in a Starter Kit containing 60 unit-dose vials packaged with one MAGNAIR, and FDA approved patient labeling. LONHALA MAGNAIR is also supplied in a Refill Kit containing 60 unit-dose vials packaged with a MAGNAIR Replacement Handset and FDA approved patient labeling.
| Package Configuration | Dosage Strength | NDC |
| Starter Kit with 30 day supply (30 foil pouches with 2 vials per pouch) and complete MAGNAIR Nebulizer System | 25 mcg | NDC: 63402-201-00 |
| Refill Kit with 30 day supply (30 foil pouches with 2 vials per pouch) and MAGNAIR Replacement Handset | 25 mcg | NDC: 63402-301-01 |
16.2 Storage and Handling
Store LONHALA Inhalation Solution in the protective foil pouch at 20°-25°C (68°-77°F) [see USP Controlled Room Temperature].
- LONHALA vials should be used with MAGNAIR only. Do not use MAGNAIR with any other vials.
- Store LONHALA vials in the protective foil pouch. After opening the foil pouch, unused unit-dose vials should be returned to, and stored in, the foil pouch. Once a foil pouch is opened, discard the vials if not used within 7 days. An opened unit-dose vial should be used right away. Discard any unit-dose vial if the solution is not colorless.
Always use the MAGNAIR Replacement Handset parts that come with each LONHALA MAGNAIR refill prescription.
Keep out of the reach of children.
17. PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
Not for Acute Symptoms: Inform patients that LONHALA MAGNAIR is not meant to relieve acute symptoms of COPD and extra doses should not be used for that purpose. Advise them to treat acute symptoms with a rescue inhaler such as albuterol. Provide patients with such medicine and instruct them in how it should be used [see Warnings and Precautions (5.1)].
Instruct patients to seek medical attention immediately if they experience any of the following:
- Symptoms get worse
- Need for more inhalations than usual of their rescue inhaler
Patients should not stop therapy with LONHALA MAGNAIR without physician/provider guidance since symptoms may recur after discontinuation.
Paradoxical Bronchospasm: Inform patients that LONHALA MAGNAIR can cause paradoxical bronchospasm. If paradoxical bronchospasm occurs, instruct patients to discontinue LONHALA MAGNAIR.
Worsening of Narrow-Angle Glaucoma: Instruct patients to be alert for signs and symptoms of acute narrow-angle glaucoma (e.g., eye pain or discomfort, blurred vision, visual halos or colored images in association with red eyes from conjunctival congestion and corneal edema). Instruct patients to consult a physician immediately if any of these signs or symptoms develop.
Worsening of Urinary Retention: Instruct patients to be alert for signs and symptoms of urinary retention (e.g., difficulty passing urine, painful urination). Instruct patients to consult a physician immediately if any of these signs or symptoms develop.
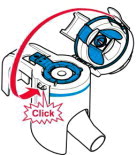
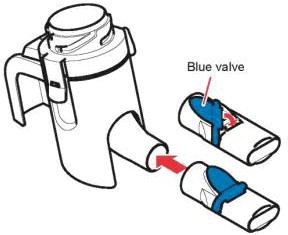
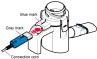
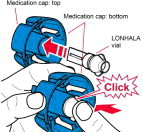
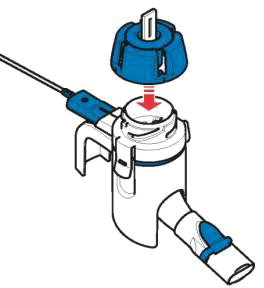
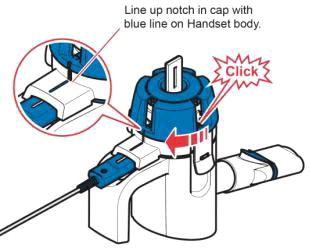
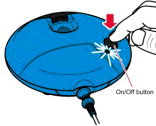
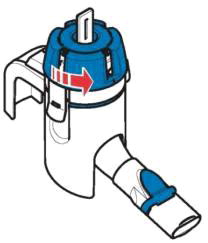
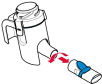
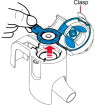
Instructions for Administering LONHALA MAGNAIR
It is important for patients to understand how to correctly administer LONHALA vials using MAGNAIR [see Instructions for Use]. Instruct patients that LONHALA vials should only be administered via MAGNAIR and MAGNAIR should not be used for administering other medications. Patients should be instructed not to inject or swallow the LONHALA solution.
Instruct patients to store LONHALA vials in the sealed foil pouch and to only open the foil pouch to remove a LONHALA vial immediately before use. Inform patients that unopened vials should be returned to the opened foil pouch for use at their next treatment and discarded if not used within 7 days or it may not be as effective.
Inform patients to use one inhalation of LONHALA MAGNAIR orally twice daily (1 vial in the morning and 1 vial in the evening) at the same time every day.
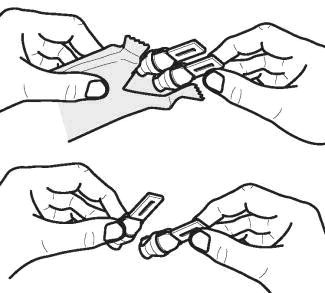
Inform patients that if they miss a dose of LONHALA MAGNAIR, they should use their next vial at the usual time. Instruct patients to not use 2 vials at one time and to not use more than 2 vials in a day. Patients should throw the plastic dispensing vials away immediately after use. Due to their small size, the vials pose a danger of choking to young children.
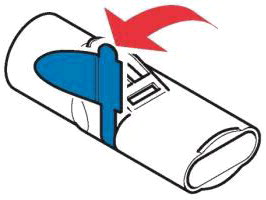
Inform patients treated with LONHALA MAGNAIR that a Refill Kit will be provided to them on a monthly basis. The Refill Kit will include foil pouches containing 60 vials of LONHALA (2 vials of LONHALA in each pouch; 1 vial per dose), and 1 MAGNAIR Replacement Handset (containing only these replacement parts: Medication cap, Handset body, Mouthpiece, and Aerosol head; Manufacturer's Instructions for Use booklet).
Important: Instruct patients to throw away the old Handset parts after using 60 vials of LONHALA and use the replacement Handset parts with the next 60 vials of LONHALA.
Manufactured for Sunovion Respiratory Development Inc.,
a direct wholly-owned subsidiary of Sunovion Pharmaceuticals Inc.
84 Waterford Drive
Marlborough, MA 01752 USA
For Sunovion customer service, call 1-888-394-7377.
For medical information, call 1-800-739-0565.
To report suspected adverse events, call 1-877-737-7226.
©2020 Sunovion Pharmaceuticals Inc. All rights reserved.
| PATIENT INFORMATION
LONHALA MAGNAIR (lon-HAH-luh MAGG-nair) (glycopyrrolate) inhalation solution, for oral inhalation use |
|||
| Important: For oral inhalation only. Do not inject or swallow the LONHALA medicine. LONHALA vials are used only with the MAGNAIR device. Do not use MAGNAIR with any other medicine. | |||
| Read this Patient Information that comes with LONHALA MAGNAIR before you start using it and each time you get a refill. There may be new information. This Patient Information does not take the place of talking to your healthcare provider about your medical condition or treatment. | |||
| What is LONHALA MAGNAIR?
LONHALA MAGNAIR is an anticholinergic medicine known as glycopyrrolate.
|
|||
Do not use LONHALA MAGNAIR if you:
|
|||
Before using LONHALA MAGNAIR, tell your healthcare provider about all of your medical conditions, including if you:
|
|||
| Tell your healthcare provider about all the medicines you take, including prescription medicines and over-the-counter medicines, vitamins, and herbal supplements. LONHALA MAGNAIR may affect the way other medicines work, and other medicines can affect how LONHALA MAGNAIR works. Using LONHALA MAGNAIR with other medicines may cause serious side effects. Especially tell your healthcare provider if you take anticholinergics (including umeclidinium, tiotropium, ipratropium, aclidinium, glycopyrrolate). Know the medicines you take. Keep a list of your medicines with you and show it to your healthcare provider and pharmacist when you get a new medicine. |
|||
| How should I use LONHALA MAGNAIR?
Read the step-by-step instructions for using LONHALA MAGNAIR at the end of this Patient Information leaflet and the Manufacturer's Instructions for Use booklet. The Manufacturer's Instructions for Use booklet provides complete information about how to put together (assemble), prepare, use, care for, and trouble-shoot your MAGNAIR nebulizer system.
|
|||
| What are the possible side effects of LONHALA MAGNAIR?
LONHALA MAGNAIR can cause serious side effects, including:
|
|||
|
| ||
|
|||
|
| ||
If you have any of these symptoms, stop taking LONHALA MAGNAIR and call your healthcare provider right away before using another dose.
|
|||
|
| ||
|
| ||
| If you have any of these symptoms, stop taking LONHALA MAGNAIR and call your healthcare provider right away before taking another dose. Common side effects of LONHALA MAGNAIR include shortness of breath, and urinary tract infections. These are not all of the possible side effects of LONHALA MAGNAIR. Call your doctor or pharmacist for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
How should I store LONHALA MAGNAIR?
|
|||
After opening the protective foil pouch, unused LONHALA vials should be returned to, and stored in, the opened foil pouch. Once a foil pouch is opened, discard the vials if not used within 7 days.
|
|||
| General information about the safe and effective use of LONHALA MAGNAIR.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use LONHALA MAGNAIR for a condition for which it was not prescribed. Do not give LONHALA MAGNAIR to other people, even if they have the same symptoms you have. It may harm them. You can ask your healthcare provider or pharmacist for information about LONHALA MAGNAIR that is written for healthcare professionals. |
|||
|
What are the ingredients in LONHALA MAGNAIR?
Active ingredient: glycopyrrolate
|
|||
PRINCIPAL DISPLAY PANEL – Starter Kit
NDC 63402-201-00
Lonhala™Magnair™
(glycopyrrolate) Inhalation Solution
25mcg/1mL
Starter Kit
Store at 20°-25°C (68°-77°F) [see USP Controlled Room Temperature].
PRINCIPAL DISPLAY PANEL – Refill Kit
NDC 63402-301-01
Lonhala™Magnair™
(glycopyrrolate) Inhalation Solution
25mcg/1mL
Refill Kit
Store at 20°-25°C (68°-77°F) [see USP Controlled Room Temperature].
PRINCIPAL DISPLAY PANEL – Product Carton - 60 vial carton
NDC 63402-101-30
Lonhala™
(glycopyrrolate) Inhalation Solution
25 mcg/1 mL
For Oral Inhalation Only
With Magnair™ Nebulizer System
Store at 20°-25°C (68°-77°F) [see USP Controlled Room Temperature].
Rx Only
Sunovion
PRINCIPAL DISPLAY PANEL – Product Foil Pouch - 2 vial pouch
NDC 63402-101-02
Lonhala™
(glycopyrrolate) Inhalation Solution
Two 1 mL Sterile Unit-Dose Vials
25 mcg/1 mL
For Oral Inhalation Only
With Magnair™ Nebulizer System
Store at 20°-25°C (68°-77°F) [see USP Controlled Room Temperature].
Rx Only
Sunovion
| LONHALA MAGNAIR
glycopyrrolate solution |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| LONHALA MAGNAIR
glycopyrrolate solution |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Sunovion Pharmaceuticals Inc. (131661746) |









 are registered trademarks of Sunovion Pharmaceuticals Inc. MAGNAIR is a registered trademark of PARI Pharma GmbH, used under license.
are registered trademarks of Sunovion Pharmaceuticals Inc. MAGNAIR is a registered trademark of PARI Pharma GmbH, used under license. are registered trademarks of Sumitomo Dainippon Pharma Co., Ltd. Sunovion Pharmaceuticals Inc. is a U.S. subsidiary of Sumitomo Dainippon Pharma Co., Ltd.
are registered trademarks of Sumitomo Dainippon Pharma Co., Ltd. Sunovion Pharmaceuticals Inc. is a U.S. subsidiary of Sumitomo Dainippon Pharma Co., Ltd. are registered trademarks of PARI GmbH.
are registered trademarks of PARI GmbH.