Label: FAMOTIDINE tablet, film coated
-
NDC Code(s):
58602-705-03,
58602-705-09,
58602-705-12,
58602-705-14, view more58602-705-15, 58602-705-19, 58602-705-32, 58602-705-34, 58602-705-39, 58602-705-40, 58602-705-41, 58602-705-44, 58602-705-84, 58602-705-88, 58602-705-93
- Packager: Aurohealth LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated April 28, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
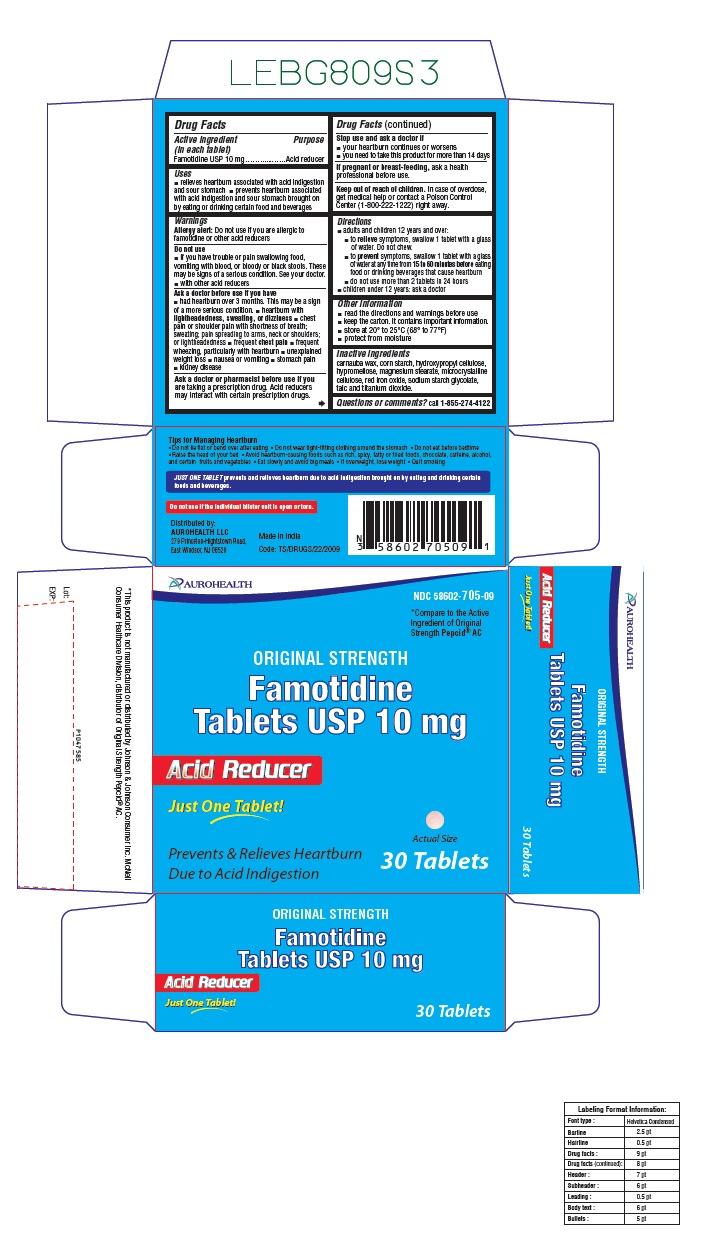
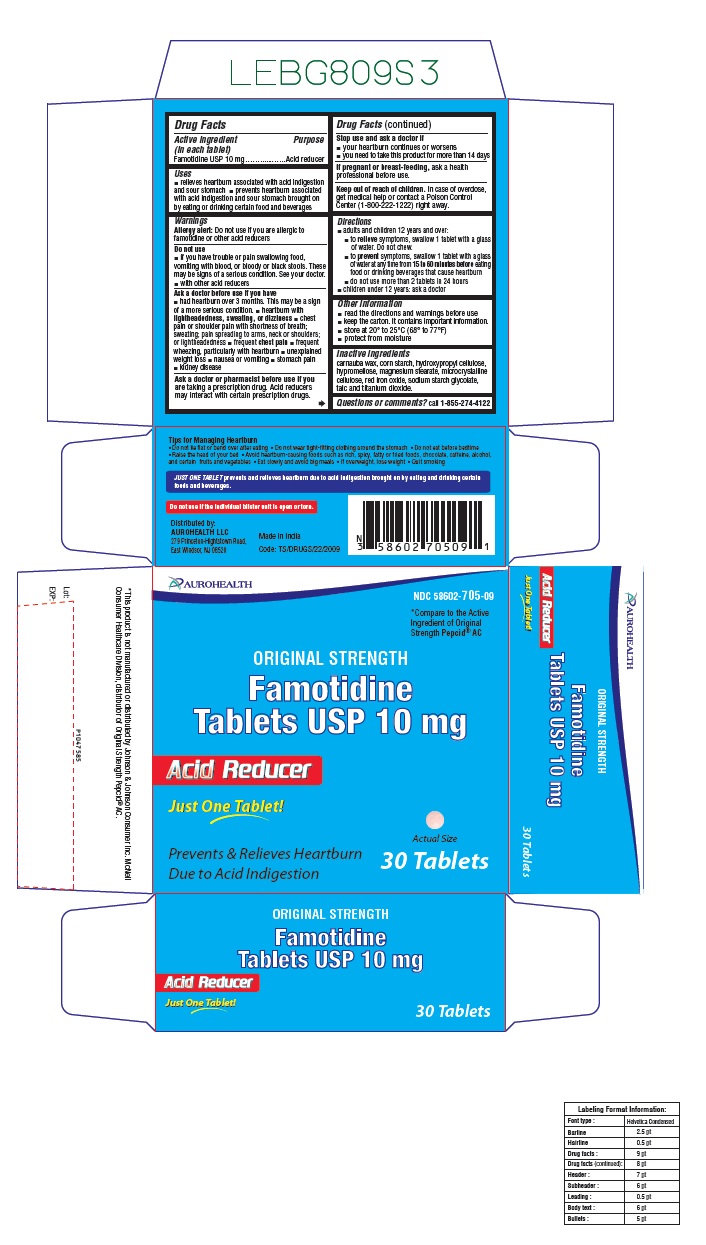
- Active ingredient (in each tablet)
- Purpose
- Uses
- Warnings
- Do not use
-
Ask a doctor before use if you have
- had heartburn over 3 months. This may be a sign of a more serious condition.
- heartburn with lightheadedness, sweating, or dizziness
- chest pain or shoulder pain with shortness of breath; sweating; pain spreading to arms, neck or shoulders; or lightheadedness
- frequent chest pain
- frequent wheezing, particularly with heartburn
- unexplained weight loss
- nausea or vomiting
- stomach pain
- kidney disease
- Ask a doctor or pharmacist before use if you are
- Stop use and ask a doctor if
- If pregnant or breast-feeding,
- Keep out of reach of children
-
Directions
- adults and children 12 years and over:
- to relieve symptoms, swallow 1 tablet with a glass of water. Do not chew.
- to prevent symptoms, swallow 1 tablet with a glass of water at any time from 15 to 60 minutes before eating food or drinking beverages that cause heartburn
- do not use more than 2 tablets in 24 hours
- children under 12 years: ask a doctor
- adults and children 12 years and over:
- Other information
- Inactive ingredients
-
Questions or comments?
call 1-855-274-4122
Tips for Managing Heartburn- Do not lie flat or bend over after eating
- Do not wear tight-fitting clothing around the stomach
- Do not eat before bedtime
- Raise the head of your bed
- Avoid heartburn-causing foods such as rich, spicy, fatty or fried foods, chocolate, caffeine, alcohol, and certain fruits and vegetables
- Eat slowly and avoid big meals
- If overweight, lose weight
- Quit smoking
JUST ONE TABLET prevents and relieves heartburn due to acid indigestion brought on by eating and drinking certain foods and beverages.
Do not use if carton is open or if printed foil seal under bottle cap is open or torn.
Distributed by:
AUROHEALTH LLC
279 Princeton-Hightstown Road,
East Windsor, NJ 08520
Made in India
Code: TS/DRUGS/22/2009
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL -10 mg (90 Tablets Container Label)
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL -10 mg (90 Tablets Container Carton Label)
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL -10 mg, Blister Carton 30 Tablets
-
INGREDIENTS AND APPEARANCE
FAMOTIDINE
famotidine tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58602-705 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FAMOTIDINE (UNII: 5QZO15J2Z8) (FAMOTIDINE - UNII:5QZO15J2Z8) FAMOTIDINE 10 mg Inactive Ingredients Ingredient Name Strength CARNAUBA WAX (UNII: R12CBM0EIZ) STARCH, CORN (UNII: O8232NY3SJ) HYDROXYPROPYL CELLULOSE (1600000 WAMW) (UNII: RFW2ET671P) HYPROMELLOSE 2910 (5 MPA.S) (UNII: R75537T0T4) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) FERRIC OXIDE RED (UNII: 1K09F3G675) SODIUM STARCH GLYCOLATE TYPE A CORN (UNII: AG9B65PV6B) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color PINK Score no score Shape ROUND (biconvex) Size 5mm Flavor Imprint Code CC;58 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58602-705-15 1 in 1 CARTON 04/26/2016 1 60 in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:58602-705-19 1 in 1 CARTON 04/26/2016 2 90 in 1 BOTTLE; Type 0: Not a Combination Product 3 NDC:58602-705-84 1 in 1 CARTON 05/13/2019 3 30 in 1 BOTTLE; Type 0: Not a Combination Product 4 NDC:58602-705-09 3 in 1 CARTON 04/26/2016 4 NDC:58602-705-03 10 in 1 BLISTER PACK; Type 0: Not a Combination Product 5 NDC:58602-705-12 4 in 1 CARTON 04/26/2016 5 NDC:58602-705-03 10 in 1 BLISTER PACK; Type 0: Not a Combination Product 6 NDC:58602-705-93 90 in 1 BOTTLE; Type 0: Not a Combination Product 04/26/2016 7 NDC:58602-705-14 1 in 1 CARTON 12/21/2019 7 50 in 1 BOTTLE; Type 0: Not a Combination Product 8 NDC:58602-705-34 1 in 1 CARTON 07/22/2020 8 200 in 1 BOTTLE; Type 0: Not a Combination Product 9 NDC:58602-705-39 1 in 1 CARTON 07/22/2020 9 365 in 1 BOTTLE; Type 0: Not a Combination Product 10 NDC:58602-705-44 1 in 1 CARTON 07/22/2020 10 400 in 1 BOTTLE; Type 0: Not a Combination Product 11 NDC:58602-705-40 1 in 1 CARTON 07/22/2020 11 500 in 1 BOTTLE; Type 0: Not a Combination Product 12 NDC:58602-705-88 1 in 1 CARTON 07/22/2020 12 750 in 1 BOTTLE; Type 0: Not a Combination Product 13 NDC:58602-705-41 1 in 1 CARTON 09/04/2020 13 1000 in 1 BOTTLE; Type 0: Not a Combination Product 14 NDC:58602-705-32 1 in 1 CARTON 02/18/2022 14 180 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA206531 04/26/2016 Labeler - Aurohealth LLC (078728447) Establishment Name Address ID/FEI Business Operations APL HEALTHCARE LIMITED 650844777 ANALYSIS(58602-705) , MANUFACTURE(58602-705) Establishment Name Address ID/FEI Business Operations Aurobindo Pharma Limited 650381903 ANALYSIS(58602-705) , MANUFACTURE(58602-705)