4 HOUR ALLERGY RELIEF- chlorpheniramine maleate tablet
Better Living Brands, LLC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Signature Care 44-194
Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- itchy, watery eyes
- sneezing
- itching of the nose or throat
Warnings
Ask a doctor before use if you have
- glaucoma
- a breathing problem such as emphysema or chronic bronchitis
- trouble urinating due to an enlarged prostate gland
Directions
| adults and children 12 years of age and over | 1 tablet every 4 to 6 hours. Do not take more than 6 tablets in 24 hours. |
| children 6 to under 12 years of age | 1/2 tablet (break tablet in half) every 4 to 6 hours. Do not exceed 3 whole tablets in 24 hours. |
|
children under 6 years | do not use this product in children under 6 years of age |
Other information
- TAMPER EVIDENT: DO NOT USE IF OUTER PACKAGE IS OPENED OR BLISTER IS TORN OR BROKEN
- store at 25ºC (77ºF); excursions permitted between 15º-30ºC (59º-86ºF)
- protect from excessive moisture
- see end flap for expiration date and lot number
Inactive ingredients
corn starch, D&C yellow #10 aluminum lake, lactose anhydrous, magnesium stearate, microcrystalline cellulose
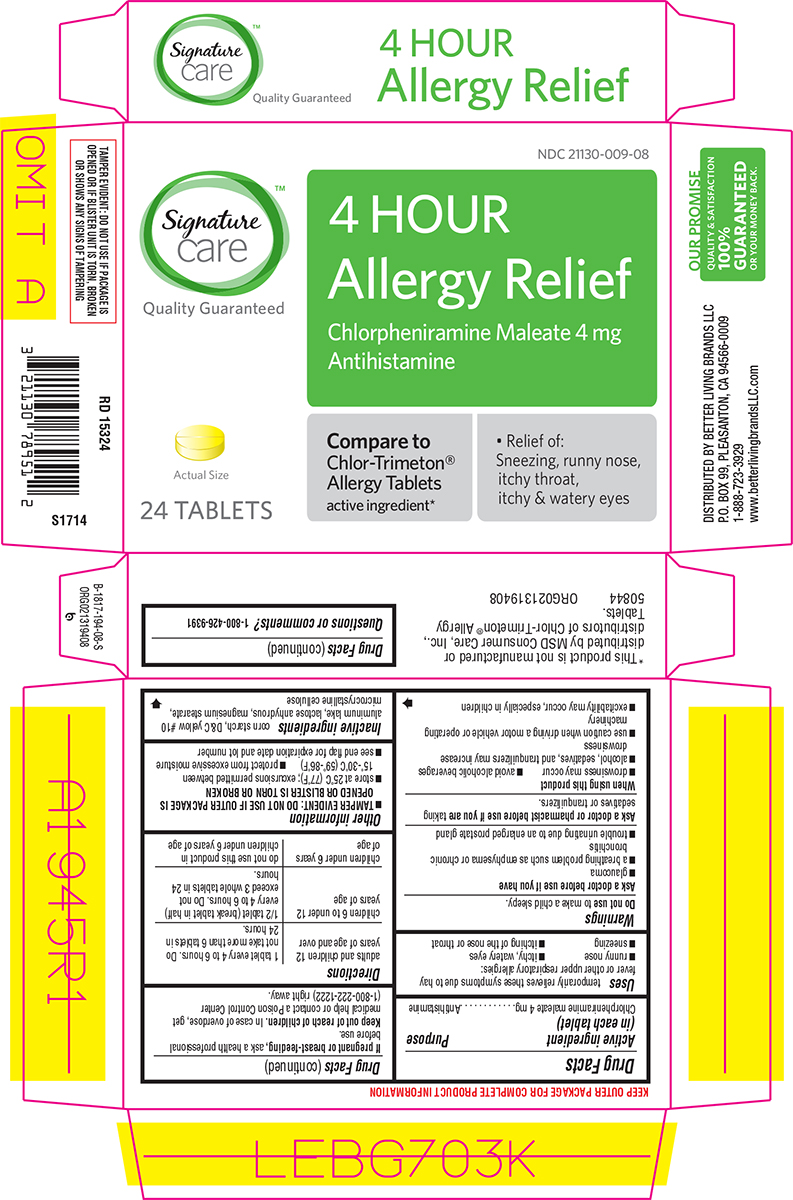
Principal Display Panel
Signature™
care
Quality Guaranteed
NDC 21130-009-08
4 HOUR
Allergy Relief
Chlorpheniramine Maleate 4 mg
Antihistamine
Actual Size
24 TABLETS
Compare to
Chlor-Trimeton®
Allergy Tablets
active ingredient*
• Relief of:
Sneezing, runny nose,
itchy throat,
itchy & watery eyes
TAMPER EVIDENT: DO NOT USE IF PACKAGE IS
OPENED OR IF BLISTER UNIT IS TORN, BROKEN
OR SHOWS ANY SIGNS OF TAMPERING
*This product is not manufactured or
distributed by MSD Consumer Care, Inc.,
distributors of Chlor-Trimeton® Allergy
Tablets.
50844 ORG021319408
DISTRIBUTED BY BETTER LIVING BRANDS LLC
P.O. BOX 99, PLEASANTON, CA 94566-0009
1-888-723-3929
www.betterlivingbrandsLLC.com
OUR PROMISE
QUALITY & SATISFACTION
100%
GUARANTEED
OR YOUR MONEY BACK.
Signature Care 44-194
| 4 HOUR ALLERGY RELIEF
chlorpheniramine maleate tablet |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Better Living Brands, LLC (009137209) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 038154464 | PACK(21130-009) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 832867894 | MANUFACTURE(21130-009) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 832867837 | PACK(21130-009) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 967626305 | PACK(21130-009) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 868734088 | PACK(21130-009) | |