BENAZEPRIL HYDROCHLORIDE AND HYDROCHLOROTHIAZIDE- benazepril hydrochloride and hydrochlorothiazide tablet, film coated
Upsher-Smith Laboratories, LLC
----------
Benazepril Hydrochloride and Hydrochlorothiazide Tablets
WARNING: FETAL TOXICITY
When pregnancy is detected, discontinue benazepril hydrochloride and hydrochlorothiazide as soon as possible.
Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus. (See WARNINGS, Fetal Toxicity).
DESCRIPTION
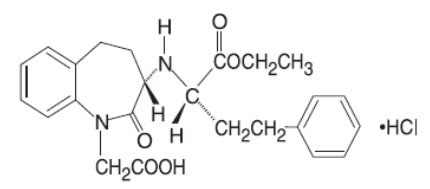
Benazepril hydrochloride, USP is a white to off-white crystalline powder, soluble (>100 mg/mL) in water, in ethanol, and in methanol. Benazepril hydrochloride's chemical name is 3-[[1-(ethoxycarbonyl)-3-phenyl-(1S)-propyl]amino]-2,3,4,5-tetrahydro-2-oxo-1H-1-(3S)-benzazepine-1-acetic acid monohydrochloride; its structural formula is:
Its molecular formula is C24H28N2O5•HCl, and its molecular weight is 460.96.
Benazeprilat, the active metabolite of benazepril, is a nonsulfhydryl angiotensin-converting enzyme inhibitor. Benazepril is converted to benazeprilat by hepatic cleavage of the ester group.
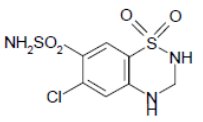
Hydrochlorothiazide, USP is a white, or practically white, practically odorless, crystalline powder. It is slightly soluble in water; freely soluble in sodium hydroxide solution, in n-butylamine, and in dimethylformamide; sparingly soluble in methanol; and insoluble in ether, in chloroform, and in dilute mineral acids. Hydrochlorothiazide's chemical name is 6-chloro-3,4-dihydro-2H-1,2,4-benzothiadiazine-7-sulfonamide 1,1-dioxide; its structural formula is:
Its molecular formula is C7H8ClN3O4S2, and its molecular weight is 297.73. Hydrochlorothiazide is a thiazide diuretic.
The tablets are a combination of benazepril hydrochloride and hydrochlorothiazide USP. They are formulated for oral administration with a combination of 10 mg, or 20 mg of benazepril hydrochloride and 12.5 mg, or 25 mg of hydrochlorothiazide USP. The inactive ingredients of the tablets are colloidal silicon dioxide, crospovidone, hydrogenated castor oil, lactose monohydrate, poloxamer, polyethylene glycol, pregelatinized starch (corn), titanium dioxide and zinc stearate. The 10 mg/12.5 mg tablets also contain FD&C red No. 40, FD&C yellow No. 6, polyvinyl alcohol and talc. The 20 mg/12.5 mg tablets also contain FD&C blue No. 2, FD&C red No. 40, hypromellose and polysorbate. The 20 mg/25 mg tablets also contain FD&C red No. 40, hypromellose and polysorbate.
CLINICAL PHARMACOLOGY
Mechanism of Action
Benazepril and benazeprilat inhibit angiotensin-converting enzyme (ACE) in human subjects and in animals. ACE is a peptidyl dipeptidase that catalyzes the conversion of angiotensin I to the vasoconstrictor substance, angiotensin II. Angiotensin II also stimulates aldosterone secretion by the adrenal cortex.
Inhibition of ACE results in decreased plasma angiotensin II, which leads to decreased vasopressor activity and to decreased aldosterone secretion. The latter decrease may result in a small increase of serum potassium. Hypertensive patients treated with benazepril alone for up to 52 weeks had elevations of serum potassium of up to 0.2 mEq/L. Similar patients treated with benazepril and hydrochlorothiazide for up to 24 weeks had no consistent changes in their serum potassium (see PRECAUTIONS).
Removal of angiotensin II negative feedback on renin secretion leads to increased plasma renin activity. In animal studies, benazepril had no inhibitory effect on the vasopressor response to angiotensin II and did not interfere with the hemodynamic effects of the autonomic neurotransmitters acetylcholine, epinephrine, and norepinephrine.
ACE is identical to kininase, an enzyme that degrades bradykinin. Whether increased levels of bradykinin, a potent vasodepressor peptide, play a role in the therapeutic effects of benazepril hydrochloride and hydrochlorothiazide remains to be elucidated.
While the mechanism through which benazepril lowers blood pressure is believed to be primarily suppression of the renin-angiotensin-aldosterone system, benazepril has an antihypertensive effect even in patients with low-renin hypertension.
Hydrochlorothiazide is a thiazide diuretic. Thiazides affect the renal tubular mechanisms of electrolyte reabsorption, directly increasing excretion of sodium and chloride in approximately equivalent amounts. Indirectly, the diuretic action of hydrochlorothiazide reduces plasma volume, with consequent increases in plasma renin activity, increases in aldosterone secretion, increases in urinary potassium loss, and decreases in serum potassium. The renin-aldosterone link is mediated by angiotensin, so coadministration of an ACE inhibitor tends to reverse the potassium loss associated with these diuretics.
The mechanism of the antihypertensive effect of thiazides is unknown.
Pharmacokinetics and Metabolism
Following oral administration of benazepril hydrochloride and hydrochlorothiazide, peak plasma concentrations of benazepril are reached within 0.5 to 1.0 hours. As determined by urinary recovery, the extent of absorption is at least 37%. In fasting subjects, the rate and extent of absorption of benazepril and hydrochlorothiazide from benazepril hydrochloride and hydrochlorothiazide are not different, respectively, from the rate and extent of absorption of benazepril and hydrochlorothiazide from immediate-release monotherapy formulations.
The estimated absolute bioavailability of hydrochlorothiazide after oral administration is about 70%. Peak plasma hydrochlorothiazide concentrations (Cmax) are reached within 2 to 5 hours after oral administration. Hydrochlorothiazide binds to albumin (40% to 70%) and distributes into erythrocytes.
The absorption of benazepril from benazepril hydrochloride tablets is not influenced by the presence of food in the gastrointestinal tract. There is no clinically significant effect of food on the bioavailability of hydrochlorothiazide.
Cleavage of the ester group (primarily in the liver) converts benazepril to its active metabolite, benazeprilat. Peak plasma concentrations of benazeprilat are reached 1 to 2 hours after drug intake in the fasting state and 2 to 4 hours after drug intake in the nonfasting state. The serum protein binding of benazepril is about 96.7% and that of benazeprilat about 95.3%, as measured by equilibrium dialysis; on the basis of in vitro studies, the degree of protein binding should be unaffected by age, hepatic dysfunction, or - over the concentration range of 0.24 to 23.6 mcmol/L - concentration.
In studies of rats given 14C-benazepril, benazepril and its metabolites crossed the blood-brain barrier only to an extremely low extent. Multiple doses of benazepril did not result in accumulation in any tissue except the lung, where, as with other ACE inhibitors in similar studies, there was a slight increase in concentration due to slow elimination in that organ.
Benazepril is almost completely metabolized to benazeprilat, which has much greater ACE inhibitory activity than benazepril, and to the glucuronide conjugates of benazepril and benazeprilat. Only trace amounts of an administered dose of benazepril can be recovered unchanged in the urine; about 20% of the dose is excreted as benazeprilat, 4% as benazepril glucuronide, and 8% as benazeprilat glucuronide.
In patients with hepatic dysfunction due to cirrhosis, levels of benazeprilat are essentially unaltered. Similarly, the pharmacokinetics of benazepril and benazeprilat do not appear to be influenced by age.
The kinetics of benazepril are dose-proportional within the dosage range of 5 mg to 20 mg. Small deviations from dose proportionality were observed when the broader range of 2 mg to 80 mg was studied, possibly due to the saturable binding of the compound to ACE.
The effective half-life of accumulation of benazeprilat following multiple dosing of benazepril is 10 to 11 hours. Thus, steady-state concentrations of benazeprilat should be reached after 2 or 3 doses of benazepril given once daily.
During chronic administration (28 days) of once-daily doses of benazepril between 5 mg and 20 mg, the kinetics did not change, and there was no significant accumulation. Accumulation ratios based on AUC and urinary recovery of benazeprilat were 1.19 and 1.27, respectively.
When dialysis was started 2 hours after ingestion of 10 mg of benazepril, approximately 6% of benazeprilat was removed in 4 hours of dialysis. The parent compound, benazepril, was not detected in the dialysate.
Benazepril and benazeprilat are cleared predominantly by renal excretion in healthy subjects with normal renal function. Nonrenal (i.e., biliary) excretion accounts for approximately 11% to 12% of benazeprilat excretion in healthy subjects. In patients with renal failure, biliary clearance may compensate to an extent for deficient renal clearance.
The disposition of benazepril and benazeprilat in patients with mild-to-moderate renal insufficiency (creatinine clearance >30 mL/min) is similar to that in patients with normal renal function. In patients with creatinine clearance ≤30 mL/min, peak benazeprilat levels and the initial (alpha phase) half-life increase, and time to steady-state may be delayed (see DOSAGE AND ADMINISTRATION).
Following oral administration, plasma hydrochlorothiazide concentrations decline bi-exponentially, with a mean distribution half-life of about 2 hours and an elimination half-life of about 10 hours. About 70% of an orally administered dose of hydrochlorothiazide is eliminated in the urine as unchanged drug. In a study in individuals with impaired renal function, the mean elimination half-life of hydrochlorothiazide was increased to 2 fold in individuals with mild/moderate renal impairment (30 ˂ CLcr ˂90 mL/min) and 3 fold and severe renal impairment (≤30 mL/min), when compared to individuals with normal renal function (CLcr >90 mL/min).
Pharmacodynamics
Benazepril
Single and multiple doses of 10 mg or more of benazeprilcause inhibition of plasma ACE activity by at least 80% to 90% for at least 24 hours after dosing. For up to 4 hours after a 10 mg dose, pressor responses to exogenous angiotensin I were inhibited by 60% to 90%.
In normal human volunteers, single doses of benazepril caused an increase in renal blood flow but had no effect on glomerular filtration rate.
Hydrochlorothiazide
After oral administration of hydrochlorothiazide, diuresis begins within 2 hours, peaks in about 4 hours and lasts about 6 to 12 hours.
Drug Interactions
Benazepril hydrochloride and hydrochlorothiazide potentiates the antihypertensive action of other antihypertensive drugs (e.g., curare derivatives, guanethidine, methyldopa, beta-blockers, vasodilators, calcium channel blockers ACE inhibitors and ARBs and DRIs).
CLINICAL STUDIES
In single-dose studies, benazepril lowered blood pressure within 1 hour, with peak reductions achieved 2 to 4 hours after dosing. The antihypertensive effect of a single dose persisted for 24 hours. In multiple-dose studies, once-daily doses of 20 mg to 80 mg decreased seated pressure (systolic/diastolic) 24 hours after dosing by about 6-12/4-7 mmHg. The reductions at trough are about 50% of those seen at peak.
Four dose-response studies of benazepril monotherapy using once-daily dosing were conducted in 470 mild-to-moderate hypertensive patients not using diuretics. The minimal effective once-daily dose of benazepril was 10 mg; further falls in blood pressure, especially at morning trough, were seen with higher doses in the studied dosing range (10 mg to 80 mg). In studies comparing the same daily dose of benazepril given as a single morning dose or as a twice-daily dose, blood pressure reductions at the time of morning trough blood levels were greater with the divided regimen.
The antihypertensive effects of benazepril were not appreciably different in patients receiving high- or low-sodium diets.
Benazepril-Hydrochlorothiazide
In 15 controlled clinical trials, 1453 healthy or hypertensive patients were exposed to benazepril and hydrochlorothiazide of which 459 were exposed for at least 6 months, 214 for at least 12 months and 25 for at least 24 months.
The combination of benazepril-hydrochlorothiazide resulted in mean placebo-adjusted decreases in sitting systolic and diastolic blood pressures of 10/6 mm Hg with 5/6.25 mg and 10/12.5 mg doses, and 20/10 mmHg with 20/25 mg dose.
In clinical trials of benazepril/hydrochlorothiazide using benazepril doses of 5 to 20 mg and hydrochlorothiazide doses of 6.25 to 25 mg, the antihypertensive effects were sustained for at least 24 hours, and they increased with increasing dose of either component. Although benazepril monotherapy is somewhat less effective in blacks than in nonblacks, the efficacy of combination therapy appears to be independent of race.
INDICATIONS AND USAGE
Benazepril hydrochloride and hydrochlorothiazide tablets are indicated for the treatment of hypertension.
This fixed combination drug is not indicated for the initial therapy of hypertension (see DOSAGE AND ADMINISTRATION).
CONTRAINDICATIONS
Benazepril hydrochloride and hydrochlorothiazide is contraindicated in patients who are anuric.
Benazepril hydrochloride and hydrochlorothiazide is also contraindicated in patients who are hypersensitive to benazepril, to any other ACE inhibitor, to hydrochlorothiazide, or to other sulfonamide-derived drugs. Hypersensitivity reactions are more likely to occur in patients with a history of allergy or bronchial asthma.
Benazepril hydrochloride and hydrochlorothiazide is also contraindicated in patients with a history of angioedema with or without previous ACE inhibitor treatment.
Benazepril hydrochloride and hydrochlorothiazide is contraindicated in combination with a neprilysin inhibitor (e.g., sacubitril). Do not administer benazepril hydrochloride and hydrochlorothiazide within 36 hours of switching to or from sacubitril/valsartan, a neprilysin inhibitor (see WARNINGSand PRECAUTIONS).
Do not coadminister aliskiren with angiotensin receptor blockers, ACE inhibitors, including benazepril hydrochloride and hydrochlorothiazide in patients with diabetes.
WARNINGS
Anaphylactoid and Possibly Related Reactions
Presumably because angiotensin-converting enzyme inhibitors affect the metabolism of eicosanoids and polypeptides, including endogenous bradykinin, patients receiving ACE inhibitors (including benazepril) may be subject to a variety of adverse reactions, some of them serious.
Head and Neck Angioedema
Angioedema of the face, extremities, lips, tongue, glottis, and larynx has been reported in patients treated with angiotensin-converting enzyme inhibitors. In U.S. clinical trials, symptoms consistent with angioedema were seen in none of the subjects who received placebo and in about 0.5% of the subjects who received benazepril. Angioedema associated with laryngeal edema can be fatal. If laryngeal stridor or angioedema of the face, tongue, or glottis occurs, treatment with benazepril hydrochloride and hydrochlorothiazide should be discontinued and appropriate therapy instituted immediately. When involvement of the tongue, glottis, or larynx appears likely to cause airway obstruction, appropriate therapy, e.g., subcutaneous epinephrine injection 1:1000 (0.3 to 0.5 mL) should be promptly administered (see PRECAUTIONSand ADVERSE REACTIONS).
Black patients receiving ACE inhibitors have been reported to have a higher incidence of angioedema compared to nonblacks.
Patients receiving coadministration of ACE inhibitor and mTOR (mammalian target of rapamycin) inhibitor (e.g., temsirolimus, sirolimus, everolimus) therapy or a neprilysin inhibitor may be at increased risk for angioedema (see PRECAUTIONS).
Intestinal Angioedema
Intestinal angioedema has been reported in patients treated with ACE inhibitors. These patients presented with abdominal pain (with or without nausea or vomiting); in some cases there was no prior history of facial angioedema and C-1 esterase levels were normal. The angioedema was diagnosed by procedures including abdominal CT scan or ultrasound, or at surgery, and symptoms resolved after stopping the ACE inhibitor. Intestinal angioedema should be included in the differential diagnosis of patients on ACE inhibitors presenting with abdominal pain.
Anaphylactoid Reactions During Desensitization
Two patients undergoing desensitizing treatment with hymenoptera venom while receiving ACE inhibitors sustained life-threatening anaphylactoid reactions. In the same patients, these reactions were avoided when ACE inhibitors were temporarily withheld, but they reappeared upon inadvertent rechallenge.
Anaphylactoid Reactions During Membrane Exposure
Anaphylactoid reactions have been reported in patients dialyzed with high-flux membranes and treated concomitantly with an ACE inhibitor. Anaphylactoid reactions have also been reported in patients undergoing low-density lipoprotein apheresis with dextran sulfate absorption.
Hypersensitivity reactions to hydrochlorothiazide are more likely in patients with allergy and asthma.
Hypotension
Benazepril hydrochloride and hydrochlorothiazide can cause symptomatic hypotension. Like other ACE inhibitors, benazepril has been only rarely associated with hypotension in uncomplicated hypertensive patients. Symptomatic hypotension is most likely to occur in patients who have been volume and/or salt depleted as a result of prolonged diuretic therapy, dietary salt restriction, dialysis, diarrhea, or vomiting. Volume and/or salt depletion should be corrected before initiating therapy with benazepril hydrochloride and hydrochlorothiazide.
Benazepril hydrochloride and hydrochlorothiazide should be used cautiously in patients receiving concomitant therapy with other antihypertensives. The thiazide component of benazepril hydrochloride and hydrochlorothiazide may potentiate the action of other antihypertensive drugs, especially ganglionic or peripheral adrenergic-blocking drugs. The antihypertensive effects of the thiazide component may also be enhanced in the postsympathectomy patient.
In patients with congestive heart failure, with or without associated renal insufficiency, ACE inhibitor therapy may cause excessive hypotension, which may be associated with oliguria, azotemia, and (rarely) with acute renal failure and death. In such patients, benazepril hydrochloride and hydrochlorothiazide therapy should be started under close medical supervision; they should be followed closely for the first 2 weeks of treatment and whenever the dose of benazepril or diuretic is increased.
If hypotension occurs, the patient should be placed in a supine position, and, if necessary, treated with intravenous infusion of physiological saline. Benazepril hydrochloride and hydrochlorothiazide treatment usually can be continued following restoration of blood pressure and volume.
Impaired Renal Function
Monitor renal function periodically in patients treated with benazepril hydrochloride and hydrochlorothiazide. Changes in renal function including acute renal failure can be caused by drugs that inhibit the renin-angiotensin system and by diuretics. Patients whose renal function may depend in part on the activity of the renin-angiotensin system (e.g., patients with renal artery stenosis, chronic kidney disease, severe congestive heart failure, or volume depletion) may be at particular risk of developing acute renal failure on benazepril hydrochloride and hydrochlorothiazide. Consider withholding or discontinuing therapy in patients who develop a clinically significant decrease in renal function on benazepril hydrochloride and hydrochlorothiazide.
In a small study of hypertensive patients with unilateral or bilateral renal artery stenosis, treatment with benazepril was associated with increases in blood urea nitrogen and serum creatinine; these increases were reversible upon discontinuation of benazepril therapy, concomitant diuretic therapy, or both.
Neutropenia/Agranulocytosis
Another angiotensin-converting enzyme inhibitor, captopril, has been shown to cause agranulocytosis and bone marrow depression, rarely in uncomplicated patients (incidence probably less than once per 10,000 exposures) but more frequently (incidence possibly as great as once per 1000 exposures) in patients with renal impairment, especially those who also have collagen-vascular diseases such as systemic lupus erythematosus or scleroderma.
Available data from clinical trials of benazepril are insufficient to show that benazepril does not cause agranulocytosis at similar rates. Monitoring of white blood cell counts should be considered in patients with collagen-vascular disease, especially if the disease is associated with impaired renal function.
Fetal Toxicity
Pregnancy
Use of drugs that act on the renin-angiotensin system during the second and third trimesters of pregnancy reduces fetal renal function and increases fetal and neonatal morbidity and death. Resulting oligohydramnios can be associated with fetal lung hypoplasia and skeletal deformations. Potential neonatal adverse effects include skull hypoplasia, anuria, hypotension, renal failure, and death. When pregnancy is detected, discontinue benazepril hydrochloride and hydrochlorothiazide as soon as possible. These adverse outcomes are usually associated with use of these drugs in the second and third trimester of pregnancy. Most epidemiologic studies examining fetal abnormalities after exposure to antihypertensive use in the first trimester have not distinguished drugs affecting the renin-angiotensin system from other antihypertensive agents. Appropriate management of maternal hypertension during pregnancy is important to optimize outcomes for both mother and fetus.
In the unusual case that there is no appropriate alternative to therapy with drugs affecting the renin-angiotensin system for a particular patient, apprise the mother of the potential risk to the fetus. Perform serial ultrasound examinations to assess the intra-amniotic environment. If oligohydramnios is observed, discontinue benazepril hydrochloride and hydrochlorothiazide unless it is considered life saving for the mother. Fetal testing may be appropriate, based on the week of pregnancy. Patients and physicians should be aware, however, that oligohydramnios may not appear until after the fetus has sustained irreversible injury. Closely observe infants with histories of in utero exposure to benazepril hydrochloride and hydrochlorothiazide for hypotension, oliguria, and hyperkalemia (see PRECAUTIONS, Pediatric Use).
No teratogenic effects of benazepril hydrochloride were seen in studies of pregnant rats, mice, and rabbits. On a mg/m2 basis, the doses used in these studies were 60 times (in rats), 9 times (in mice), and more than 0.8 times (in rabbits) the maximum recommended human dose (assuming a 50-kg woman). On a mg/kg basis these multiples are 300 times (in rats), 90 times (in mice), and more than 3 times (in rabbits) the maximum recommended human dose. When hydrochlorothiazide was orally administered without benazepril to pregnant mice and rats during their respective periods of major organogenesis, at doses up to 3000 and 1000 mg/kg/day respectively, there was no evidence of harm to the fetus. Similarly, no teratogenic effects of benazepril were seen in studies of pregnant rats, mice, and rabbits; on a mg/kg basis, the doses used in these studies were 300 times (in rats), 90 times (in mice), and more than 3 times (in rabbits) the maximum recommended human dose.
Thiazides can cross the placenta, and concentrations reached in the umbilical vein approach those in the maternal plasma. Hydrochlorothiazide, like other diuretics, can cause placental hypoperfusion. It accumulates in the amniotic fluid, with reported concentrations up to 19 times higher than in umbilical vein plasma. Use of thiazides during pregnancy is associated with a risk of fetal or neonatal jaundice or thrombocytopenia. Since they do not prevent or alter the course of EPH (Edema, Proteinuria, Hypertension) gestosis (pre-eclampsia), these drugs must not be used to treat hypertension in pregnant women. The use of hydrochlorothiazide for other indications (e.g., heart disease) in pregnancy should be avoided.
Hepatic Failure
Rarely, ACE inhibitors have been associated with a syndrome that starts with cholestatic jaundice and progresses to fulminant hepatic necrosis and (sometimes) death. The mechanism of this syndrome is not understood. Patients receiving ACE inhibitors who develop jaundice or marked elevations of hepatic enzymes should discontinue the ACE inhibitor and receive appropriate medical follow-up.
Systemic Lupus Erythematosus
Thiazide diuretics have been reported to cause exacerbation or activation of systemic lupus erythematosus.
Acute Myopia and Secondary Angle-Closure Glaucoma
Hydrochlorothiazide, a sulfonamide, can cause an idiosyncratic reaction, resulting in acute transient myopia and acute angle-closure glaucoma. Symptoms include acute onset of decreased visual acuity or ocular pain and typically occur within hours to weeks of drug initiation. Untreated acute angle-closure glaucoma can lead to permanent vision loss. The primary treatment is to discontinue hydrochlorothiazide as rapidly as possible. Prompt medical or surgical treatments may need to be considered if the intraocular pressure remains uncontrolled. Risk factors for developing acute angle-closure glaucoma may include a history of sulfonamide or penicillin allergy.
PRECAUTIONS
General
Serum Electrolyte Abnormalities
In clinical trials of benazepril hydrochloride and hydrochlorothiazide, the average change in serum potassium was near zero in subjects who received 5/6.25 mg or 20/12.5 mg, but the average subject who received 10/12.5 mg or 20/25 mg experienced a mild reduction in serum potassium, similar to that experienced by the average subject receiving the same dose of hydrochlorothiazide monotherapy.
Hydrochlorothiazide can cause hypokalemia and hyponatremia. Hypomagnesemia can result in hypokalemia which appears difficult to treat despite potassium repletion. Drugs that inhibit the renin-angiotensin system can cause hyperkalemia. Monitor serum electrolytes periodically.
Metabolic Disturbances
Hydrochlorothiazide
Hydrochlorothiazide may alter glucose tolerance and raise serum levels of cholesterol and triglycerides.
Hydrochlorothiazide may raise the serum uric acid level due to reduced clearance of uric acid and may cause or exacerbate hyperuricemia and precipitate gout in susceptible patients.
Thiazides decrease urinary calcium excretion and may cause mild elevation of serum calcium. Avoid using benazepril hydrochloride and hydrochlorothiazide in patients with hypercalcemia.
Cough
Presumably due to the inhibition of the degradation of endogenous bradykinin, persistent nonproductive cough has been reported with all ACE inhibitors, always resolving after discontinuation of therapy. ACE inhibitor-induced cough should be considered in the differential diagnosis of cough.
Surgery/Anesthesia
In patients undergoing surgery or during anesthesia with agents that produce hypotension, benazepril will block the angiotensin II formation that could otherwise occur secondary to compensatory renin release. Hypotension that occurs as a result of this mechanism can be corrected by volume expansion.
Information for Patients
Angioedema
Angioedema, including laryngeal edema, can occur at any time with treatment with ACE inhibitors. A patient receiving benazepril hydrochloride and hydrochlorothiazide should be told to report immediately any signs or symptoms suggesting angioedema (swelling of face, eyes, lips, or tongue, or difficulty in breathing) and to take no more drugs until after consulting with the prescribing physician.
Pregnancy
Female patients of childbearing age should be told about the consequences of exposure to benazepril hydrochloride and hydrochlorothiazide during pregnancy. Discuss treatment options with women planning to become pregnant. Patients should be asked to report pregnancies to their physicians as soon as possible.
Symptomatic Hypotension
A patient receiving benazepril hydrochloride and hydrochlorothiazide should be cautioned that lightheadedness can occur, especially during the first days of therapy, and that it should be reported to the prescribing physician. The patient should be told that if syncope occurs, benazepril hydrochloride and hydrochlorothiazide should be discontinued until the physician has been consulted.
All patients should be cautioned that inadequate fluid intake, excessive perspiration, diarrhea, or vomiting can lead to an excessive fall in blood pressure, with the same consequences of lightheadedness and possible syncope.
Hyperkalemia
A patient receiving benazepril hydrochloride and hydrochlorothiazide should be told not to use potassium supplements or salt substitutes containing potassium without consulting the prescribing physician.
Neutropenia
Patients should be told to promptly report any indication of infection (e.g., sore throat, fever), which could be a sign of neutropenia.
Non-melanoma Skin Cancer
Instruct patients taking hydrochlorothiazide to protect skin from the sun and undergo regular skin cancer screening.
Laboratory Tests
The hydrochlorothiazide component of benazepril hydrochloride and hydrochlorothiazide may decrease serum PBI levels without signs of thyroid disturbance.
Therapy with benazepril hydrochloride and hydrochlorothiazide should be interrupted for a few days before carrying out tests of parathyroid function.
Drug Interactions
Neprilysin Inhibitors
Patients taking concomitant neprilysin inhibitors may be at increased risk for angioedema.
Interactions Common for Both Benazepril and Hydrochlorothiazide
Potassium Supplements and Potassium-Sparing Diuretics
Concomitant use with benazepril hydrochloride and hydrochlorothiazide may effect potassium levels. Monitor potassium periodically.
mTOR (mammalian target of rapamycin) inhibitors
Patients receiving coadministration of ACE inhibitor and mTOR inhibitor (e.g., temsirolimus, sirolimus, everolimus) therapy may be at increased risk for angioedema (see WARNINGS).
Lithium
Renal clearance of lithium is reduced by thiazides and increase the risk of lithium toxicity. Increased serum lithium levels and symptoms of lithium toxicity have been reported in patients receiving ACE inhibitors during therapy with lithium. Monitor lithium levels when used concomitantly with benazepril hydrochloride and hydrochlorothiazide.
Dual Blockade of the Renin-Angiotensin System (RAS)
Dual Blockade of the RAS with angiotensin receptor blockers, ACE inhibitors, or aliskiren is associated with increased risks of hypertension, hyperkalemia, and changes in renal function (including acute renal failure) compared to monotherapy. Most patients receiving the combination of two RAS inhibitors do not obtain any additional benefit compared to monotherapy. In general, avoid combined use of RAS inhibitors. Closely monitor blood pressure, renal function and electrolytes in patients on benazepril hydrochloride and hydrochlorothiazide and other agents that affect the RAS.
Do not coadminister aliskiren with benazepril hydrochloride and hydrochlorothiazide in patients with diabetes. Avoid use of aliskiren with benazepril hydrochloride and hydrochlorothiazide in patients with renal impairment (GFR < 60 mL/min).
NSAIDs and Cox-2 Selective Agents
In patients who are elderly, volume-depleted (including those on diuretic therapy), or with compromised renal function, coadministration of NSAIDs, including selective COX-2 inhibitors, with ACE inhibitors, including benazepril, may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. Monitor renal function periodically in patients receiving benazepril and NSAID therapy.
The antihypertensive effect of benazepril and hydrochlorothiazide may be attenuated by NSAIDs.
Benazepril
Benazepril has been used concomitantly with beta-adrenergic-blocking agents, calcium-blocking agents, cimetidine, diuretics, digoxin, hydralazine, and naproxen without evidence of clinically important adverse interactions. Other ACE inhibitors have had less than additive effects with beta-adrenergic blockers, presumably because drugs of both classes lower blood pressure by inhibiting parts of the renin-angiotensin system.
Interaction studies with warfarin and acenocoumarol have failed to identify any clinically important effects of benazepril on the serum concentrations or clinical effects of these anticoagulants.
Gold
Nitritoid reactions (symptoms include facial flushing, nausea, vomiting and hypotension) have been reported rarely in patients on therapy with injectable gold (sodium aurothiomalate) and concomitant ACE inhibitor therapy.
Hydrochlorothiazide
Ion exchange resins
Stagger the dosage of hydrochlorothiazide and ion exchange resins such that hydrochlorothiazide is administered at least 4 hours before or 4 to 6 hours after the administration of resins. Single doses of either cholestyramine or colestipol resins bind the hydrochlorothiazide and reduce its absorption from the gastrointestinal tract by up to 85% and 43%, respectively.
Digitalis Glycosides
Thiazide-induced hypokalemia or hypomagnesemia may predispose the patients to digoxin toxicity.
Skeletal Muscle Relaxants
Possible increased responsiveness to muscle relaxants such as curare derivatives.
Antidiabetic Agents
Dosage adjustment of antidiabetic drug may be required.
Antineoplastic Agents (e.g., cyclophosphamide, methotrexate)
Concomitant use of thiazide diuretics may reduce renal excretion of cytotoxic agents and enhance their myelosuppressive effects.
Drugs that Alter Gastrointestinal Motility
The bioavailability of thiazide-type diuretics may be increased by anticholinergic agents (e.g., atropine, biperiden), apparently due to a decrease in gastrointestinal motility and the stomach emptying rate. Conversely, pro-kinetic drugs may decrease the bioavailability of thiazide diuretics.
Cyclosporin
Concomitant treatment with diuretics may increase the risk of hyperuricaemia and gout-type complications.
Alcohol, Barbiturates or Narcotics
Concomitant administration of thiazide diuretics with alcohol, barbiturates, or narcotics may potentiate orthostatic hypotension.
Pressor Amines
Hydrochlorothiazide may reduce the response to pressor amines such as noradrenaline but the clinical significance of this effect is not sufficient to preclude their use.
Non-Clinical Safety Data
Carcinogenesis, Mutagenicity, Fertility
No evidence of carcinogenicity was found when benazepril was given to rats and mice for 104 weeks at doses up to 150 mg/kg/day. On a body-surface-area basis, this dose is 18 times (rats) and 9 times (mice) the maximum recommended human dose. No mutagenic activity was detected in the Ames test in bacteria (with or without metabolic activation), in an in vitro test for forward mutations in cultured mammalian cells, or in a nucleus anomaly test. At doses of 50 to 500 mg/kg/day (6 to 61 times the maximum recommended dose on a body-surface-area basis), benazepril had no adverse effect on the reproductive performance of male and female rats.
Under the auspices of the National Toxicology Program, rats and mice received hydrochlorothiazide in their feed for 2 years, at doses up to 600 mg/kg/day in mice and up to 100 mg/kg/day in rats. These studies uncovered no evidence of a carcinogenic potential of hydrochlorothiazide in rats or female mice, but there was equivocal evidence of hepatocarcinogenicity in male mice.
Hydrochlorothiazide was not genotoxic in in vitro assays using strains TA 98, TA 100, TA 1535, TA 1537, and TA 1538 of Salmonella typhimurium (the Ames test); in the Chinese Hamster Ovary (CHO) test for chromosomal aberrations; or in in vivo assays using mouse germinal cell chromosomes, Chinese hamster bone marrow chromosomes, and the Drosophila sex-linked recessive lethal trait gene. Positive test results were obtained in the in vitro CHO Sister Chromatid Exchange (clastogenicity) test and in the Mouse Lymphoma Cell (mutagenicity) assays, using concentrations of hydrochlorothiazide of 43 to 1300 mcg/mL. Positive test results were also obtained in the Aspergillus nidulans nondisjunction assay, using an unspecified concentration of hydrochlorothiazide.
Fertility
There are no human fertility data for hydrochlorothiazide. In animal studies, benazepril and hydrochlorothiazide alone or in combination had no effect on fertility and conception (see PRECAUTIONS, Non-Clinical Safety Data).
Use in Specific Populations
NursingMothers
Minimal amounts of unchanged benazepril and benazeprilat are excreted into the breast milk of lactating women treated with benazepril, so that a newborn child ingesting nothing but breast milk would receive less than 0.1% of the maternal doses of benazepril and benazeprilat. Thiazides, on the other hand, are definitely excreted into breast milk. Because of the potential for serious adverse reactions in nursing infants from hydrochlorothiazide and the unknown effects of benazepril in infants, a decision should be made whether to discontinue nursing or to discontinue benazepril hydrochloride and hydrochlorothiazide, taking into account the importance of the drug to the mother.
Geriatric Use
Of the total number of patients who received benazepril hydrochloride and hydrochlorothiazide in U.S. clinical studies of benazepril hydrochloride and hydrochlorothiazide, 19% were 65 or older while about 1.5% were 75 or older. Overall differences in effectiveness or safety were not observed between these patients and younger patients.
A limited amount of data suggests that the systemic clearance of hydrochlorothiazide is reduced in both healthy and hypertensive elderly subjects compared to young healthy volunteers.
Pediatric Use
Neonates with a history of in utero exposure to benazepril hydrochloride and hydrochlorothiazide:
If oliguria or hypotension occurs, direct attention toward support of blood pressure and renal perfusion. Exchange transfusions or dialysis may be required as a means of reversing hypotension and/or substituting for disordered renal function. Benazepril, which crosses the placenta, can theoretically be removed from the neonatal circulation by these means; there are occasional reports of benefit from these maneuvers with another ACE inhibitor, but experience is limited.
Safety and effectiveness in pediatric patients have not been established.
Renal Impairment
Safety and effectiveness of benazepril hydrochloride and hydrochlorothiazide in patients with severe renal impairment (CrCL ≤30 mL/min) have not been established. No dose adjustment is required in patients with mild (CrCL 60 to 90 mL/min) or moderate (CrCL 30 to 60 mL/min) renal impairment.
Hepatic Impairment
No adjustment of the initial dose is required for patients with mild to moderate hepatic impairment (see CLINICAL PHARMACOLOGY).
Hydrochlorothiazide
Minor alterations of fluid and electrolyte imbalance may precipitate hepatic coma in patients with impaired hepatic function or progressive liver disease.
ADVERSE REACTIONS
Benazepril hydrochloride and hydrochlorothiazide has been evaluated for safety in over 2500 patients with hypertension; over 500 of these patients were treated for at least 6 months, and over 200 were treated for more than 1 year.
The reported side effects were generally mild and transient, and there was no relationship between side effects and age, sex, race, or duration of therapy. Discontinuation of therapy due to side effects was required in approximately 7% of U.S. patients treated with benazepril hydrochloride and hydrochlorothiazide and in 4% of patients treated with placebo.
The most common reasons for discontinuation of therapy with benazepril hydrochloride and hydrochlorothiazide in U.S. studies were cough (1.0%; see PRECAUTIONS), “dizziness” (1.0%), headache (0.6%), and fatigue (0.6%).
The side effects considered possibly or probably related to study drug that occurred in U.S. placebo-controlled trials in more than 1% of patients treated with benazepril hydrochloride and hydrochlorothiazide are shown in the table below.
| Benazepril Hydrochloride and Hydrochlorothiazide N=665 | Placebo
N=235 |
|||
|---|---|---|---|---|
| N | % | N | % | |
|
“Dizziness” |
41 |
6.3 |
8 |
3.4 |
|
Fatigue |
34 |
5.2 |
6 |
2.6 |
|
Postural Dizziness |
23 |
3.5 |
1 |
0.4 |
|
Headache |
20 |
3.1 |
10 |
4.3 |
|
Cough |
14 |
2.1 |
3 |
1.3 |
|
Hypertonia |
10 |
1.5 |
3 |
1.3 |
|
Vertigo |
10 |
1.5 |
2 |
0.9 |
|
Nausea |
9 |
1.4 |
2 |
0.9 |
|
Impotence |
8 |
1.2 |
0 |
0.0 |
|
Somnolence |
8 |
1.2 |
1 |
0.4 |
Other side effects considered possibly or probably related to study drug that occurred in U.S. placebo-controlled trials in 0.3% to 1.0% of patients treated with benazepril hydrochloride and hydrochlorothiazide were the following:
Cardiovascular
Palpitations, flushing.
Gastrointestinal
Vomiting, diarrhea, dyspepsia, anorexia, and constipation.
Neurologic and Psychiatric
Insomnia, nervousness, paresthesia, libido decrease, dry mouth, taste perversion, and tinnitus.
Dermatologic
Rash and sweating.
Other
Urinary frequency, arthralgia, myalgia, asthenia, and pain (including chest pain and abdominal pain).
Other adverse experiences reported in 0.3% or more of benazepril hydrochloride and hydrochlorothiazide patients in U.S. controlled clinical trials, and rarer events seen in post-marketing experience, were the following; asterisked entries occurred in more than 1% of patients (in some, a causal relationship to benazepril hydrochloride and hydrochlorothiazide is uncertain):
Cardiovascular
Syncope, peripheral vascular disorder, and tachycardia.
Body as a Whole
Infection, back pain*, flu syndrome*, fever, chills, and neck pain.
Dermatologic
Photosensitivity and pruritus.
Gastrointestinal
Gastroenteritis, flatulence, and tooth disorder.
Neurologic and Psychiatric
Hypesthesia, abnormal vision, abnormal dreams, and retinal disorder.
Respiratory
Upper respiratory infection*, epistaxis, bronchitis, rhinitis*, sinusitis*, and voice alteration.
Other
Conjunctivitis, arthritis, urinary tract infection, alopecia, and urinary frequency*.
Post-Marketing Experience
The following adverse reactions have been identified during post-approval use of either benazepril or hydrochlorothiazide. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate their frequency or establish a causal relationship to drug exposure:
Non-Melanoma Skin Cancer
Hydrochlorothiazide is associated with an increased risk of non-melanoma skin cancer. In a study conducted in the Sentinel System, increased risk was predominantly for squamous cell carcinoma (SCC) and in white patients taking large cumulative doses. The increased risk for SCC in the overall population was approximately 1 additional case per 16,000 patients per year, and for white patients taking a cumulative dose of ≥50,000mg the risk increase was approximately 1 additional SCC case for every 6,700 patients per year.
Benazepril
Stevens-Johnson syndrome, pancreatitis, hemolytic anemia, pemphigus, and thrombocytopenia, eosinophilic pneumonitis.
Hydrochlorothiazide
Digestive
Pancreatitis, small bowel angioedema, jaundice (intrahepatic cholestatic), sialadenitis, vomiting, diarrhea, cramping, nausea, gastric irritation, constipation, and anorexia.
Neurologic
Vertigo, lightheadedness, transient blurred vision, headache, paresthesia, xanthopsia, weakness, and restlessness.
Musculoskeletal
Muscle spasm.
Hematologic
Aplastic anemia, agranulocytosis, leukopenia, neutropenia and thrombocytopenia.
Metabolic
Hyperglycemia, glycosuria, and hyperuricemia, pyrexia, asthenia, parathyroid gland changes with hypercalcemia and hypophosphatemia.
Hypersensitivity
Anaphylactoid reactions, necrotizing angiitis, respiratory distress (including pneumonitis and pulmonary edema), purpura, urticaria, rash, and photosensitivity.
Skin
Erythema multiforme including Stevens-Johnson syndrome, and exfoliative dermatitis including toxic epidermal necrolysis.
Clinical Laboratory Test Findings
Serum Electrolytes
See PRECAUTIONS.
Creatinine and BUN
Minor reversible increases in serum creatinine and BUN were observed in patients with essential hypertension treated with benazepril hydrochloride and hydrochlorothiazide. Such increases occurred most frequently in patients with renal artery stenosis (see PRECAUTIONS).
To report SUSPECTED ADVERSE REACTIONS, contact Upsher-Smith Laboratories, Inc. at 1-855-899-9180 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
OVERDOSAGE
No specific information is available on the treatment of overdosage with benazepril hydrochloride and hydrochlorothiazide; treatment should be symptomatic and supportive. Therapy with benazepril hydrochloride and hydrochlorothiazide should be discontinued, and the patient should be observed. Dehydration, electrolyte imbalance, and hypotension should be treated by established procedures.
Single oral doses of 1 g/kg of benazepril caused reduced activity in mice, and doses of 3 g/kg were associated with significant lethality. Reduction of activity in rats was not seen until they had received doses of 5 g/kg, and doses of 6 g/kg were not lethal. In single-dose studies of hydrochlorothiazide, most rats survived doses up to 2.75 g/kg.
Data from human overdoses of benazepril are scanty, but the most common manifestation of human benazepril overdosage is likely to be hypotension. In human hydrochlorothiazide overdose, the most common signs and symptoms observed have been those of dehydration and electrolyte depletion (hypokalemia, hypochloremia, hyponatremia). If digitalis has also been administered, hypokalemia may accentuate cardiac arrhythmias.
Laboratory determinations of serum levels of benazepril and its metabolites are not widely available, and such determinations have, in any event, no established role in the management of benazepril overdose.
No data are available to suggest physiological maneuvers (e.g., maneuvers to change the pH of the urine) that might accelerate elimination of benazepril and its metabolites. Benazeprilat is only slightly dialyzable, but dialysis might be considered in overdosed patients with severely impaired renal function (see WARNINGS).
Angiotensin II could presumably serve as a specific antagonist-antidote in the setting of benazepril overdose, but angiotensin II is essentially unavailable outside of scattered research facilities. Because the hypotensive effect of benazepril is achieved through vasodilation and effective hypovolemia, it is reasonable to treat benazepril overdose by infusion of normal saline solution.
DOSAGE AND ADMINISTRATION
Dose once daily. The dosage may then be increased after 2 to 3 weeks as needed to help achieve blood pressure goals. The maximum recommended dose is 20/25 mg.
Switch Therapy
A patient whose blood pressure is not adequately controlled with benazepril alone or with hydrochlorothiazide alone may be switched to combination therapy with benazepril hydrochloride and hydrochlorothiazide tablets. The usual recommended starting dose is 10 mg/12.5 mg once daily to control blood pressure.
HOW SUPPLIED
Benazepril Hydrochloride and Hydrochlorothiazide Tablets, for oral administration, are available as
10 mg/12.5 mg
Pink, oblong, film-coated tablets, debossed "E 204" on one side and scored on the other side and supplied as:
NDC 0832-0483-11 bottles of 100
20 mg/12.5 mg
Lavender, oblong, film-coated tablets, debossed "E 211" on one side and scored on the other side and supplied as:
NDC 0832-0484-11 bottles of 100
20 mg/25 mg
Maroon, oblong, film-coated tablets, debossed "E 277" on one side and scored on the other side and supplied as:
NDC 0832-0485-11 bottles of 100
Each strength is supplied in bottles that contain a desiccant.
Dispense in a tight, light-resistant container as defined in the USP with a child-resistant closure, as required.
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Protect from light and moisture. Keep tightly closed.
KEEP OUT OF THE REACH OF CHILDREN.
Manufactured for
UPSHER-SMITH LABORATORIES, LLC.
Maple Grove, MN 55369
© 2020 Upsher-Smith Laboratories, LLC.
Rev. October 2020
MF0483REV10/20
46193071
Package/Label Display Panel
NDC 0832-0483-11
Benazepril Hydrochloride and Hydrochlorothiazide Tablets
10 mg/12.5 mg
100 Tablets Rx only
UPSHER-SMITH
| BENAZEPRIL HYDROCHLORIDE AND HYDROCHLOROTHIAZIDE
benazepril hydrochloride and hydrochlorothiazide tablet, film coated |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| BENAZEPRIL HYDROCHLORIDE AND HYDROCHLOROTHIAZIDE
benazepril hydrochloride and hydrochlorothiazide tablet, film coated |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| BENAZEPRIL HYDROCHLORIDE AND HYDROCHLOROTHIAZIDE
benazepril hydrochloride and hydrochlorothiazide tablet, film coated |
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| Labeler - Upsher-Smith Laboratories, LLC (809088862) |