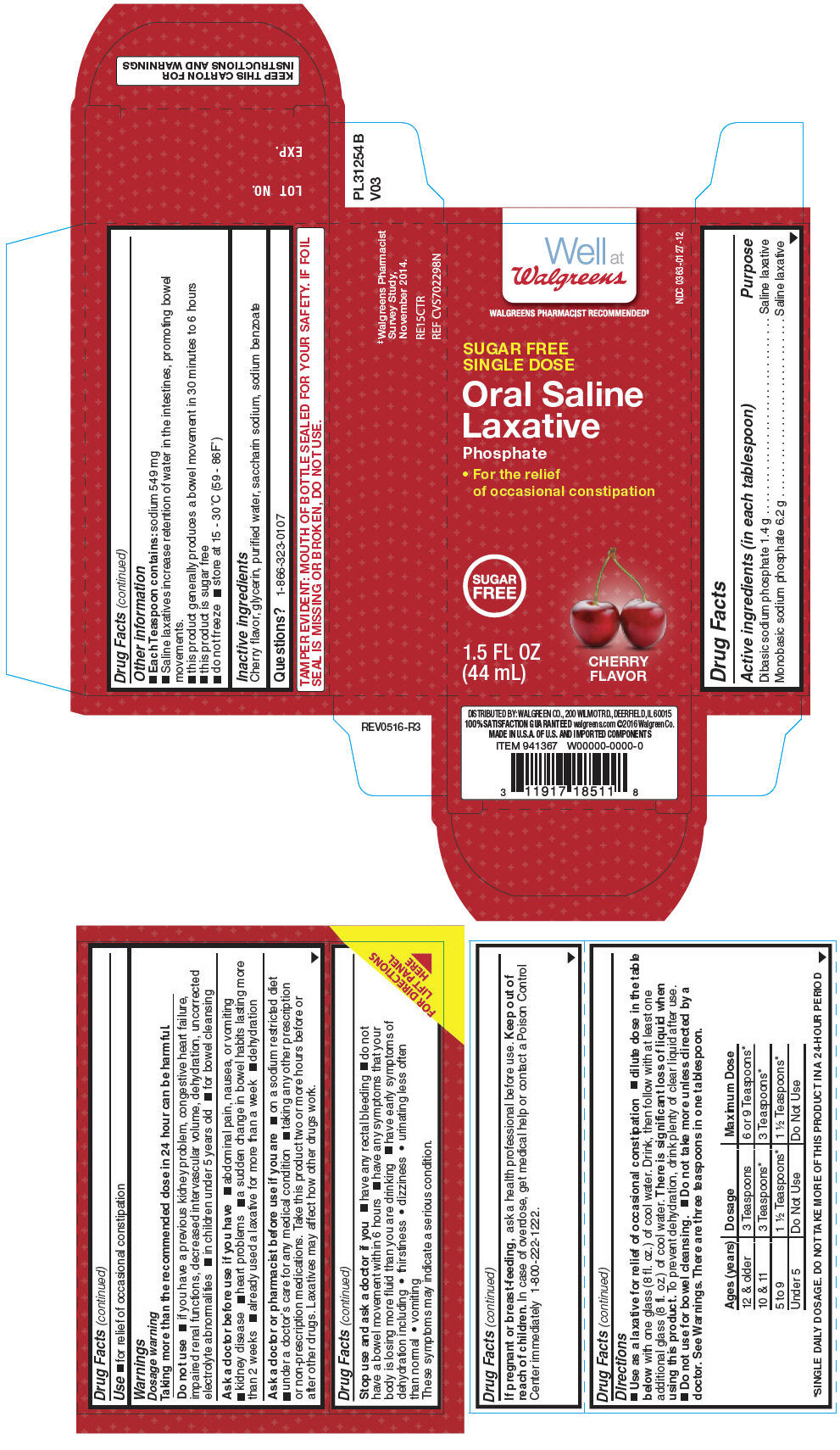
Label: ORAL SALINE LAXATIVE- dibasic sodium phosphate, monobasic sodium phosphate liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 0363-0127-12 - Packager: Walgreen Co
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 3, 2017
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- Use
-
Warnings
Do not use
- if you have a previous kidney problem, congestive heart failure, impaired renal functions, decreased intervascular volume, dehydration, uncorrected electrolyte abnormalities
- in children under 5 years old
- for bowel cleansing
Ask a doctor before use if you have
- abdominal pain, nausea, or vomiting
- kidney disease
- heart problems
- a sudden change in bowel habits lasting more than 2 weeks
- already used a laxative for more than a week
- dehydration
Ask a doctor or pharmacist before use if you are
- on a sodium restricted diet
- under a doctor's care for any medical condition
- taking any other prescription or non-prescription medications. Take this product two or more hours before or after other drugs. Laxatives may affect how other drugs work.
Stop use and ask a doctor if you
- have any rectal bleeding
- do not have a bowel movement within 6 hours
- have any symptoms that your body is losing more fluid than you are drinking
- have early symptoms of dehydration including
- thirstiness
- dizziness
- urinating less often than normal
- vomiting
These symptoms may indicate a serious condition.
-
Directions
- Use as a laxative for relief of occasional constipation
- dilute dose in the table below with one glass (8 fl. oz.) of cool water. Drink, then follow with at least one additional glass (8 fl. oz.) of cool water. There is significant loss of liquid when using this product. To prevent dehydration, drink plenty of clear liquid after use.
- Do not use for bowel cleansing.
- Do not take more unless directed by a doctor. See Warnings. There are three teaspoons in one tablespoon.
- Other information
- Inactive ingredients
- Questions?
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 44 mL Bottle Carton
-
INGREDIENTS AND APPEARANCE
ORAL SALINE LAXATIVE
dibasic sodium phosphate, monobasic sodium phosphate liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0363-0127 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM PHOSPHATE, DIBASIC (UNII: GR686LBA74) (PHOSPHATE ION - UNII:NK08V8K8HR) SODIUM PHOSPHATE, DIBASIC 1.4 g in 15 mL SODIUM PHOSPHATE, MONOBASIC, UNSPECIFIED FORM (UNII: 3980JIH2SW) (PHOSPHATE ION - UNII:NK08V8K8HR) SODIUM PHOSPHATE, MONOBASIC, UNSPECIFIED FORM 6.2 g in 15 mL Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) SACCHARIN SODIUM (UNII: SB8ZUX40TY) SODIUM BENZOATE (UNII: OJ245FE5EU) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0363-0127-12 1 in 1 CARTON 10/05/2012 1 45 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part334 10/05/2012 Labeler - Walgreen Co (008965063) Establishment Name Address ID/FEI Business Operations Natureplex LLC 062808196 MANUFACTURE(0363-0127)