HEALTH MART LOPERAMIDE HYDROCHLORIDE- loperamide hydrochloride suspension
Strategic Sourcing Services LLC
----------
McKesson Loperamide Hydrochloride Oral Suspension Drug Facts
Warnings
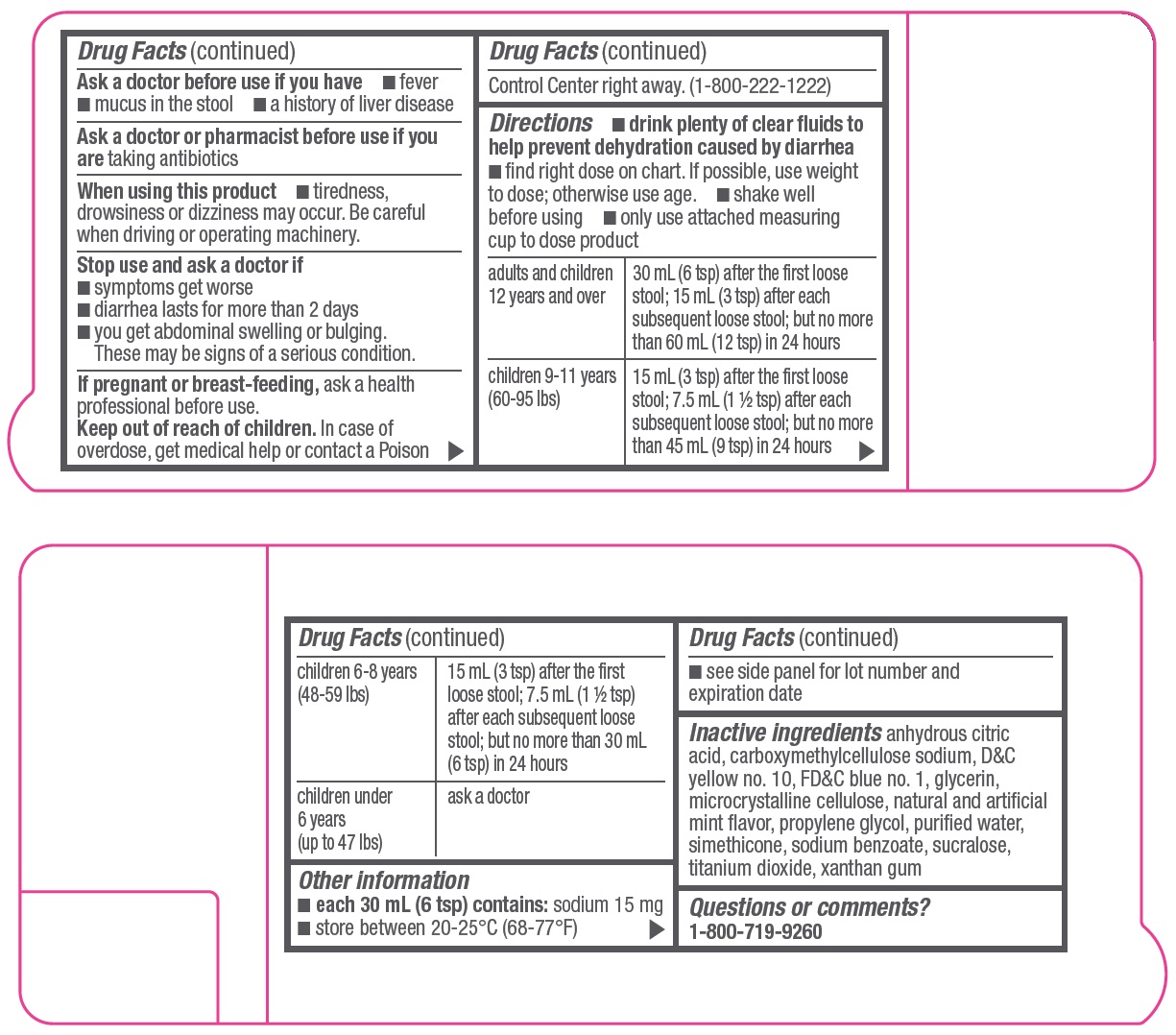
Allergy alert: Do not use if you have ever had a rash or other allergic reaction to loperamide HCl
When using this product
tiredness, drowsiness or dizziness may occur. Be careful when driving or operating machinery.
Directions
- •
- drink plenty of clear fluids to help prevent dehydration caused by diarrhea
- •
- find right dose on chart. If possible, use weight to dose; otherwise use age.
- •
- shake well before using
- •
- only use attached measuring cup to dose product
|
adults and children 12 years and over |
30 mL (6 tsp) after the first loose stool; 15 mL (3 tsp) after each subsequent loose stool; but no more than 60 mL (12 tsp) in 24 hours |
|
children 9-11 years (60-95 lbs) |
15 mL (3 tsp) after the first loose stool; 7.5 mL (1 ½ tsp) after each subsequent loose stool; but no more than 45 mL (9 tsp) in 24 hours |
|
children 6-8 years (48-59 lbs) |
15 mL (3 tsp) after the first loose stool; 7.5 mL (1 ½ tsp) after each subsequent loose stool; but no more than 30 mL (6 tsp) in 24 hours |
|
children under 6 years (up to 47 lbs) |
ask a doctor |
Other information
- •
- each 30 mL (6 tsp) contains: sodium 15 mg
- •
- store between 20-25°C (68-77°F)
- •
- see side panel for lot number and expiration date
| HEALTH MART LOPERAMIDE HYDROCHLORIDE
loperamide hydrochloride suspension |
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| Labeler - Strategic Sourcing Services LLC (116956644) |