Label: NEOMYCIN POLYMYXIN B SULFATES AND DEXAMETHASONE- neomycin sulfate, polymyxin b sulfate and dexamethasone suspension/ drops
-
Contains inactivated NDC Code(s)
NDC Code(s): 57319-347-01 - Packager: Phoenix Pharmaceutical, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
Drug Label Information
Updated November 3, 2010
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION:
Neomycin and Polymyxin B Sulfates and Dexamethasone Ophthalmic Suspension USP is a multiple dose anti-infective steroid combination in a sterile suspension for topical application. The active ingredient, Dexamethasone, is represented by the following structural formula:
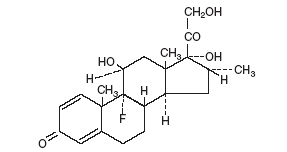
C22H29FO5
Mol. Wt. 392.47
Chemical Name: Pregna-1,4-diene-3,20-dione, 9-fluoro-11,17,21-trihydroxy-16-methyl-, (11β,16α)-.
Neomycin Sulfate is the sulfate salt of neomycin B and C which are produced by the growth of Streptomyces fradiae Waksman (Fam. Streptomycetaceae). It has a potency equivalent to not less than 600 micrograms of neomycin base per milligram, calculated on an anhydrous basis. The structural formula is:
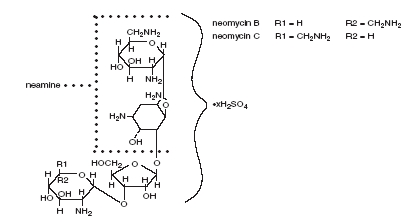
Polymyxin B Sulfate is the sulfate salt of polymyxin B1 and B2 which are produced by the growth of Bacillus polymyxa (Prazmowski) Migula (Fam. Bacillaceae). It has a potency of not less than 6,000 polymyxin B units per milligram, calculated on an anhydrous basis. The structural formula is:
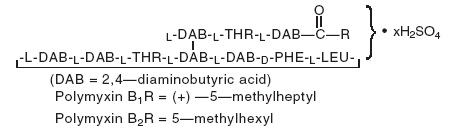
Each mL Contains: ACTIVES: Neomycin Sulfate (equivalent to 3.5 mg Neomycin), Polymyxin B Sulfate equal to 10,000 polymyxin B units, Dexamethasone 1 mg (0.1%); INACTIVES: Sodium Chloride, Hypromellose, Polysorbate 20, Purified Water. Hydrochloric Acid and/or Sodium Hydroxide may be added to adjust pH (3.5 – 6.0).
PRESERVATIVE ADDED: Benzalkonium Chloride 0.004% -
CLINICAL PHARMACOLOGY:
Corticoids suppress the inflammatory response to a variety of agents and they probably delay or slow the healing. Since corticoids may inhibit the body’s defense mechanism against infection, a concomitant antimicrobial drug may be used when this inhibition is considered to be clinically significant in a particular case.
When a decision to administer both a corticoid and an antimicrobial is made, the administration of such drugs in combination has the advantage of greater patient compliance and convenience, with the added assurance that the appropriate dosage of both drugs is administered, plus assured compatibility of ingredients when both types of drugs are in the same formulation and, particularly, that the correct volume of drug is delivered and retained.
The relative potency of corticosteroids depends on the molecular structure, concentration, and release from the vehicle.
-
INDICATIONS AND USAGE:
For steroid-responsive inflammatory ocular conditions for which a corticosteroid is indicated and where bacterial infection or a risk of bacterial ocular infection exists.
Ocular steroids are indicated in inflammatory conditions of the palpebral and bulbar conjunctiva, cornea, and anterior segment of the globe where the inherent risk of steroid use in certain infective conjunctivitis is accepted to obtain a diminution in edema and inflammation. They are also indicated in chronic anterior uveitis and corneal injury from chemical, radiation or thermal burns, or penetration of foreign bodies.
The use of a combination drug with an anti-infective component is indicated where the risk of infection is high or where there is an expectation that potentially dangerous numbers of bacteria will be present in the eye.
The particular anti-infective drug in this product is active against the following common bacterial eye pathogens: Staphylococcus aureus,Escherichia coli, Haemophilus influenzae, Klebsiella/Enterobacter species, Neisseria species, Pseudomonas aeruginosa.
This product does not provide adequate coverage against Serratia marcescens, and Streptococci, including Streptococcus pneumoniae.
-
CONTRAINDICATIONS:
Neomycin and polymyxin B sulfates and dexamethasone ophthalmic suspension is contraindicated in most viral diseases of the cornea and conjunctiva, including epithelial herpes simplex keratitis (dendritic keratitis), vaccinia, varicella, and also in mycobacterial infection of the eye and fungal diseases of ocular structures. Neomycin and polymyxin B sulfates and dexamethasone ophthalmic suspension is also contraindicated in individuals with known or suspected hypersensitivity to any of the ingredients of this preparation and to other corticosteroids.
-
WARNINGS:
Prolonged use of corticosteroids may result in glaucoma with damage to the optic nerve, defects in visual acuity and fields of vision, and in posterior subcapsular cataract formation. Prolonged use may also suppress the host immune response and thus increase the hazard of secondary ocular infections.
Various ocular diseases and long-term use of topical corticosteroids have been known to cause corneal and scleral thinning. Use of topical corticosteroids in the presence of thin corneal or scleral tissue may lead to perforation.
Acute purulent infections of the eye may be masked or activity enhanced by the presence of corticosteroid medication.
If this product is used for 10 days or longer, intraocular pressure should be routinely monitored even though it may be difficult in children and uncooperative patients. Steroids should be used with caution in the presence of glaucoma. Intraocular pressure should be checked frequently.
The use of steroids after cataract surgery may delay healing and increase the incidence of bleb formation.
Use of ocular steroids may prolong the course and may exacerbate the severity of many viral infections of the eye (including herpes simplex). Employment of a corticosteroid medication in the treatment of patients with a history of herpes simplex requires great caution; frequent slit lamp microscopy is recommended.
Neomycin and polymyxin B sulfates and dexamethasone ophthalmic suspension is not for injection. It should never be injected subconjunctivally, nor should it be directly introduced into the anterior chamber of the eye.
Products containing neomycin sulfate may cause cutaneous sensitization.
-
PRECAUTIONS:
General
The initial prescription and renewal of the medication order beyond 20 mL should be made by a physician only after examination of the patient with the aid of magnification, such as a slit lamp biomicroscopy and, where appropriate, fluorescein staining. If signs and symptoms fail to improve after two days, the patient should be re-evaluated.
As fungal infections of the cornea are particularly prone to develop coincidentally with long-term corticosteroid applications, fungal invasion should be suspected in any persistent corneal ulceration where a corticosteroid has been used or is in use. Fungal cultures should be taken when appropriate.
If this product is used for 10 days or longer, intraocular pressure should be monitored (see WARNINGS).
Prolonged use of topical anti-bacterial agents may give rise to overgrowth of nonsusceptible organisms including fungi.
Information for Patients:
If inflammation or pain persists longer than 48 hours or becomes aggravated, the patient should be advised to discontinue use of the medication and consult a physician.
This product is sterile when packaged. To prevent contamination, care should be taken to avoid touching the bottle tip to eyelids or to any other surface. The use of this bottle by more than one person may spread infection. Keep bottle tightly closed when not in use. Keep out of reach of children.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals to evaluate carcinogenic or mutagenic potential have not been conducted with polymyxin B sulfate. Treatment of cultured human lymphocytes in vitro with neomycin increased the frequency of chromosome aberrations at the highest concentration (80 µg/mL) tested. However, the effects of neomycin on carcinogenesis and mutagenesis in humans are unknown.
Polymyxin B has been reported to impair the motility of equine sperm, but its effects on male or female fertility are unknown.
Pregnancy
Teratogenic Effects: Pregnancy Category C.
Dexamethasone has been shown to be teratogenic in mice and rabbits following topical ophthalmic application in multiples of the therapeutic dose.
In the mouse, corticosteroids produce fetal resorptions and a specific abnormality, cleft palate. In the rabbit, corticosteroids have produced fetal resorptions and multiple abnormalities involving the head, ears, limbs, palate, etc.
There are no adequate or well-controlled studies in pregnant women. Neomycin and polymyxin B sulfates and dexamethasone ophthalmic suspension should be used during pregnancy only if the potential benefit to the mother justifies the potential risk to the embryo or fetus. Infants born of mothers who have received substantial doses of corticosteroids during pregnancy should be observed carefully for signs of hypoadrenalism.
Nursing Mothers
Systemically administered corticosteroids appear in human milk and could suppress growth, interfere with endogenous corticosteroid production, or cause other untoward effects. It is not known whether topical administration of corticosteroids could result in sufficient systemic absorption to produce detectable quantities in human milk. Because many drugs are excreted in human milk, caution should be exercised when neomycin and polymyxin B sulfates and dexamethasone ophthalmic suspension is administered to a nursing woman.
-
ADVERSE REACTIONS:
Adverse reactions have occurred with steroid/anti-infective combination drugs which can be attributed to the steroid component, the antiinfective component, or the combination. Exact incidence figures are not available since no denominator of treated patients is available.
Reactions occurring most often from the presence of the anti-infective ingredients are allergic sensitizations. The reactions due to the steroid component are: elevation of intraocular pressure (IOP) with possible development of glaucoma, and infrequent optic nerve damage; posterior subcapsular cataract formation; and delayed wound healing.
Corticosteroid-containing preparations have also been reported to cause perforation of the globe. Keratitis, conjunctivitis, corneal ulcers, and conjunctival hyperemia have occasionally been reported following use of steroids.
-
DOSAGE AND ADMINISTRATION:
One or two drops topically in the conjunctival sac(s). In severe disease, drops may be used hourly, being tapered to discontinuation as the inflammation subsides. In mild disease, drops may be used up to four to six times daily.
Not more than 20 mL should be prescribed initially and the prescription should not be refilled without further evaluation as outlined in PRECAUTIONS above.
SHAKE WELL BEFORE USING.
- HOW SUPPLIED:
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
NEOMYCIN POLYMYXIN B SULFATES AND DEXAMETHASONE
neomycin sulfate, polymyxin b sulfate and dexamethasone suspension/ dropsProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:57319-347 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Neomycin Sulfate (UNII: 057Y626693) (NEOMYCIN - UNII:I16QD7X297) Neomycin Sulfate 3.5 mg in 1 mL Polymyxin B Sulfate (UNII: 19371312D4) (POLYMYXIN B - UNII:J2VZ07J96K) Polymyxin B Sulfate 10000 [USP'U] in 1 mL Dexamethasone (UNII: 7S5I7G3JQL) (DEXAMETHASONE - UNII:7S5I7G3JQL) Dexamethasone 1 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) Hydrochloric Acid (UNII: QTT17582CB) HYPROMELLOSES (UNII: 3NXW29V3WO) POLYSORBATE 20 (UNII: 7T1F30V5YH) Water (UNII: 059QF0KO0R) Sodium Chloride (UNII: 451W47IQ8X) Sodium Hydroxide (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:57319-347-01 1 in 1 CARTON 1 5 mL in 1 BOTTLE, DROPPER Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA064135 09/13/1995 Labeler - Phoenix Pharmaceutical, Inc. (150711039) Registrant - Bausch & Lomb Incorporated (196603781) Establishment Name Address ID/FEI Business Operations Bausch & Lomb Incorporated 807927397 MANUFACTURE


