M.V.I. ADULT- retinol, ergocalciferol, .alpha.-tocopherol acetate, dl-, phytonadione, ascorbic acid, niacinamide, riboflavin 5-phosphate sodium, thiamine hydrochloride, pyridoxine hydrochloride, dexpanthenol, biotin, folic acid, and cyanocobalamin
Hospira, Inc.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use M.V.I.-Adult™ safely and effectively. See full prescribing information for M.V.I.-Adult™.
M.V.I.-Adult™ (Multi-Vitamin Injection), for intravenous use. Initial U.S. Approval: 2004 INDICATIONS AND USAGEM.V.I.-Adult™ is a combination of vitamins indicated for prevention of vitamin deficiency in adults and children aged 11 years and above receiving parenteral nutrition (1) DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONSWARNINGS AND PRECAUTIONS
ADVERSE REACTIONSAdverse reactions have included anaphylaxis, rash, erythema, pruritus, headache, dizziness, agitation, anxiety, diplopia (6) To report SUSPECTED ADVERSE REACTIONS, contact Hospira, Inc. at 1-800-441-4100, or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. DRUG INTERACTIONSEffect of M.V.I.-Adult on other drugs:
Effects of other drugs on M.V.I.-Adult: USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION. Revised: 1/2015 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
M.V.I.-Adult™ is a combination of vitamins indicated for the prevention of vitamin deficiency in adults and children aged 11 and older receiving parenteral nutrition.
The physician should not await the development of clinical signs of vitamin deficiency before initiating vitamin therapy.
2 DOSAGE AND ADMINISTRATION
2.1 Important Dosage and Administration Instructions
M.V.I.-Adult™ is a combination product that contains the following vitamins: ascorbic acid, vitamin A, vitamin D, thiamine, riboflavin, pyridoxine, niacinamide, dexpanthenol, vitamin E, vitamin K, folic acid, biotin, and vitamin B12.
M.V.I.-Adult is supplied as a single dose or as a pharmacy bulk package for intravenous use intended for administration by intravenous infusion after dilution:
- •
- M.V.I-Adult Single Dose consists of two vials which must be mixed prior to use. The mixed solution will provide a single 10 mL dose which must be diluted prior to intravenous administration [see Dosage and Administration (2.3)].
- •
- M.V.I-Adult Pharmacy Bulk Package consists of two pharmacy bulk vials which must be mixed prior to use. The mixed solution will provide ten 10 mL single doses which must be diluted prior to intravenous administration. Pharmacy bulk package of M.V.I-Adult is intended for dispensing of single doses to multiple patients in a pharmacy admixture program and is restricted to the preparation of admixtures for infusion [see Dosage and Administration (2.3)].
Do not administer M.V.I.-Adult™ as a direct, undiluted intravenous injection as it may cause dizziness, faintness, and tissue irritation.
2.2 Dosage Information
The recommended daily dosage volume is 10 mL. One daily dose (10 mL) is diluted by adding directly to a specified volume of an intravenous fluid [see Dosage and Administration (2.3)].
Patients with multiple vitamin deficiencies or with increased vitamin requirements may need multiple daily dosages as indicated or additional doses of individual vitamins.
2.3 Preparation and Administration Instructions
M.V.I.-Adult supplied as a single dose:
- •
- M.V.I- Adult is to be used only in a suitable work area such as a laminar flow hood (or an equivalent clean air compounding area).
- •
- Transfer the contents of Vial 1 (5 mL of solution) into the contents of Vial 2 (5 mL of solution). The mixed solution (10 mL) will provide a single 10 mL dose.
- •
- Once the closure system has been penetrated, complete withdrawal of vial contents within 4 hours. Mixed solution may be stored for up to 4 hours refrigerated.
- •
- Visually inspect for particulate matter and discoloration prior to intravenous administration.
- •
- Utilizing a suitable sterile automated compounding device or dispensing pin for accuracy, aseptically transfer the 10 mL dose into a plastic or glass bottle containing at least 500 to 1000 mL intravenous parenteral nutrition solution containing dextrose or saline.
- •
- After M.V.I.-Adult™ is diluted in an intravenous infusion, refrigerate the resulting solution unless it is to be used immediately, and use the solution within 24 hours after dilution.
- •
- Minimize exposure to light because some of the vitamins in M.V.I.-Adult, particularly A, D and riboflavin, are light sensitive.
M.V.I.-Adult supplied as a pharmacy bulk package:
- •
- M.V.I- Adult is to be used only in a suitable work area such as a laminar flow hood (or an equivalent clean air compounding area).
- •
- Transfer the contents of Vial 1 (50 mL) into Vial 2 (50 mL). The mixed solution (100 mL) will provide ten 10 mL single doses to patients in a pharmacy admixture program.
- •
- Each bulk vial closure shall be penetrated only one time with a suitable sterile transfer device or dispensing set that allows measured dispensing of the contents.
- •
- Once the closure system has been penetrated, complete dispensing from the pharmacy bulk vial within 4 hours. Mixed solution may be stored for up to 4 hours refrigerated.
- •
- Discard unused portion.
- •
- Visually inspect for particulate matter and discoloration prior to administration.
- •
- Utilizing a suitable sterile automated compounding device or dispensing pin for accuracy, aseptically transfer each 10 mL dose into a plastic or glass bottle containing at least 500 to 1000 mL intravenous parenteral nutrition solution containing dextrose or saline.
- •
- After M.V.I.-Adult™ is diluted in an intravenous infusion, refrigerate the resulting solution unless it is to be used immediately, and use the solution within 24 hours after dilution.
- •
- Minimize exposure to light because some of the vitamins in M.V.I.-Adult, particularly A, D and riboflavin, are light sensitive.
2.4 Monitoring Vitamin Blood Levels
Blood vitamin concentrations should be monitored to ensure maintenance of adequate levels, particularly in patients receiving parenteral multivitamins as the only source of vitamins for long periods of time.
2.5 Drug Incompatibilities
- •
- M.V.I.-Adult™ is not physically compatible with moderately alkaline solutions such as a sodium bicarbonate solution and other alkaline drugs such as acetazolamide sodium, aminophylline, ampicillin sodium, and chlorothiazide sodium.
- •
- Folic acid is unstable in the presence of calcium salts such as calcium gluconate.
- •
- Vitamin A and thiamine in M.V.I.-Adult™ may react with bisulfite solutions such as sodium bisulfite or vitamin K bisulfite. Do not add M.V.I.-Adult™ directly to intravenous fat emulsions.
- •
- Consult appropriate references for listings of physical and chemical compatibility of solutions and drugs with the vitamin infusion. In such circumstances, admixture or Y-site administration with vitamin solutions should be avoided.
3 DOSAGE FORMS AND STRENGTHS
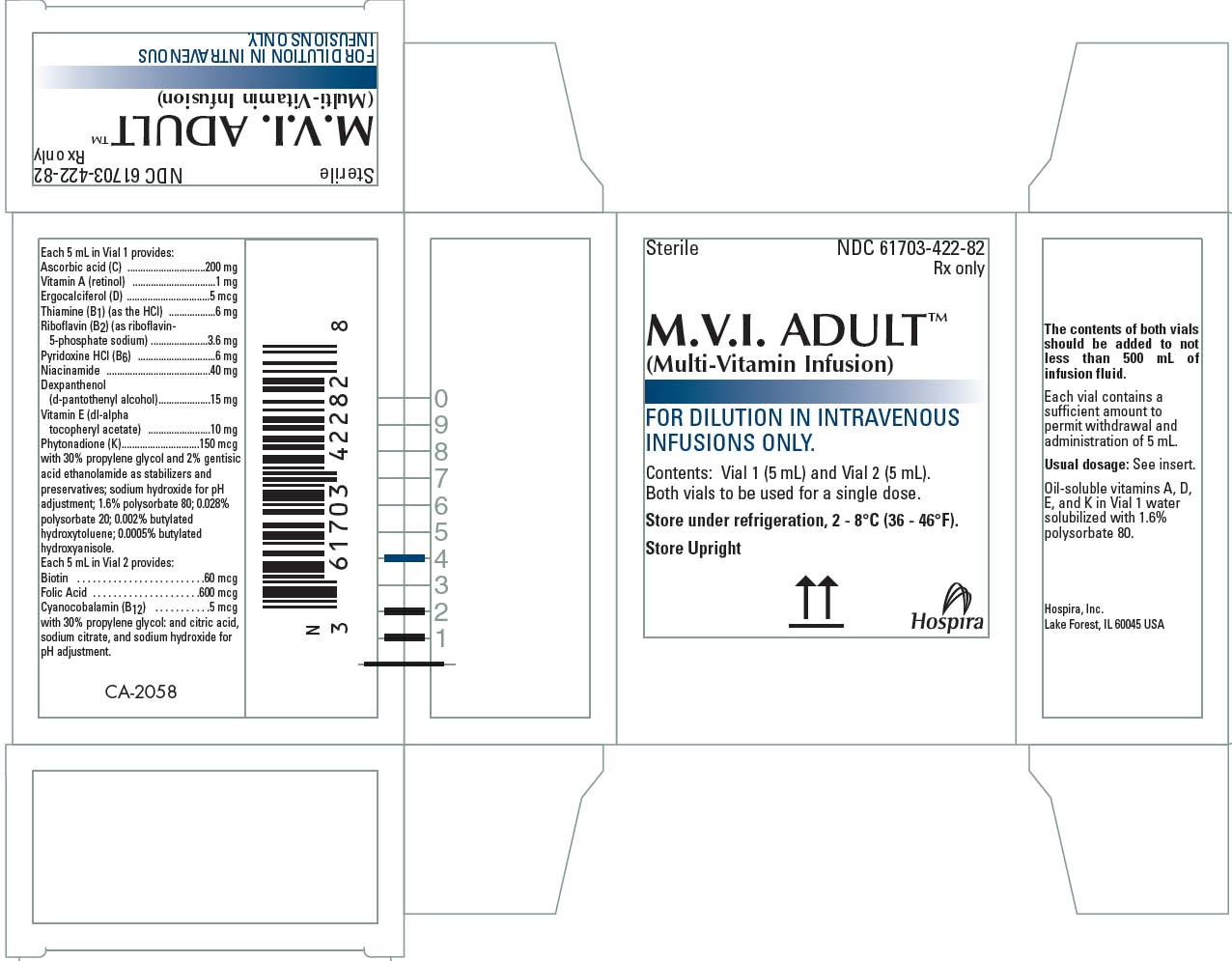
M.V.I.-Adult™ single dose is an injection consisting of two vials labeled Vial 1 and Vial 2. Both vials must be mixed prior to use. The mixed solution (10 mL) will provide a single 10 mL dose [see Dosage and Administration (2.3) and Description (11)].
M.V.I.-Adult™ pharmacy bulk package is an injection consisting of two vials labeled Vial 1 and Vial 2. Both vials must be mixed prior to use. The mixed solution (100 mL) will provide ten 10 mL single doses [see Dosage and Administration (2.3) and Description (11)].
4 CONTRAINDICATIONS
M.V.I.-Adult™ is contraindicated in patients who have:
- •
- A history of hypersensitivity to any of the vitamins or excipients contained in this formulation, or
- •
- An existing hypervitaminosis.
5 WARNINGS AND PRECAUTIONS
5.1 Decreased Anticoagulant Effect of Warfarin
M.V.I.-Adult contains Vitamin K which may decrease the anticoagulant effect of warfarin. In patients who are on warfarin anticoagulant therapy receiving M.V.I-Adult, prothrombin time/INR should be periodically monitored to determine if the dose of warfarin needs to be adjusted.
5.2 Aluminum Toxicity
M.V.I.-Adult™ contains aluminum that may be toxic. Aluminum may reach toxic levels with prolonged parenteral administration in patients with renal impairment. Premature neonates are particularly at risk because their kidneys are immature, and they require large amounts of calcium and phosphate solutions, which contain aluminum.
Patients with impaired kidney function, including premature neonates, who receive parenteral levels of aluminum at greater than 4 to 5 micrograms per kg per day, accumulate aluminum at levels associated with central nervous system and bone toxicity. Tissue loading may occur at even lower rates of administration. To prevent aluminum toxicity monitor periodically aluminum levels with prolonged parenteral administration of M.V.I.-Adult.
5.3 Risk of Low Vitamin A Levels
Vitamin A may adhere to plastic, resulting in lower vitamin A concentrations after administration of M.V.I.-Adult™. Therefore, blood vitamin concentrations should be periodically monitored and the administration of additional therapeutic doses of Vitamin A may be required.
5.4 Allergic Reactions to Thiamine
Allergic reactions such as urticaria, periorbital and digital edema, have been reported following intravenous administration of thiamine, which is found in M.V.I.-Adult™. There have been rare reports of anaphylaxis following intravenous doses of thiamine. No fatal anaphylaxis reactions associated with M.V.I. Adult™ have been reported.
5.5 Hypervitaminosis A
Hypervitaminosis A, manifested by nausea, vomiting, headache, dizziness, blurred vision, has been reported in patients with renal failure receiving 1.5 mg/day retinol and in patients with liver disease. Therefore, supplementation of renal failure patients and patients with liver diseases with vitamin A, an ingredient found in M.V.I.-Adult™, should be undertaken with caution [see Use in Specific Populations (8.6, 8.7)]. Blood levels of Vitamin A should be monitored periodically.
5.6 Interference with Diagnosis of Megaloblastic Anemia
M.V.I-Adult contains folic acid and cyanocobalamin which can mask serum deficits of folic acid and cyanocobalamin in patients with megaloblastic anemia. Avoid the use of M.V.I.-Adult™ in patients with suspected or diagnosed megaloblastic anemia prior to blood sampling for the detection of the folic acid and cyanocobalamin deficiencies.
5.7 Potential to Develop Vitamin Deficiencies or Excesses
In patients receiving parenteral multivitamins, such as with M.V.I.-Adult™, blood vitamin concentrations should be periodically monitored to determine if vitamin deficiencies or excesses are developing. M.V.I.-Adult™ may not correct long-standing specific vitamin deficiencies. The administration of additional doses of specific vitamins may be required [see Dosage and Administration (2.2)].
6 ADVERSE REACTIONS
The following adverse reactions are discussed in greater detail in other section of the labeling.
- •
- Allergic reactions to thiamine [see Warnings and Precautions (5.4)].
- •
- Hypervitaminosis A [see Warnings and Precautions (5.5)].
The following adverse reactions have been identified during post approval use of M.V.I.-Adult™. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Dermatologic: rash, erythema, pruritus
CNS: headache, dizziness, agitation, anxiety
Ophthalmic: diplopia
7 DRUG INTERACTIONS
A number of interactions between vitamins and drugs have been reported. The following are examples of these types of interactions:
7.1 Effect of M.V.I.-Adult on Other Drugs
Phenytoin: Folic acid may increase phenytoin metabolism and lower the serum concentration of phenytoin resulting in increased seizure activity.
Methotrexate: Folic acid may decrease a patient’s response to methotrexate therapy.
Levodopa: Pyridoxine may increase the metabolism of levodopa (decrease blood level of levodopa) and decrease its efficacy.
Antibiotics: Thiamine, riboflavin, pyridoxine, niacinamide, and ascorbic acid decrease antibiotic activities of erythromycin, kanamycin, streptomycin, doxycycline, and lincomycin.
Bleomycin: Ascorbic acid and riboflavin inactivate bleomycin in vitro, thus the activity of bleomycin may be reduced.
7.2 Effect of Other Drugs on M.V.I.-Adult
Hydralazine or Isoniazid:
Concomitant administration of hydralazine or isoniazid may increase pyridoxine requirements.
Chloramphenicol:
In patients with pernicious anemia, the hematologic response to vitamin B12 therapy may be inhibited by concomitant administration of chloramphenicol.
Phenytoin:
Phenytoin may decrease serum folic acid concentrations.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category C
M.V.I.-Adult™ has not been studied in pregnant women. Pregnant women should follow the U.S. recommended daily allowances for pregnancy because their vitamin requirements may exceed those of nonpregnant women. Animal reproduction studies have not been conducted with M.V.I-Adult (Multi-Vitamin Injection) administered by intravenous infusion).
8.3 Nursing Mothers
M.V.I.-Adult™ has not been studied in lactating women. Lactating women may have vitamin requirements that exceed those of nonlactating woman. Caution should be exercised when M.V.I.-Adult™ is administered to a nursing woman.
8.6 Renal Impairment
M.V.I.-Adult™ has not been studied in patients with renal impairment. Monitor renal function, calcium, phosphorus and vitamin A levels in patients with renal impairment [see Warning and Precautions (5.2, 5.5)].
8.7 Hepatic Impairment
M.V.I.-Adult™ has not been studied in patients with hepatic impairment. Monitor vitamin A level in patients with liver disease, high alcohol consumption [see Warning and Precautions (5.5)].
10 OVERDOSAGE
Signs and symptoms of acute or chronic overdosage may be those of individual M.V.I.-Adult™ component toxicity. There is no clinical experience with M.V.I.-Adult™ overdosage.
11 DESCRIPTION
M.V.I.-Adult™ (Multi-Vitamin Injection) is a sterile product consisting of two vials provided as a single dose or as a pharmacy bulk package for intravenous use intended for administration by intravenous infusion after dilution:
M.V.I.-Adult™ supplied as single dose
(a) Vial 1 (5 mL)
(b) Vial 2 (5 mL).
The mixed solution will provide a single 10 mL dose.
M.V.I.-Adult™ supplied as pharmacy bulk package
(a) Vial 1 (50 mL)
(b) Vial 2 (50 mL).
The mixed solution will provide ten single doses of 10 mL each.
Each 5 mL of Vial 1 contains 10 vitamins and each 5 mL of Vial 2 contains 3 vitamins (see Table 1).
|
Vial 1* |
|
|
Fat Soluble Vitamins** |
|
|
Ingredient |
Amount per Unit Dose |
|
Vitamin A (retinol) |
1 mga |
|
Vitamin D (ergocalciferol) |
5 mcgb |
|
Vitamin E (dl-alpha-tocopheryl acetate) |
10 mgc |
|
Vitamin K (phytonadione) |
150 mcg |
|
Water Soluble Vitamins |
|
|
Vitamin C (ascorbic acid) |
200 mg |
|
Niacinamide |
40 mg |
|
Vitamin B2 (as riboflavin 5-phosphate sodium) |
3.6 mg |
|
Vitamin B1 (thiamine) |
6 mg |
|
Vitamin B6 (pyridoxine HCl) |
6 mg |
|
Dexpanthenol (d-pantothenyl alcohol) |
15 mg |
|
* With 30% propylene glycol and 2% gentisic acid ethanolamide as stabilizers and preservatives; sodium hydroxide for pH adjustment; 1.6% polysorbate 80; 0.028% polysorbate 20; 0.002% butylated hydroxytoluene; 0.0005% butylated hydroxyanisole. ** Fat soluble vitamins A, D, E and K are water solubilized with polysorbate 80. (a) 1 mg vitamin A equals 3,300 USP units. |
|
|
Vial 2* |
|
|
Biotin |
60 mcg |
|
Folic acid |
600 mcg |
|
B12 (cyanocobalamin) |
5 mcg |
|
* With 30% propylene glycol; and citric acid, sodium citrate, and sodium hydroxide for pH adjustment. |
|
Multiple vitamin preparation for intravenous infusion:
M.V.I.-Adult™ (Multi-Vitamin Injection) makes available a combination of fat-soluble and water-soluble vitamins in an aqueous solution, formulated for incorporation into intravenous infusions. The liposoluble vitamins A, D, E, and K have been solubilized in an aqueous medium with polysorbate 80, permitting intravenous administration of these vitamins.
16 HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
M.V.I.-ADULT™ is supplied in the following package configurations:
M.V.I.-ADULT™ SINGLE DOSE
NDC 61703-422-82. Boxes of 10 and cartons of 100. Each box contains two vials – Vial 1 (5 mL) and Vial 2 (5 mL), both vials to be used for a single 10 mL dose [see Dosage and Administration (2.3)].
M.V.I.-ADULT™ PHARMACY BULK PACKAGE
NDC 61703-422-83. 2 Boxes of 10 vials (5 Vial 1 and 5 Vial 2), 50 mL each. Mix contents of Vial 1 and Vial 2 to provide ten single 10 mL doses [see Dosage and Administration (2.3)].
Storage and Handling
Minimize the exposure of M.V.I.-Adult™ to light, because vitamins A, D and riboflavin are light sensitive.
Store at 2-8°C (36-46°F).
17 PATIENT COUNSELING INFORMATION
Instruct patients (if age appropriate) and caregivers:
- •
- To watch for signs of allergic reactions (i.e. urticaria, periorbital and digital edema) to thiamine in M.V.I-Adult. Hypersensitivity reactions may occur to any of the vitamins or excipients contained in M.V.I.-Adult.
- •
- To watch for and immediately report signs of hypervitaminosis A, manifested by nausea, vomiting, headache, dizziness, blurred vision, if patients have renal impairment.
- •
- To report other adverse reactions that patients may experience such as rash, erythema, pruritus, headache, dizziness, agitation, anxiety, and diplopia.
- •
- That the patients on warfarin anticoagulant therapy will be monitored periodically for blood prothrombin/ INR levels to determine if the dose of warfarin needs to be adjusted.
- •
- About the significance of periodic monitoring of blood vitamin concentrations to determine if vitamin deficiencies or excesses are developing and the need to monitor renal function, calcium, phosphorus, aluminum, and vitamin A levels in patients with renal impairment.
EN-3764
| M.V.I. ADULT
retinol, ergocalciferol, .alpha.-tocopherol acetate, dl-, phytonadione, ascorbic acid, niacinamide, riboflavin 5-phosphate sodium, thiamine hydrochloride, pyridoxine hydrochloride, dexpanthenol, biotin, folic acid, and cyanocobalamin kit |
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
| Labeler - Hospira, Inc. (141588017) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Hospira, Inc. | 093132819 | ANALYSIS(61703-422) , LABEL(61703-422) , MANUFACTURE(61703-422) , PACK(61703-422) | |