PSEUDOEPHEDRINE HCL- pseudoephedrine hcl tablet, film coated
L.N.K. International, Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Amerisource 44-112 Delisted
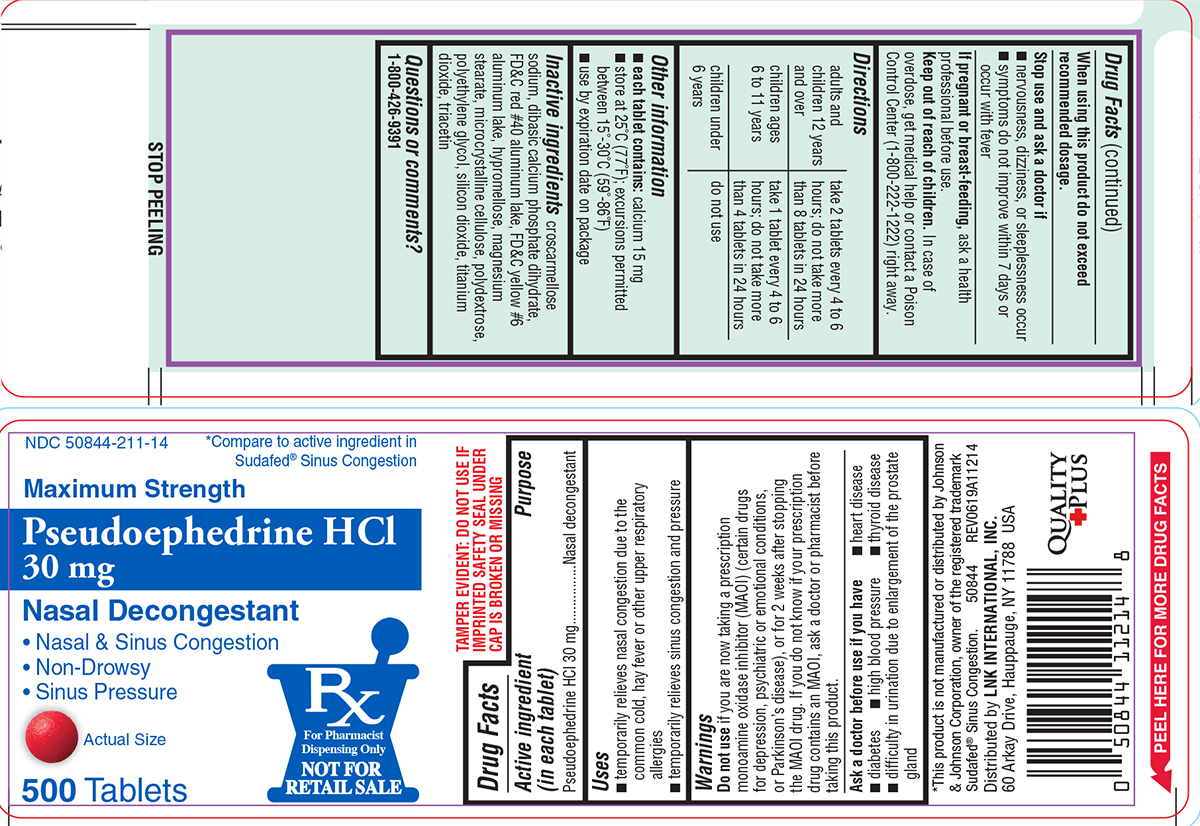
Uses
- temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies
- temporarily relieves sinus congestion and pressure
Warnings
Do not use
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- diabetes
- heart disease
- high blood pressure
- thyroid disease
- difficulty in urination due to enlargement of the prostate gland
Directions
| adults and children 12 years and over | take 2 tablets every 4 to 6 hours; do not take more than 8 tablets in 24 hours |
| children ages 6 to 11 years | take 1 tablet every 4 to 6 hours; do not take more than 4 tablets in 24 hours |
| children under 6 years | do not use |
Other information
-
each tablet contains: calcium 15 mg
- store at 25ºC (77ºF); excursions permitted between 15º-30ºC (59º-86ºF)
- use by expiration date on package
Inactive ingredients
croscarmellose sodium, dibasic calcium phosphate dihydrate, FD&C red #40 aluminum lake, FD&C yellow #6 aluminum lake, hypromellose, magnesium stearate, microcrystalline cellulose, polydextrose, polyethylene glycol, silicon dioxide, titanium dioxide, triacetin
Principal Display Panel
NDC 50844-211-14
*Compare to active ingredient in Sudafed® Sinus Congestion
Maximum Strength
Pseudoephedrine HCl
30 mg
Nasal Decongestant
• Nasal & Sinus Congestion
• Non-Drowsy
• Sinus Pressure
Actual Size
500 Tablets
Rx
For Pharmacist
Dispensing Only
NOT FOR
RETAIL SALE
TAMPER EVIDENT: DO NOT USE IF IMPRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
*This product is not manufactured or distributed by Johnson & Johnson Corporation, owner of the registered trademark Sudafed® Sinus Congestion.
50844 REV0619A11214
Distributed by LNK INTERNATIONAL, INC.
60 Arkay Drive, Hauppauge, NY 11788 USA
Amerisource 44-112
| PSEUDOEPHEDRINE HCL
pseudoephedrine hcl tablet, film coated |
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| Labeler - L.N.K. International, Inc. (038154464) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 038154464 | pack(50844-211) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 832867837 | pack(50844-211) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 832867894 | manufacture(50844-211) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 868734088 | pack(50844-211) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 967626305 | pack(50844-211) | |