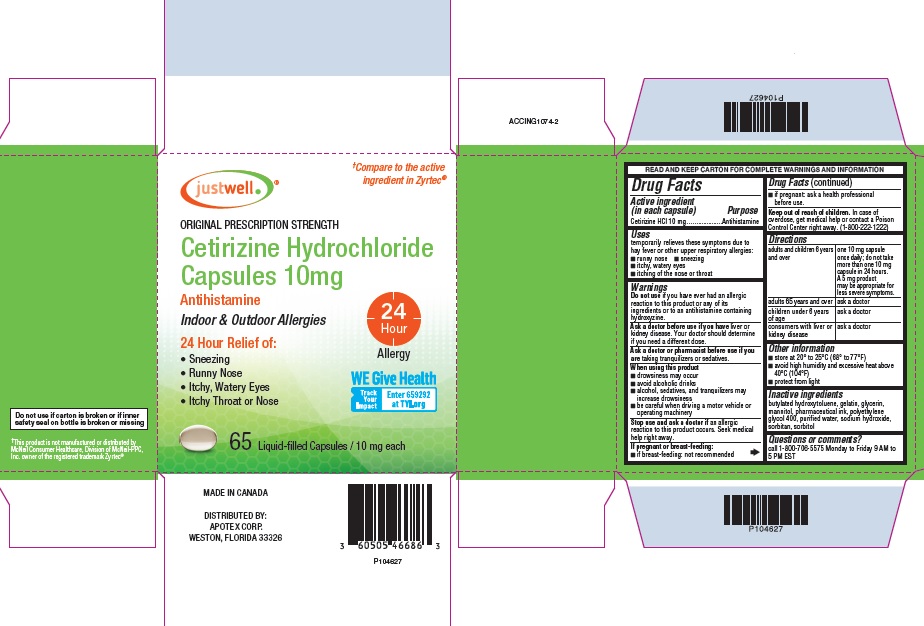
CETIRIZINE- cetirizine capsule, liquid filled
Apotex Corp.
----------
Drug Facts
Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- sneezing
- itchy, watery eyes
- itching of the nose or throat
Warnings
Do not use if you have ever had an allergic reaction to this product or any of its ingredients or to an antihistamine containing hydroxyzine.
Ask a doctor before use if you have liver or kidney disease.
Your doctor should determine if you need a different dose.
When using this product
- drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
Stop use and ask a doctor if an allergic reaction to this product occurs. Seek medical help right away.
Directions
| adults and children 6 years and over | one 10 mg capsule once daily; do not take more than one 10 mg capsule in 24 hours. A 5 mg product may be appropriate for less severe symptoms. |
| adults 65 years and over | ask a doctor |
| children under 6 years of age | ask a doctor |
| consumers with liver or kidney disease | ask a doctor |
Other information
- store at 20° to 25°C (68° to 77°F)
- avoid high humidity and excessive heat above 40°C (104°F)
- protect from light
Inactive ingredients
butylated hydroxytoluene, gelatin, glycerin, mannitol, pharmaceutical ink, polyethylene glycol 400, purified water, sodium hydroxide, sorbitan, sorbitol
| CETIRIZINE
cetirizine capsule, liquid filled |
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
| Labeler - Apotex Corp. (845263701) |
| Registrant - Apotex Inc. (209429182) |
Revised: 2/2021
Document Id: c7ca2e17-5968-18b0-8d37-8bf875adc114
Set id: 5ddd5bfe-eb1b-2129-decc-eec4788c956d
Version: 4
Effective Time: 20210203
Apotex Corp.