BUDESONIDE- budesonide capsule
Par Pharmaceutical Inc.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use budesonide safely and effectively. See full prescribing information for budesonide.
Budesonide Capsules, for oral use Initial U.S. Approval: 1997 INDICATIONS AND USAGEBudesonide is a glucocorticosteroid indicated for: DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHSCapsules: 3 mg (3) CONTRAINDICATIONSHypersensitivity to any of the ingredients in budesonide. (4) WARNINGS AND PRECAUTIONS
ADVERSE REACTIONSMost common adverse reactions (≥ 5%) are headache, respiratory infection, nausea, back pain, dyspepsia, dizziness, abdominal pain, flatulence, vomiting, fatigue, pain. (6.1) To report SUSPECTED ADVERSE REACTIONS, contact AstraZeneca at 1-800-236-9933 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. DRUG INTERACTIONSUSE IN SPECIFIC POPULATIONSSee 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling. Revised: 12/2011 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Mild to Moderate Active Crohn’s Disease
The recommended adult dosage for the treatment of mild to moderate active Crohn's disease involving the ileum and/or the ascending colon is 9 mg orally taken once daily in the morning for up to 8 weeks. Repeated 8 week courses of budesonide can be given for recurring episodes of active disease.
2.2 Maintenance of Clinical Remission of Mild to Moderate Crohn’s Disease
Following an 8 week course(s) of treatment for active disease and once the patient’s symptoms are controlled (CDAI less than 150), budesonide 6 mg orally is recommended once daily for maintenance of clinical remission up to 3 months. If symptom control is still maintained at 3 months an attempt to taper to complete cessation is recommended. Continued treatment with budesonide 6 mg for more than 3 months has not been shown to provide substantial clinical benefit.
Patients with mild to moderate active Crohn’s disease involving the ileum and/or ascending colon have been switched from oral prednisolone to budesonide with no reported episodes of adrenal insufficiency. Since prednisolone should not be stopped abruptly, tapering should begin concomitantly with initiating budesonide treatment.
2.3 CYP3A4 inhibitors
If concomitant administration with ketoconazole, or any other CYP3A4 inhibitor, is indicated, patients should be closely monitored for increased signs and/or symptoms of hypercorticism. Grapefruit juice, which is known to inhibit CYP3A4, should also be avoided when taking budesonide. In these cases, reduction in the dose of budesonide capsules should be considered [see Drug Interactions (7) and Clinical Pharmacology (12.3)].
3 DOSAGE FORMS AND STRENGTHS
Budesonide 3 mg capsules are hard gelatin capsules with an opaque light grey body and an opaque pink cap, coded with ENTOCORT EC 3 mg on the capsule.
4 CONTRAINDICATIONS
Budesonide is contraindicated in patients with hypersensitivity to budesonide or any of the ingredients of budesonide. Anaphylactic reactions have occurred [see Adverse Reactions (6.2)].
5 WARNINGS AND PRECAUTIONS
5.1 Hypercorticism and Adrenal Suppression
When glucocorticosteroids are used chronically, systemic effects such as hypercorticism and adrenal suppression may occur. Glucocorticosteroids can reduce the response of the hypothalamus-pituitary-adrenal (HPA) axis to stress. In situations where patients are subject to surgery or other stress situations, supplementation with a systemic glucocorticosteroid is recommended. Since budesonide is a glucocorticosteroid, general warnings concerning glucocorticoids should be followed.
5.2 Transferring Patients from Systemic Glucocorticosteroid Therapy
Care is needed in patients who are transferred from glucocorticosteroid treatment with high systemic effects to corticosteroids with lower systemic availability, such as budesonide, since symptoms attributed to withdrawal of steroid therapy, including those of acute adrenal suppression or benign intracranial hypertension, may develop. Adrenocortical function monitoring may be required in these patients and the dose of glucocorticosteroid treatment with high systemic effects should be reduced cautiously.
5.3 Immunosuppression
Patients who are on drugs that suppress the immune system are more susceptible to infection than healthy individuals. Chicken pox and measles, for example, can have a more serious or even fatal course in susceptible patients or patients on immunosuppressant doses of glucocorticosteroids. In patients who have not had these diseases, particular care should be taken to avoid exposure.
How the dose, route and duration of glucocorticosteroid administration affect the risk of developing a disseminated infection is not known. The contribution of the underlying disease and/or prior glucocorticosteroid treatment to the risk is also not known. If exposed, therapy with varicella zoster immune globulin (VZIG) or pooled intravenous immunoglobulin (IVIG), as appropriate, may be indicated. If exposed to measles, prophylaxis with pooled intramuscular immunoglobulin (IG) may be indicated. (See prescribing information for VZIG and IG.) If chicken pox develops, treatment with antiviral agents may be considered.
Glucocorticosteroids should be used with caution, if at all, in patients with active or quiescent tuberculosis infection, untreated fungal, bacterial, systemic viral or parasitic infections.
Replacement of systemic glucocorticosteroids with budesonide capsules may unmask allergies (e.g., rhinitis and eczema), which were previously controlled by the systemic drug.
5.4 Increased Systemic Glucocorticosteroid Susceptibility
Reduced liver function affects the elimination of glucocorticosteroids, and increased systemic availability of oral budesonide has been demonstrated in patients with liver cirrhosis [see Use in Specific Populations (8.6)].
6 ADVERSE REACTIONS
- Systemic glucocorticosteroid use may result in the following:
- •
- Hypercorticism and Adrenal Suppression [see Warnings and Precautions (5.1)]
- •
- Symptoms of steroid withdrawal in those patients transferring from Systemic Glucocorticosteroid Therapy [see Warnings and Precautions (5.2)]
- •
- Immunosuppression [see Warnings and Precautions (5.3)]
- •
- Increased Systemic Glucocorticosteroid Susceptibilty [see Warnings and Precautions (5.4)]
- •
- Other Glucocorticosteroid Effects [see Warnings and Precautions (5.5)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of budesonide was evaluated in 651 patients in five short-term, active disease state studies. They ranged in age from 17 to 74 (mean 35), 40% were male and 97% were white, 2.6% were greater than or equal to 65 years of age. Five hundred and twenty patients were treated with budesonide 9 mg (total daily dose). The most common adverse reactions reported were headache, respiratory infection, nausea, and symptoms of hypercorticism. Clinical studies have shown that the frequency of glucocorticosteroid-associated adverse reactions was substantially reduced with budesonide capsules compared with prednisolone at therapeutically equivalent doses. Adverse reactions occurring in greater than or equal to 5% of the patients are listed in Table 1:
|
||||
|
Budesonide 9 mg n=520 |
Placeob n=107 |
Prednisolone 40 mg n=145 |
Comparator* n=88 |
|
|
Adverse Reaction |
Number (%) |
Number (%) |
Number (%) |
Number (%) |
|
Headache |
107 (21) |
19 (18) |
31 (21) |
11 (13) |
|
Respiratory Infection |
55 (11) |
7 (7) |
20 (14) |
5 (6) |
|
Nausea |
57 (11) |
10 (9) |
18 (12) |
7 (8) |
|
Back Pain |
36 (7) |
10 (9) |
17 (12) |
5 (6) |
|
Dyspensia |
31 (6) |
4 (4) |
17 (12) |
3 (3) |
|
Dizziness |
38 (7) |
5 (5) |
18 (12) |
5 (6) |
|
Abdominal Pain |
32 (6) |
18 (17) |
6 (4) |
10 (11) |
|
Flatulence |
30 (6) |
6 (6) |
12 (8) |
5 (6) |
|
Vomiting |
29 (6) |
6 (6) |
6 (4) |
6 (7) |
|
Fatigue |
25 (5) |
8 (7) |
11 (8) |
0 (0) |
|
Pain |
24 (5) |
8 (7) |
17 (12) |
2 (2) |
The safety of budesonide was evaluated in 233 patients in four long-term clinical trials (52 weeks). A total of 145 patients were treated with budesonide 6 mg. A total of 8% of budesonide patients discontinued treatment due to adverse reactions compared with 10% in the placebo group. The adverse reaction profile in long-term treatment of Crohn’s disease was similar to that of short-term treatment with budesonide 9 mg in active Crohn’s disease.
In the long-term clinical trials, the following adverse reactions occurred in greater than or equal to 5% of the 6 mg budesonide patients and are not listed in (Table 1) or by body system below: diarrhea (10%); sinusitis (8%); infection viral (6%); and arthralgia (5%).
Adverse reactions, occurring in patients treated with budesonide 9 mg (total daily dose) in short-term active disease state studies and/or budesonide 6 mg (total daily dose) long-term, with an incidence less than 5% and greater than placebo are listed below by system organ class:
Blood and lymphatic system disorders: leukocytosis
Cardiac disorders: palpitation, tachycardia
Eye disorders: eye abnormality, vision abnormal
General disorders and administration site conditions: asthenia, chest pain, dependent edema, face edema, flu-like disorder, malaise, fever
Gastrointestinal disorders: anus disorder, Crohn’s disease aggravated, enteritis, epigastric pain, gastrointestinal fistula, glossitis, hemorrhoids, intestinal obstruction, tongue edema, tooth disorder
Infections and infestations: Ear infection-not otherwise specified, bronchitis, abscess, rhinitis, urinary tract infection, thrush
Investigations: c-reactive protein increased, weight increased
Metabolism and nutrition disorders: appetite increased, hypokalemia
Musculoskeletal and connective tissue disorders: arthritis, cramps, myalgia
Nervous system disorders: hyperkinesia, parasthesia, tremor, vertigo, dizziness, somnolence, amnesia
Psychiatric disorders: agitation, confusion, insomnia, nervousness, sleep disorder
Renal and urinary disorders: dysuria, micturition frequency, nocturia
Reproductive system and breast disorders: intermenstrual bleeding, menstrual disorder
Respiratory, thoracic and mediastinal disorders: dyspnea, pharynx disorder
Skin and subcutaneous tissue disorders: acne, alopecia, dermatitis, eczema, skin disorder, sweating increased, purpura
Vascular disorders: flushing,hypertension
Table 2 displays the frequency and incidence of signs/symptoms of hypercorticism by active questioning of patients in short-term clinical trials.
|
Budesonide 9 mg n=427 |
Placebo n=107 |
Prednisolome Taper 40 mg n=145 |
|
|
Sign/Symptom |
Number (%) |
Number (%)) |
Number (%) |
|
Acne |
63 (15) |
14 (13) |
33 (23)* |
|
Bruising Easily |
63 (15) |
12 (11) |
13 (9) |
|
Moon Face |
46 (11) |
4 (4) |
53 (37)* |
|
Swollen Ankles |
32 (7) |
6 (6) |
13 (9) |
|
Hirsutism† |
22 (5) |
2 (2) |
5 (3) |
|
Buffalo Hump |
6 (1) |
2 (2) |
5 (3) |
|
Skin Striae |
4 (1) |
2 (2) |
0 (0) |
Table 3 displays the frequency and incidence of signs/symptoms of hypercorticism by active questioning of patients in long-term clinical trials.
|
Budesonide 3 mg n=88 |
Budesonide 6 mg n=145 |
Placebo n=143 |
|
|
Sign/Symptom |
Number (%) |
Number (%) |
Number (%) |
|
Bruising easily |
4 (5) |
15 (10) |
5 (4) |
|
Acne |
4 (5) |
14 (10) |
3 (2) |
|
Moon face |
3 (3) |
6 (4) |
0 |
|
Hirsutism |
2 (2) |
5 (3) |
1 (1) |
|
Swollen ankles |
2 (2) |
3 (2) |
3 (2) |
|
Buffalo hump |
1 (1) |
1 (1) |
0 |
|
Skin Striae |
2 (2) |
0 |
0 |
The incidence of signs/symptoms of hypercorticism as described above in long-term clinical trials was similar to that seen in the short-term clinical trials.
A randomized, open, parallel-group multicenter safety study specifically compared the effect of budesonide (less than 9 mg per day) and prednisolone (less than 40 mg per day) on bone mineral density over 2 years when used at doses adjusted to disease severity. Bone mineral density decreased significantly less with budesonide than with prednisolone in steroid-naïve patients, whereas no difference could be detected between treatment groups for steroid-dependent patients and previous steroid users. The incidence of symptoms associated with hypercorticism was significantly higher with prednisolone treatment.
Clinical Laboratory Test Findings
The following potentially clinically significant laboratory changes in clinical trials, irrespective of relationship to budesonide, were reported in greater than or equal to 1% of patients: hypokalemia, leukocytosis, anemia, hematuria, pyuria, erythrocyte sedimentation rate increased, alkaline phosphatase increased, atypical neutrophils, C-reactive protein increased, and adrenal insufficiency.
6.2 Postmarketing Experience
The following adverse reactions have been reported during post-approval use of budesonide. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune System Disorders: Anaphylactic reactions
Nervous System Disorders:Benign intracranial hypertension
Psychiatric Disorders: Mood swings
7 DRUG INTERACTIONS
Concomitant oral administration of ketoconazole (a known inhibitor of CYP3A4 activity in the liver and in the intestinal mucosa) caused an eight-fold increase of the systemic exposure to oral budesonide. If treatment with inhibitors of CYP3A4 activity (such as ketoconazole, itraconazole, ritonavir, indinavir, saquinavir, erythromycin, etc.) is indicated, reduction of the budesonide dose should be considered. After extensive intake of grapefruit juice (which inhibits CYP3A4 activity predominantly in the intestinal mucosa), the systemic exposure for oral budesonide increased about two times. Ingestion of grapefruit or grapefruit juice should be avoided in connection with budesonide administration [see Clinical Pharmacology (12.3)]..
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Teratogenic Effects:Pregnancy Category C: Budesonide was teratogenic and embryocidal in rabbits and rats. Budesonide produced fetal loss, decreased pup weights, and skeletal abnormalities at subcutaneous doses of 25 mcg/kg in rabbits (approximately 0.05 times the maximum recommended human dose on a body surface area basis) and 500 mcg/kg in rats (approximately 0.5 times the maximum recommended human dose on a body surface area basis).
There are no adequate and well-controlled studies in pregnant women. Budesonide should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nonteratogenic Effects: Hypoadrenalism may occur in infants born of mothers receiving corticosteroids during pregnancy. Such infants should be carefully observed.
8.3 Nursing Mothers
The disposition of budesonide when delivered by inhalation from a dry powder inhaler at doses of 200 or 400 mcg twice daily for at least 3 months was studied in eight lactating women with asthma from 1 to 6 months postpartum1. Systemic exposure to budesonide in these women appears to be comparable to that in non-lactating women with asthma from other studies. Breast milk obtained over eight hours post-dose revealed that the maximum budesonide concentration for the 400 and 800 mcg total daily doses was 0.39 and 0.78 nmol/L, respectively, and occurred within 45 minutes after inhalation. The estimated oral daily dose of budesonide from breast milk to the infant is approximately 0.007 and 0.014 mcg/kg per day for the two dose regimens used in this study, which represents approximately 0.3% to 1% of the dose inhaled by the mother. Budesonide plasma concentrations obtained from five infants at about 90 minutes after breast feeding (and about 140 minutes after drug administration to the mother) were below quantifiable levels (less than 0.02 nmol/L in four infants and less than 0.04 nmol/L in one infant).
The recommended daily dose of budesonide capsules is higher (up to 9 mg daily) compared with inhaled budesonide (up to 800 mg daily) given to mothers in the above study. The maximum budesonide plasma concentration following a 9 mg daily dose (in both single- and repeated-dose pharmacokinetic studies) of oral budesonide is approximately 5-10 nmol/L which is up to 10 times higher than the 1-2 nmol/L for a 800 mg daily dose of inhaled budesonide at steady state in the above inhalation study.
Since there are no data from controlled trials on the use of budesonide by nursing mothers or their infants, and because of the potential for serious adverse reactions in nursing infants from budesonide, a decision should be made whether to discontinue nursing or to discontinue budesonide, taking into account the clinical importance of budesonide to the mother.
Budesonide is secreted in human milk. Data from budesonide delivered via dry powder inhaler indicates that the total daily oral dose of budesonide available in breast milk to the infant is approximately 0.3% to 1% of the dose inhaled by the mother. Assuming the coefficient of extrapolation between the inhaled and oral doses is constant across all dose levels, at therapeutic doses of budesonide, budesonide exposure to the nursing child may be up to 10 times higher than that by budesonide inhalation.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established. Systemic and inhaled corticosteroids, including budesonide, may cause a reduction of growth velocity in pediatric patients.
8.5 Geriatric Use
Clinical studies of budesonide did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
8.6 Hepatic Insufficiency
Patients with moderate to severe liver disease should be monitored for increased signs and/or symptoms of hypercorticism. Reducing the dose of budesonide capsules should be considered in these patients [see Warnings and Precautions (5.4)].
10 OVERDOSAGE
Reports of acute toxicity and/or death following overdosage of glucocorticosteroids are rare. Treatment consists of immediate gastric lavage or emesis followed by supportive and symptomatic therapy.
If glucocorticosteroids are used at excessive doses for prolonged periods, systemic glucocorticosteroid effects such as hypercorticism and adrenal suppression may occur. For chronic overdosage in the face of severe disease requiring continuous steroid therapy, the dosage may be reduced temporarily.
Single oral doses of 200 and 400 mg/kg were lethal in female and male mice, respectively. The signs of acute toxicity were decreased motor activity, piloerection and generalized edema.
11 DESCRIPTION
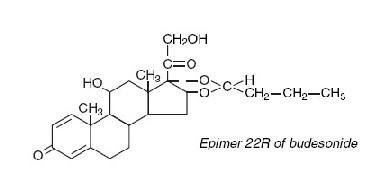
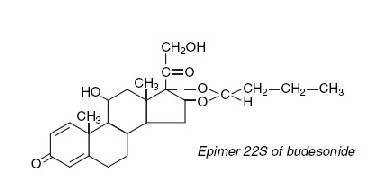
Budesonide, the active ingredient of budesonide capsules, is a synthetic corticosteroid. Budesonide is designated chemically as (RS)-11β, 16α, 17,21-tetrahydroxypregna-1,4-diene-3,20-dione cyclic 16,17-acetal with butyraldehyde. Budesonide is provided as a mixture of two epimers (22R and 22S). The empirical formula of budesonide is C25H34O6 and its molecular weight is 430.5. Its structural formula is:
Budesonide is a white to off-white, tasteless, odorless powder that is practically insoluble in water and heptane, sparingly soluble in ethanol, and freely soluble in chloroform. Its partition coefficient between octanol and water at pH 5 is 1.6 x 103 ionic strength 0.01.
Each capsule for oral administration contains 3 mg of micronized budesonide with the following inactive ingredients: ethylcellulose, acetyltributyl citrate, methacrylic acid copolymer type C, triethyl citrate, antifoam M, polysorbate 80, talc, and sugar spheres. The capsule shells have the following inactive ingredients: gelatin, iron oxide, and titanium dioxide.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Budesonide has a high topical glucocorticosteroid (GCS) activity and a substantial first pass elimination. The formulation contains granules which are coated to protect dissolution in gastric juice, but which dissolve at pH greater than 5.5, ie, normally when the granules reach the duodenum. Thereafter, a matrix of ethylcellulose with budesonide controls the release of the drug into the intestinal lumen in a time-dependent manner.
12.2 Pharmacodynamics
Budesonide has a high glucocorticoid effect and a weak mineralocorticoid effect, and the affinity of budesonide to GCS receptors, which reflects the intrinsic potency of the drug, is about 200-fold that of cortisol and 15-fold that of prednisolone.
Treatment with systemically active GCS is associated with a suppression of endogenous cortisol concentrations and an impairment of the hypothalamus-pituitary-adrenal (HPA) axis function. Markers, indirect and direct, of this are cortisol levels in plasma or urine and response to ACTH stimulation.
Plasma cortisol suppression was compared following five days’ administration of budesonide capsules and prednisolone in a crossover study in healthy volunteers. The mean decrease in the integrated 0-24 hour plasma cortisol concentration was greater (78%) with prednisolone 20 mg per day compared to 45% with budesonide 9 mg per day.
12.3 Pharmacokinetics
Absorption
The absorption of budesonide seems to be complete, although Cmax and Tmax are variable. Time to peak concentration varies in individual patients between 30 and 600 minutes. Following oral administration of 9 mg of budesonide in healthy subjects, a peak plasma concentration of approximately 5 nmol/L is observed and the area under the plasma concentration time curve is approximately 30 nmol·hr/L. The systemic availability after a single dose is higher in patients with Crohn's disease compared to healthy volunteers, (21% vs 9%) but approaches that in healthy volunteers after repeated dosing.
Distribution
The mean volume of distribution (Vss) of budesonide varies between 2.2 and 3.9 L/kg in healthy subjects and in patients. Plasma protein binding is estimated to be 85 to 90% in the concentration range 1 to 230 nmol/L, independent of gender. The erythrocyte/plasma partition ratio at clinically relevant concentrations is about 0.8.
Metabolism
Following absorption, budesonide is subject to high first pass metabolism (80-90%). In vitro experiments in human liver microsomes demonstrate that budesonide is rapidly and extensively biotransformed, mainly by CYP3A4, to its 2 major metabolites, 6β-hydroxy budesonide and 16α-hydroxy prednisolone. The glucocorticoid activity of these metabolites is negligible (less than 1/100) in relation to that of the parent compound.
In vivo investigations with intravenous doses in healthy subjects are in agreement with the in vitro findings and demonstrate that budesonide has a high plasma clearance, 0.9-1.8 L/min. Similarly, high plasma clearance values have been shown in patients with Crohn’s disease. These high plasma clearance values approach the estimated liver blood flow, and, accordingly, suggest that budesonide is a high hepatic clearance drug.
The plasma elimination half-life, t1/2, after administration of intravenous doses ranges between 2 and 3.6 hours, and does not differ between healthy adults and patients with Crohn’s disease.
Excretion
Budesonide is excreted in urine and feces in the form of metabolites. After oral as well as intravenous administration of micronized [3H]-budesonide, approximately 60% of the recovered radioactivity is found in urine. The major metabolites, including 6β-hydroxy budesonide and 16α-hydroxy prednisolone, are mainly renally excreted, intact or in conjugated forms. No unchanged budesonide is detected in urine.
Special Populations
Gender
No significant pharmacokinetic differences have been identified due to gender.
Hepatic Insufficiency
In patients with liver cirrhosis, systemic availability of orally administered budesonide correlates with disease severity and is, on average, 2.5-fold higher compared with healthy controls. Patients with mild liver disease are minimally affected. Patients with severe liver dysfunction were not studied. Absorption parameters are not altered, and for the intravenous dose, no significant differences in CL or Vss are observed.
Renal Insufficiency
The pharmacokinetics of budesonide in patients with renal impairment has not been studied. Intact budesonide is not renally excreted, but metabolites are to a large extent, and might therefore reach higher levels in patients with impaired renal function. However, these metabolites have negligible corticosteroid activity as compared with budesonide (less than 1/100).
Drug-Drug Interactions
Budesonide is metabolized via CYP3A4. Potent inhibitors of CYP3A4 can increase the plasma levels of budesonide several-fold. Co-administration of ketoconazole results in an eight-fold increase in AUC of budesonide, compared to budesonide alone. Grapefruit juice, an inhibitor of gut mucosal CYP3A, approximately doubles the systemic exposure of oral budesonide. Conversely, induction of CYP3A4 can result in the lowering of budesonide plasma levels.
Oral contraceptives containing ethinyl estradiol, which are also metabolized by CYP3A4, do not affect the pharmacokinetics of budesonide. Budesonide does not affect the plasma levels of oral contraceptives (ie, ethinyl estradiol) [see Drug Interactions (7)].
Since the dissolution of the coating of budesonide is pH dependent (dissolves at pH greater than 5.5), the release properties and uptake of the compound may be altered after treatment with drugs that change the gastrointestinal pH. However, the gastric acid inhibitory drug omeprazole, 20 mg once daily does not affect the absorption or pharmacokinetics of budesonide. When an uncoated oral formulation of budesonide is co-administered with a daily dose of cimetidine 1 g, a slight increase in the budesonide peak plasma concentration and rate of absorption occurs, resulting in significant cortisol suppression.
Food Effects
A mean delay in time to peak concentration of 2.5 hours is observed with the intake of a high-fat meal, with no significant differences in AUC.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies with budesonide were conducted in rats and mice. In a two-year study in Sprague-Dawley rats, budesonide caused a statistically significant increase in the incidence of gliomas in male rats at an oral dose of 50 mcg/kg (approximately 0.05 times the maximum recommended human dose on a body surface area basis). In addition, there were increased incidences of primary hepatocellular tumors in male rats at 25 mcg/kg (approximately 0.023 times the maximum recommended human dose on a body surface area basis) and above. No tumorigenicity was seen in female rats at oral doses up to 50 mcg/kg (approximately 0.05 times the maximum recommended human dose on a body surface area basis). In an additional two-year study in male Sprague-Dawley rats, budesonide caused no gliomas at an oral dose of 50 mcg/kg (approximately 0.05 times the maximum recommended human dose on a body surface area basis). However, it caused a statistically significant increase in the incidence of hepatocellular tumors at an oral dose of 50 mcg/kg (approximately 0.05 times the maximum recommended human dose on a body surface area basis). The concurrent reference corticosteroids (prednisolone and triamcinolone acetonide) showed similar findings. In a 91-week study in mice, budesonide caused no treatment-related carcinogenicity at oral doses up to 200 mcg/kg (approximately 0.1 times the maximum recommended human dose on a body surface area basis).
Budesonide was not genotoxic in the Ames test, the mouse lymphoma cell forward gene mutation (TK +/-) test, the human lymphocyte chromosome aberration test, the Drosophila melanogaster sex-linked recessive lethality test, the rat hepatocyte UDS test and the mouse micronucleus test.
In rats, budesonide had no effect on fertility at subcutaneous doses up to 80 mcg/kg (approximately 0.07 times the maximum recommended human dose on a body surface area basis). However, it caused a decrease in prenatal viability and viability in pups at birth and during lactation, along with a decrease in maternal body-weight gain, at subcutaneous doses of 20 mcg/kg (approximately 0.02 times the maximum recommended human dose on a body surface area basis) and above. No such effects were noted at 5 mcg/kg (approximately 0.005 times the maximum recommended human dose on a body surface area basis).
14 CLINICAL STUDIES
The safety and efficacy of budesonide were evaluated in 994 patients with mild to moderate active Crohn’s disease of the ileum and/or ascending colon in 5 randomized and double-blind studies. The study patients ranged in age from 17 to 85 (mean 35), 40% were male and 97% were white. Of the 651 patients treated with budesonide, 17 (2.6%) were greater than or equal to 65 years of age and none were greater than 74 years of age. The Crohn’s Disease Activity Index (CDAI) was the main clinical assessment used for determining efficacy in these 5 studies. The CDAI is a validated index based on subjective aspects rated by the patient (frequency of liquid or very soft stools, abdominal pain rating and general well-being) and objective observations (number of extraintestinal symptoms, need for antidiarrheal drugs, presence of abdominal mass, body weight and hematocrit). Clinical improvement, defined as a CDAI score of less than or equal to 150 assessed after 8 weeks of treatment, was the primary efficacy variable in these 5 comparative efficacy studies of budesonide capsules. Safety assessments in these studies included monitoring of adverse reactions. A checklist of potential symptoms of hypercorticism was used.
One study (Study 1) compared the safety and efficacy of budesonide 9 mg daily in the morning to a comparator. At baseline, the median CDAI was 272. budesonide 9 mg daily resulted in a significantly higher clinical improvement rate at Week 8 than the comparator. See Table 4.
|
|||||
|
Clinical Study |
Budesonide |
Comparator* |
Placebo |
Prednisolone |
|
|
9 mg Daily |
4.5 mg Twice Daily | ||||
|
1 |
62/91 (69%) |
37/83 (45%) | |||
|
2 |
31/61 (51%) |
13/64 (20%) | |||
|
3 |
38/79 (48%) |
41/78 (53%) |
13/40 (33%) | ||
|
4 |
35/58 (60%) |
25/60 (42%) |
35/58 (60%) |
||
|
5 |
45/86 (52%) |
56/85 (65%) |
|||
Two placebo-controlled clinical trials (Studies 2 and 3) were conducted. Study 2 involved 258 patients and tested the effects of graded doses of budesonide (1.5 mg twice daily, 4.5 mg twice daily, or 7.5 mg twice daily) versus placebo. At baseline, the median CDAI was 290. The 3 mg per day dose level (data not shown) could not be differentiated from placebo. The 9 mg per day arm was statistically different from placebo (Table 4), while no additional benefit was seen when the daily budesonide dose was increased to 15 mg per day (data not shown). In Study 3, the median CDAI at baseline was 263. Neither 9 mg daily nor 4.5 mg twice daily budesonide dose levels was statistically different from placebo (Table 4).
Two clinical trials (Studies 4 and 5) compared budesonide capsules with oral prednisolone (initial dose 40 mg per day). At baseline, the median CDAI was 277. Equal clinical improvement rates (60%) were seen in the budesonide 9 mg daily and the prednisolone groups in Study 4. In Study 5, 13% fewer patients in the budesonide group experienced clinical improvement than in the prednisolone group (no statistical difference) (Table 4).
The proportion of patients with normal plasma cortisol values (greater than 150 nmol/L) was significantly higher in the budesonide groups in both trials (60 to 66%) than in the prednisolone groups (26 to 28%) at Week 8.
The efficacy and safety of budesonide for maintenance of clinical remission were evaluated in four double-blind, placebo-controlled, 12-month trials in which 380 patients were randomized and treated once daily with 3 mg or 6 mg budesonide or placebo. Patients ranged in age from 18 to 73 (mean 37) years. Sixty percent of the patients were female and 99% were Caucasian. The mean CDAI at entry was 96. Among the four clinical trials, approximately 75% of the patients enrolled had exclusively ileal disease. Colonoscopy was not performed following treatment. budesonide 6 mg per day prolonged the time to relapse, defined as an increase in CDAI of at least 60 units to a total score greater than 150 or withdrawal due to disease deterioration. The median time to relapse in the pooled population of the 4 studies was 154 days for patients taking placebo, and 268 days for patients taking budesonide 6 mg per day. budesonide 6 mg per day reduced the proportion of patients with loss of symptom control relative to placebo in the pooled population for the 4 studies at 3 months (28% vs. 45% for placebo).
16 HOW SUPPLIED/STORAGE AND HANDLING
Budesonide 3 mg capsules are hard gelatin capsules with an opaque light grey body and an opaque pink cap, coded with ENTOCORT EC 3 mg on the capsule and are supplied as follows:
- NDC 49884-501-01 Bottles of 100
Store at 25°C (77°F); excursions permitted to 15-30°C (59-86°F) [See USP Controlled Room Temperature].
Keep container tightly closed.
17 PATIENT COUNSELING INFORMATION
“See FDA-Approved Patient Labeling (Patient Information)”
Patients being treated with Budesonide Capsules should receive the following information and instructions. This information is intended to aid the patient in the safe and effective use of the medication. It is not a disclosure of all possible adverse or intended effects. For proper use of Budesonide Capsules and to attain maximum improvement, the patient should read and follow the accompanying FDA-Approved Patient Labeling.
17.1 Hypercorticism and Adrenal Suppression
Patients should be advised that Budesonide Capsules may cause systemic glucocorticosteroid effects of hypercorticism and adrenal suppression. Patients should taper slowly from systemic glucocorticosteroids if transferring to Budesonide Capsules [see Warnings and Precautions (5.1) and (5.2)].
17.2 Immunosuppression
Patients who are on immunosuppressant doses of glucocorticosteroids should be warned to avoid exposure to chicken pox or measles and, if exposed, to consult their physician without delay. Patients should be informed of potential worsening of existing tuberculosis, fungal, bacterial, viral or parasitic infections [see Warnings and Precautions (5.3)].
17.3 How to Take Budesonide Capsules
Budesonide Capsules should be swallowed whole and NOT CHEWED OR BROKEN. Patients should be advised to avoid the consumption of grapefruit juice for the duration of their budesonide therapy.
© AstraZeneca 2010, 2011
Distributed by:
Par Pharmaceutical Companies, Inc
Spring Valley, NY 10977 U.S.A
Patient Information
Budesonide Capsules
Read the Patient Information that comes with budesonide before you start taking it and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or your treatment.
What is budesonide?
Budesonide is a prescription glucocorticosteroid medicine used in people with mild to moderate Crohn’s disease that affects part of the small intestine (ileum) and part of the large intestine (ascending colon):
- •
- to treat active Crohn’s disease
- •
- to help keep symptoms from coming back for up to 3 months
It is not known if budesonide is safe and effective in children.
Who should not take budesonide?
Do not take budesonide if:
- •
- you are allergic to budesonide or any of the ingredients in budesonide. See the end of this leaflet for a complete list of ingredients in budesonide.
What should I tell my healthcare provider before taking budesonide?
Before you take budesonide tell your healthcare provider if you:
- •
- have liver problems
- •
- are planning to have surgery
- •
- have chicken pox or measles or have recently been near anyone with chicken pox or measles
- •
- have or had a family history of diabetes, cataracts or glaucoma
- •
- have or had tuberculosis
- •
- have high blood pressure (hypertension)
- •
- have decreased bone mineral density (osteoporosis)
- •
- stomach ulcers
- •
- any other medical condition
- •
- are pregnant or plan to become pregnant. It is not known if budesonide may harm your unborn baby.
- •
- are breastfeeding or plan to breastfeed. budesonide can pass into breast milk and may harm your baby. You and your healthcare provider should decide if you will take budesonide or breastfeed. You should not do both.
Tell your healthcare provider about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements.Budesonide and other medicines may affect each other causing side effects.
Especially tell your healthcare providerif you take:
- •
- a glucocorticosteroid medicine
- •
- medicines that suppress your immune system (immunosuppressant)
- •
- ketoconazale or other medicines that affect how your liver works.
Ask your healthcare provideror pharmacist if you are not sure if your medicine is one listed above.
How should I take budesonide?
- •
- Take budesonide exactly as your healthcare providertells you.
- •
- Your healthcare providerwill tell you how many budesonide capsules to take. Your healthcare providermay change your dose if needed.
- •
- Take budesonide in the morning.
- •
- Take budesonide capsules whole. Do not chew or crush budesonide capsules before swallowing.
What should I avoid while taking budesonide?
- •
- Do not eat grapefruit or drink grapefruit juice while taking budesonide. Eating grapefruit or drinking grapefruit juice can increase the level of budesonide in your blood.
What are the possible side effects of budesonide?
- •
-
Effects of having too much glucocorticosteroid medicine in your blood (hypercorticism). Long-time use of budesonide can cause you to have too much glucocorticosteroid medicine in your blood. Tell your healthcare provider if you have any of the following signs and symptoms of hypercorticism:
- ∘
- acne
- ∘
- bruise easily
- ∘
- rounding of your face (moon face)
- ∘
- ankle swelling
- ∘
- thicker or more hair on your body and face
- ∘
- a fatty pad or hump between your shoulders (buffalo hump)
- ∘
- pink or purple stretch marks on the skin of your abdomen, thighs, breasts and arms
- •
- Adrenal suppression. When budesonide is taken for a long period of time (chronic use),adrenal suppression can happen. This is a condition in which the adrenal glands do not make enough steroid hormones. Symptoms of adrenal suppression include: tiredness, weakness, nausea and vomiting and low blood pressure. Tell your healthcare provider if you are under stress or have any symptoms of adrenal suppression during treatment with budesonide. Immune system effects and a higher chance of infections.
- •
-
Immune system effects and a higher chance of infections. Budesonide weakens your immune system. Taking medicines that weaken your immune system makes you more likely to get infections. Avoid contact with people who have contagious diseases such as chicken pox or measles, while taking budesonide.
Tell your healthcare provider about any signs or symptoms of infection during treatment with budesonide, including:- ∘
- fever
- ∘
- pain
- ∘
- aches
- ∘
- chills
- ∘
- feeling tired
- ∘
- nausea and vomiting
- •
- Worsening of allergies. If you take certain other glucocorticosteroid medicines to treat allergies, switching to budesonide may cause your allergies to come back. These allergies may include eczema (a skin disease) or rhinitis (inflammation inside your nose). Tell your healthcare provider if any of your allergies become worse while taking budesonide.
The most common side effects of budesonide include:
-
- ∘
- headache
- ∘
- infection in your air passages (respiratory infection)
- ∘
- back pain
- ∘
- upset stomach
- ∘
- dizziness
- ∘
- abdominal pain
- ∘
- excessive stomach or intestinal gas
- ∘
- diarrhea
- ∘
- sinus infection
- ∘
- viral infection
- ∘
- joint pain
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of budesonide. For more information, ask your healthcare provider or pharmacist.
Call your healthcare provider for medical advice about side effects. You may report side effects to AstraZeneca at 1-800-236-9933 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
How should I store budesonide?
- •
- Store budesonide at 59°F to 86°F (15°C to 30°C).
- •
- Keep budesonide in a tightly closed container.
Keep budesonide and all medicines out of reach from children.
General information about budesonide
Medicines are sometimes prescribed for purposes other than those listed in Patient Information leaflets. Do not use budesonide for a condition for which it was not prescribed. Do not give budesonide to other people, even if they have the same symptoms you have. It may harm them.
This Patient Information leaflet summarizes the most important information about budesonide. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about budesonide that is written for health professionals.
For more information go to.www.ENTOCORTEC.com or call 1-800-236-9933.
What are the ingredients in budesonide?
Active ingredient: budesonide
Inactive ingredients: ethylcellulose, acetyltributyl citrate, methacrylic acid copolymer type C, triethyl citrate, antifoam M, polysorbate 80, talc, and sugar spheres.
The capsule shell contains: gelatin, iron oxide, and titanium dioxide.
Distributed by:
Par Pharmaceutical Companies, Inc
Spring Valley, NY 10977 U.S.A.
This Patient Information has been approved by the U.S. Food and Drug Administration.
Revised: 2011
| BUDESONIDE
budesonide capsule |
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| Labeler - Par Pharmaceutical Inc. (092733690) |
| Registrant - AstraZeneca PLC (230790719) |