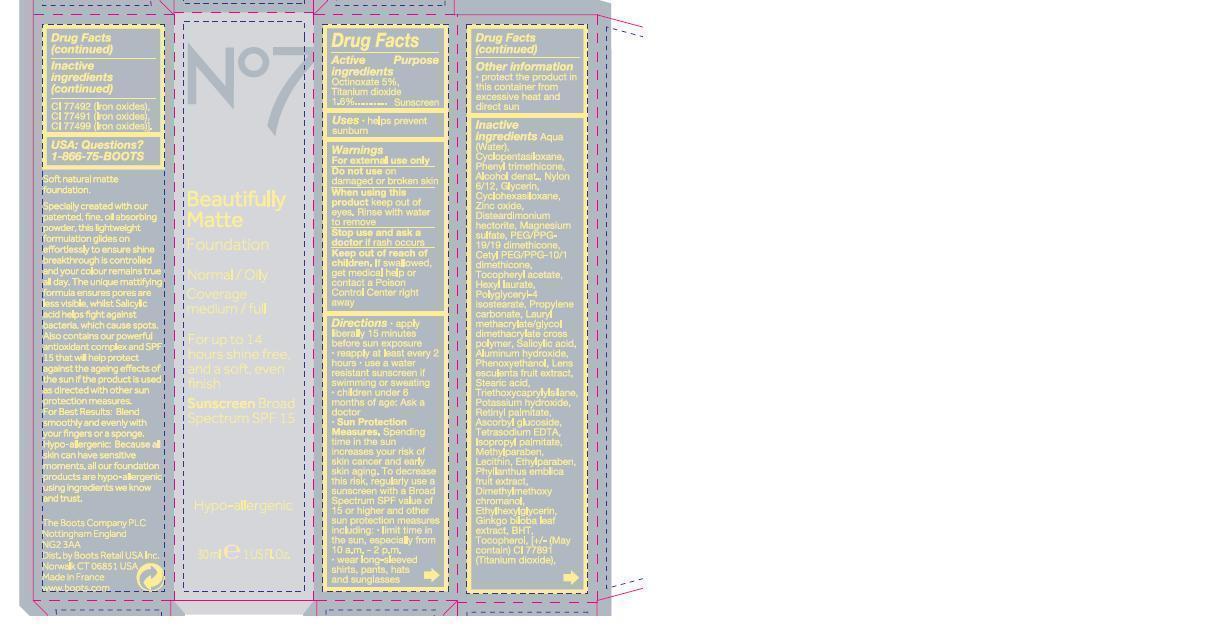
Label: NO7 BEAUTIFULLY MATTE FOUNDATION SUNSCREEN BROAD SPECTRUM SPF 15 DEEPLY BEIGE- octinoxate and titanium dioxide emulsion
-
Contains inactivated NDC Code(s)
NDC Code(s): 61589-6504-1 - Packager: BCM Cosmetique SAS
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 30, 2012
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- WARNINGS AND PRECAUTIONS
- ACTIVE INGREDIENT
- INDICATIONS & USAGE
-
INSTRUCTIONS FOR USE
Directions
apply liberally 15 minutes before sun exposure
reapply at least every 2 hours
use a water resistant sunscreen if swimming or sweating
children under 6 months of age ask a doctor
sun protectin measures
spending time in the sun increases your risk of skin cancer and early skin aging. to decrease this risk, regularly use a sunscreen with a broad spectrum SPF value of 15 or higher and other sun protection measures including limit time in the sun especially from 10 am to 2 pm, wear long sleeved shirts, pants, hats and sunglasses
- STORAGE AND HANDLING
-
DESCRIPTION
Soft natural matte foundation
Specially created with our patented, fine, oil absorbing powder, this lightweight formulation glides on effortlessly to ensure shine breakthrough is controlled and your colour remains true all day. The unique mattifying formula ensures pores are less visble, whilst Salicylic acid helps fight against bacteria, which cause spots. Also contains our powerful anitoxidant complex and SPF 15 that will help protect against the ageing effects of the sun if the product is used as directed with other sun protection measures. For best results blend smoothly and evenly with your fingers or a sponge. Hypo-allergenic because all skin can have sensitive moments, all our foundation products are hypo-allergenic using ingredients we know and trust.
-
INACTIVE INGREDIENT
inactive ingredients
aqua (water), cyclopentasiloxane, phenyl trimethicone, alcohol denat, nylon 6/12, glycerin, cyclohexsiloxane, zinc oxide, disteardimonium hectorite, magnesium sulfate, PEG PPG 19/19 dimethicone, tocopheryl acetate, hexyl laurate, polyglyceryl-4- isostearate, propylene carbonate,lauryl methacrylate/glycol dimethacrylate cross polymer, salicylic acid, aluminum hydroxide, phenoxyethanol, lens esculenta fruit extract, stearic acid, triethoxycaprylylsilane, potassium hydroxide, retinyl palmitate, ascorbyl glucoside, tetrasodium edta, isopropyl palmitate, methylparaben, lecithin, ethylparaben, phyllanthus emblica fruit extract, dimethoxyl chromanol, ethylhexylglycerin, ginkgo biloba leaf extract, BHT, tocopherol, may contain CI 77891 (titanium dioxide).
- INFORMATION FOR PATIENTS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
NO7 BEAUTIFULLY MATTE FOUNDATION SUNSCREEN BROAD SPECTRUM SPF 15 DEEPLY BEIGE
octinoxate and titanium dioxide emulsionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:61589-6504 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 1.5 g in 30 g TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 0.48 g in 30 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) GLYCERIN (UNII: PDC6A3C0OX) NYLON-12 (UNII: 446U8J075B) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) ZINC OXIDE (UNII: SOI2LOH54Z) CYCLOMETHICONE 6 (UNII: XHK3U310BA) MAGNESIUM SULFATE (UNII: DE08037SAB) ASCORBYL GLUCOSIDE (UNII: 2V52R0NHXW) PHENOXYETHANOL (UNII: HIE492ZZ3T) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) PEG/PPG-19/19 DIMETHICONE (UNII: EHH90CO7TL) .ALPHA.-TOCOPHEROL, DL- (UNII: 7QWA1RIO01) PHENYL TRIMETHICONE (UNII: DR0K5NOJ4R) PROPYLENE CARBONATE (UNII: 8D08K3S51E) LAURYL METHACRYLATE/GLYCOL DIMETHACRYLATE CROSSPOLYMER (UNII: EX0F4CZ66H) CETYL PEG/PPG-10/1 DIMETHICONE (HLB 1.5) (UNII: V2W71V8T0X) HEXYL LAURATE (UNII: 4CG9F9W01Q) POLYGLYCERYL-4 ISOSTEARATE (UNII: 820DPX33S7) METHYLPARABEN (UNII: A2I8C7HI9T) SALICYLIC ACID (UNII: O414PZ4LPZ) ETHYLPARABEN (UNII: 14255EXE39) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) VITAMIN A PALMITATE (UNII: 1D1K0N0VVC) TRIETHOXYCAPRYLYLSILANE (UNII: LDC331P08E) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) ALCOHOL (UNII: 3K9958V90M) STEARIC ACID (UNII: 4ELV7Z65AP) ETIDRONATE TETRASODIUM (UNII: CZZ9T1T1X4) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) ISOPROPYL PALMITATE (UNII: 8CRQ2TH63M) TOCOPHEROL (UNII: R0ZB2556P8) PHYLLANTHUS EMBLICA FRUIT (UNII: YLX4CW2576) GINKGO BILOBA LEAF OIL (UNII: Y5967KO1JH) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:61589-6504-1 1 in 1 CARTON 1 30 g in 1 BOTTLE, GLASS Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 10/26/2012 Labeler - BCM Cosmetique SAS (275359578) Registrant - The Boots Company PLC (218622660) Establishment Name Address ID/FEI Business Operations BCM Cosmetique SAS 275359578 manufacture(61589-6504)