Label: SORBITOL-MANNITOL- sorbitol and mannitol irrigant
- NDC Code(s): 0990-7981-08
- Packager: ICU Medical Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
Drug Label Information
Updated April 7, 2021
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
Sorbitol-Mannitol Irrigation is a sterile, nonpyrogenic, hypotonic, aqueous solution for urologic nonelectrolyte irrigation during transurethral surgical procedures. Each 100 mL contains sorbitol 2.70 g and mannitol 0.54 g in water for injection. The solution is nonelectrolytic and hypotonic (178 mOsmol/liter calc.); pH 5.2 (4.0 to 7.0).
The solution contains no bacteriostat, antimicrobial agent or added buffer and is intended only for use as a single-dose irrigation. When smaller volumes are required the unused portion should be discarded.
Sorbitol-Mannitol Irrigation is a nonelectrolyte urologic irrigant.
Sorbitol, NF is chemically designated D-glucitol (C6H14O6), white powder, granules or flakes very soluble in water. It has the following structural formula:
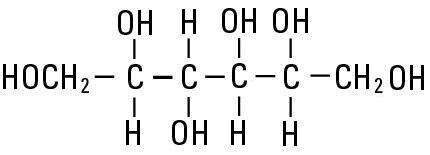
Mannitol, USP is chemically designated D-mannitol (C6H14O6), white crystalline powder or free-flowing granules, freely soluble in water. It has the following structural formula:
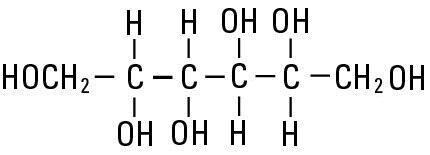
Water for Injection, USP is chemically designated H20.
The flexible plastic container is fabricated from a specially formulated polyvinylchloride. Water can permeate from inside the container into the overwrap but not in amounts sufficient to affect the solution significantly.
The semi-rigid container is fabricated from a specially formulated polyolefin. It is a copolymer of ethylene and propylene. The container requires no vapor barrier to maintain the proper drug concentration.
Solutions in contact with the plastic container may leach out certain chemical components from the plastic in very small amounts; however, biological testing was supportive of the safety of the plastic container materials.
Exposure to temperatures above 25°C/77°F during transport and storage will lead to minor losses in moisture content. Higher temperatures lead to greater losses. It is unlikely that these minor losses will lead to clinically significant changes within the expiration period.
-
CLINICAL PHARMACOLOGY
Sorbitol and mannitol are hexitols and are nonelectrolytes. A solution of these constituents in water is therefore nonconductive and suitable for urologic irrigation during electrosurgical procedures. A 3% (approx.) total concentration of sorbitol-mannitol contains sufficient solute to minimize the risk of intravascular hemolysis which can occur from absorption of plain water through open prostatic veins during transurethral resection (TUR). Any solution that is absorbed intravascularly during transurethral prostatic or bladder surgery, although variable in amount depending primarily on the extent of surgery, will be excreted by the kidney. When absorbed intravascularly, sorbitol and mannitol act as osmotic diuretics.
Intravascular absorption of sorbitol has been shown to produce elevations of serum lactate after TUR above preoperative values owing to sorbitol's favored metabolism to lactate from pyruvate. Increased lactate levels were not sufficient to produce evidence of metabolic acidosis. Mannitol is only slightly metabolized and rapidly excreted by the kidney.
- INDICATIONS AND USAGE
- CONTRAINDICATIONS
-
WARNINGS
FOR UROLOGIC IRRIGATION ONLY.
Solutions for urologic irrigation must be used with caution in patients with severe cardiopulmonary or renal dysfunction.
Irrigating fluids used during transurethral prostatectomy have been demonstrated to enter the systemic circulation in relatively large volumes; thus, sorbitol-mannitol irrigant must be regarded as a systemic drug. Absorption of large amounts of fluids containing sorbitol-mannitol and the osmotic diuresis it produces may significantly alter cardiopulmonary and renal dynamics.
Hyperglycemia from metabolism of sorbitol may occur in patients with diabetes mellitus.
Hyperlactatemia from metabolism of sorbitol may potentially produce a significant lactic acidemia in metabolically compromised patients.
The contents of an opened container should be used promptly to minimize the possibility of bacterial growth or pyrogen formation.
Discard the unused portion of irrigation solution since it contains no preservatives. Do not heat over 66°C (150°F).
-
PRECAUTIONS
Cardiovascular status, especially of the patient with cardiac disease, should be carefully observed before and during transurethral resection of the prostate when using Sorbitol-Mannitol Irrigation, because the quantity of fluid absorbed into the systemic circulation by opened prostatic veins may produce significant expansion of the extracellular fluid and lead to fulminating congestive heart failure.
Shift of sodium-free intracellular fluid into the extracellular compartment following systemic absorption of solution may lower serum sodium concentration and aggravate pre-existing hyponatremia.
Excessive loss of water and electrolytes may lead to serious imbalances. With continuous irrigation, loss of water may occur in excess of electrolytes, producing hypernatremia.
Sustained diuresis that results from transurethral irrigation with Sorbitol-Mannitol Irrigation may obscure and intensify inadequate hydration or hypovolemia.
Aseptic technique is essential for the use of sterile solutions for irrigation. The administration set should be attached promptly. Unused portions should be discarded and a fresh container of appropriate size used for the start-up of each cycle or repeat procedure.
Do not administer unless solution is clear, seal is intact and container is undamaged. Discard unused portion.
Carcinogenesis, Mutagenesis, Impairment of Fertility: Studies with Sorbitol-Mannitol Irrigation have not been performed to evaluate carcinogenic potential, mutagenic potential, or effects on fertility.
Nursing Mothers: Caution should be exercised when Sorbitol-Mannitol Irrigation is administered to a nursing woman.
Pregnancy: Teratogenic Effects.
Pregnancy Category C. Animal reproduction studies have not been conducted with Sorbitol-Mannitol Irrigation. It is also not known whether Sorbitol-Mannitol Irrigation can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Sorbitol-Mannitol Irrigation should be given to a pregnant woman only if clearly needed.
Pediatric Use: The safety and effectiveness of Sorbitol-Mannitol Irrigation have not been established. Its limited use in pediatric patients has been inadequate to fully define proper dosage and limitations for use.
-
ADVERSE REACTIONS
Adverse reactions may result from intravascular absorption of sorbitol and mannitol. The literature reports occasional adverse reactions from intravenous sorbitol-mannitol infusions. Consequences of absorption of urologic irrigating solutions include fluid and electrolyte disturbances such as acidosis, electrolyte loss, marked diuresis, urinary retention, edema, dryness of mouth, thirst and dehydration; cardiovascular disorders such as hypotension, tachycardia, angina-like pains; pulmonary disorders such as pulmonary congestion; and other general reactions such as blurred vision, convulsions, nausea, vomiting, diarrhea, rhinitis, chills, vertigo, backache and urticaria. Allergic reactions from sorbitol-mannitol have also been reported.
Should any adverse reaction occur, discontinue the irrigant, evaluate the patient, institute appropriate therapeutic countermeasures and save the remainder of the fluid for examination if deemed necessary.
-
OVERDOSAGE
In the event of dehydration, fluid or solute overload, discontinue the irrigation, evaluate the patient and institute corrective measures as indicated. (See WARNINGS, PRECAUTIONS and ADVERSE REACTIONS.)
-
DOSAGE AND ADMINISTRATION
Sorbitol-Mannitol Irrigation should be administered only by transurethral instillation with appropriate urologic instrumentation. A disposable administration set should be used. The total volume of solution used for irrigation is solely at the discretion of the surgeon.
Height of container(s) above the operating table in excess of 60 cm (approx. 2 ft) has been reported to increase intravascular absorption of the irrigating fluid.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever container and solution permit. (See PRECAUTIONS.)
-
HOW SUPPLIED
Sorbitol-Mannitol Irrigation is supplied in single-dose 3000 mL flexible irrigation container
(NDC No. 0409-7981-08 / 0990-7981-08).ICU Medical is transitioning NDC codes from the "0409" to a "0990" labeler code. Both NDC codes are expected to be in the market for a period of time.
Store at 20 to 25ºC (68 to 77ºF). [See USP Controlled Room Temperature.] Protect from freezing.
Revised: July, 2018
EN-4664
ICU Medical, Inc., Lake Forest, Illinois, 60045, USA
-
PRINCIPAL DISPLAY PANEL - 3000 mL Bag Label
3000 mL
NDC 0990-7981-08SORBITOL-
MANNITOL
IRRIGATIONEACH 100 mL CONTAINS
SORBITOL 2.70 g; MANNITOL
0.54 g. pH 5.2 (4.0 TO 7.0)
178 mOsmol/LITER (CALC.)
STERILE, NONPYROGENIC.INDICATIONS:
FOR UROLOGIC IRRIGATION.CONTRAINDICATIONS:
NOT FOR INJECTION. USE ONLY
IF SOLUTION IS CLEAR AND
CONTAINER IS UNDAMAGED.WARNINGS:
DO NOT HEAT OVER 66°C (150°F)
OR STORE ABOVE 40°C (104°F).
SINGLE-DOSE CONTAINER.
CONTAINS NO BACTERIOSTAT.
DISCARD UNUSED PORTION.
USE ASEPTIC TECHNIQUE.USUAL DOSAGE:
SEE INSERT.RX ONLY
IM-4383
3
V
CONTAINS DEHPICU Medical, Inc.,
Lake Forest, Illinois, 60045, USAicumedical

-
PRINCIPAL DISPLAY PANEL - Overwrap Label
2
HDPETO OPEN TEAR AT NOTCH
DO NOT REMOVE FROM OVERWRAP UNTIL READY FOR USE. AFTER REMOVING
THE OVERWRAP, CHECK FOR MINUTE LEAKS BY SQUEEZING CONTAINER FIRMLY.
IF LEAKS ARE FOUND, DISCARD SOLUTION AS STERILITY MAY BE IMPAIRED.
RECOMMENDED STORAGE: ROOM TEMPERATURE (25°C). AVOID EXCESSIVE
HEAT. PROTECT FROM FREEZING. SEE INSERT.
98-4321-R14-3/98
-
INGREDIENTS AND APPEARANCE
SORBITOL-MANNITOL
sorbitol and mannitol irrigantProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0990-7981 Route of Administration URETHRAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SORBITOL (UNII: 506T60A25R) (SORBITOL - UNII:506T60A25R) SORBITOL 2.7 g in 100 mL MANNITOL (UNII: 3OWL53L36A) (MANNITOL - UNII:3OWL53L36A) MANNITOL 0.54 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0990-7981-08 4 in 1 CASE 10/01/2019 1 1 in 1 POUCH 1 3000 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA018316 10/01/2019 Labeler - ICU Medical Inc. (118380146) Establishment Name Address ID/FEI Business Operations Hospira, Inc. 827731089 ANALYSIS(0990-7981)


