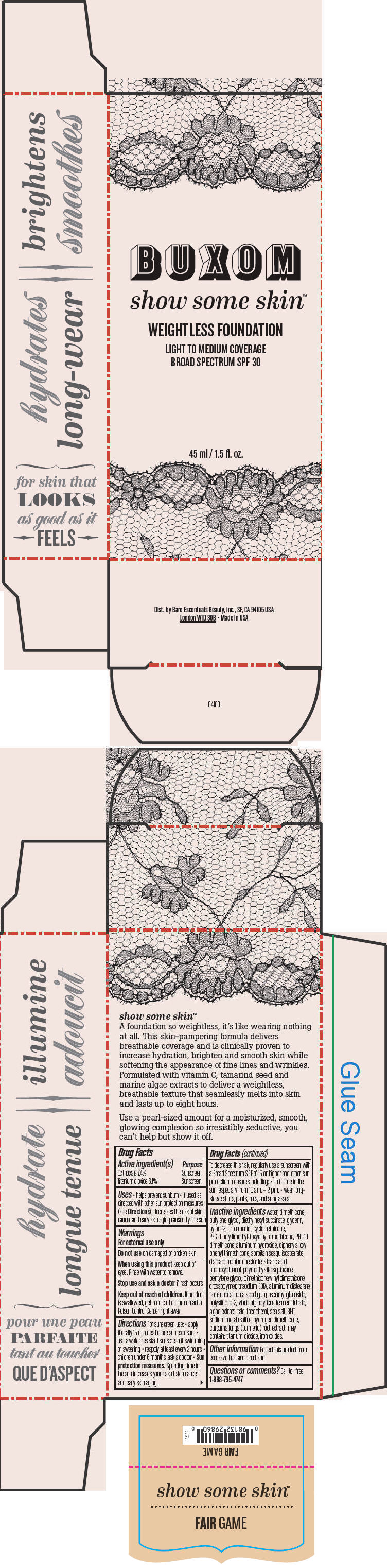
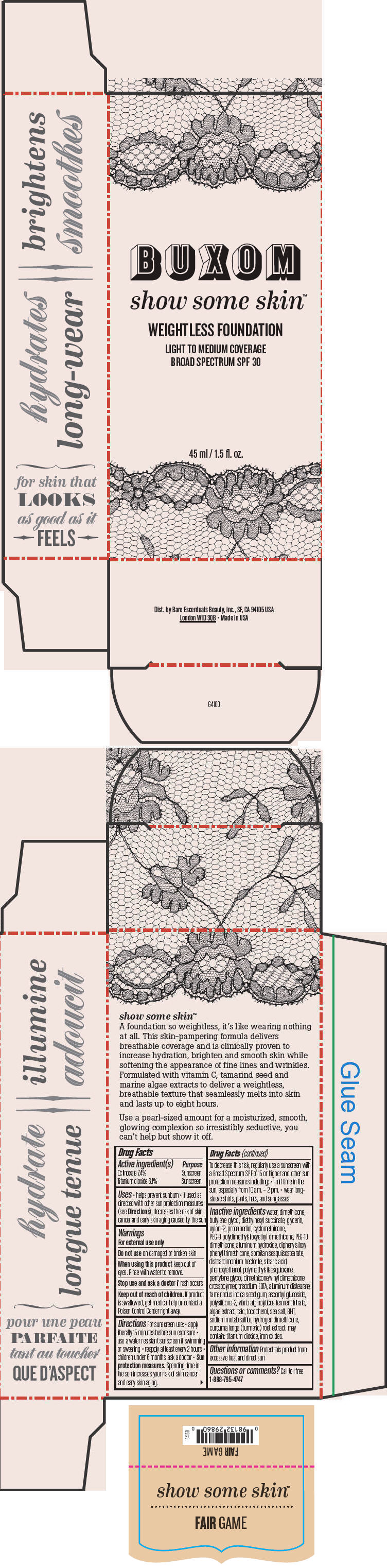
Label: BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 FAIR GAME- octinoxate and titanium dioxide liquid
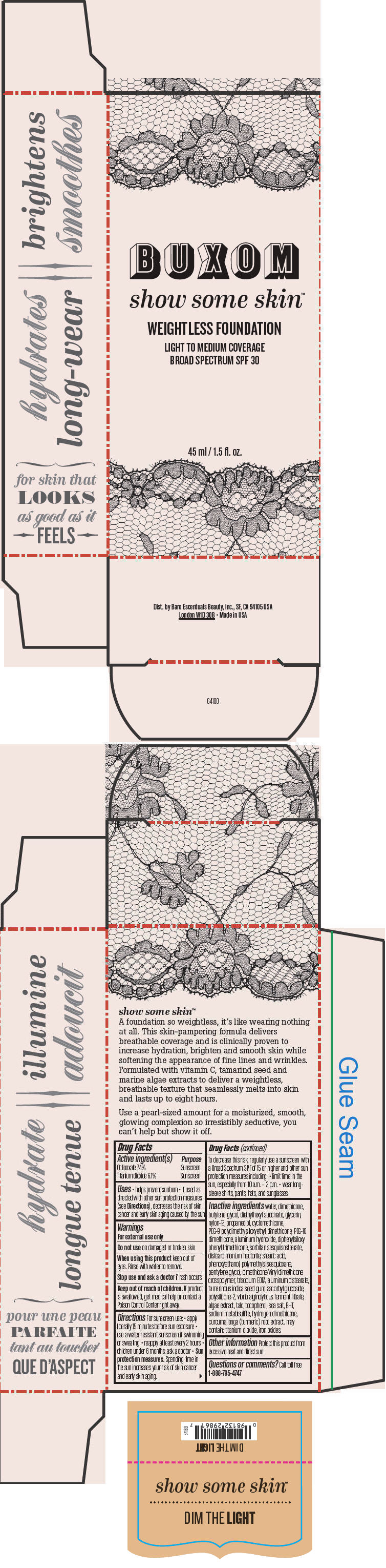
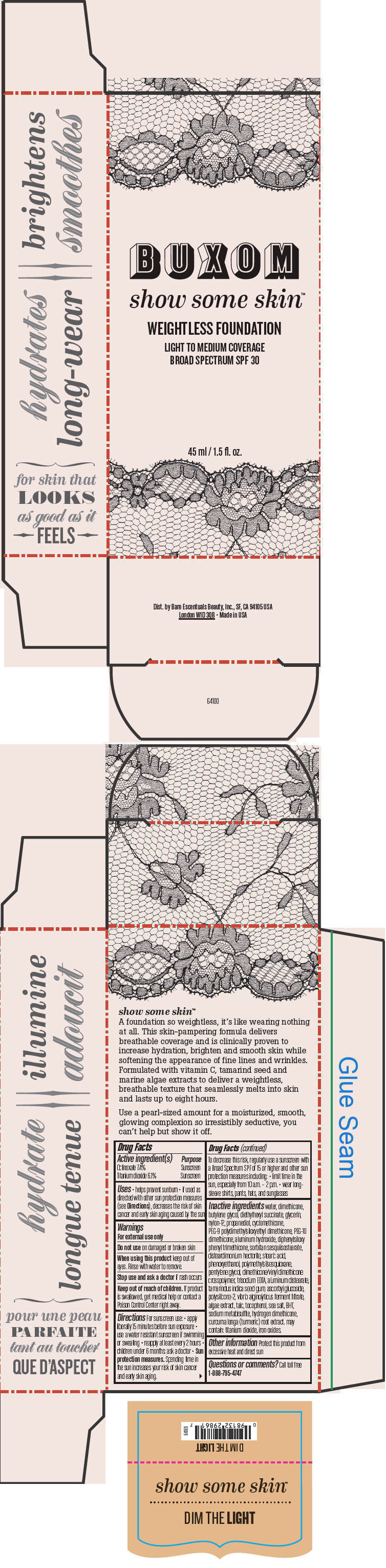
BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 DIM THE LIGHT- octinoxate and titanium dioxide liquid
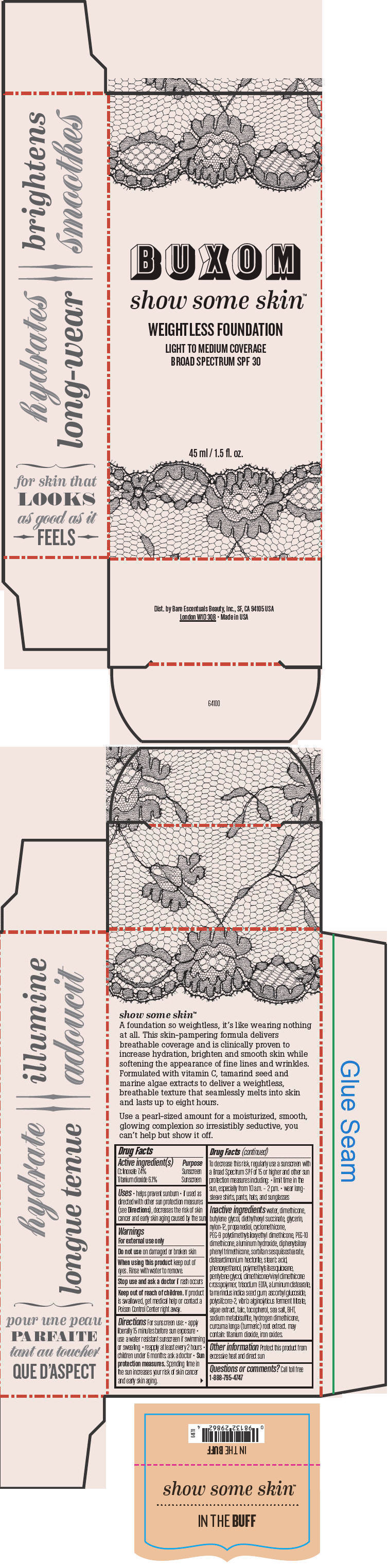
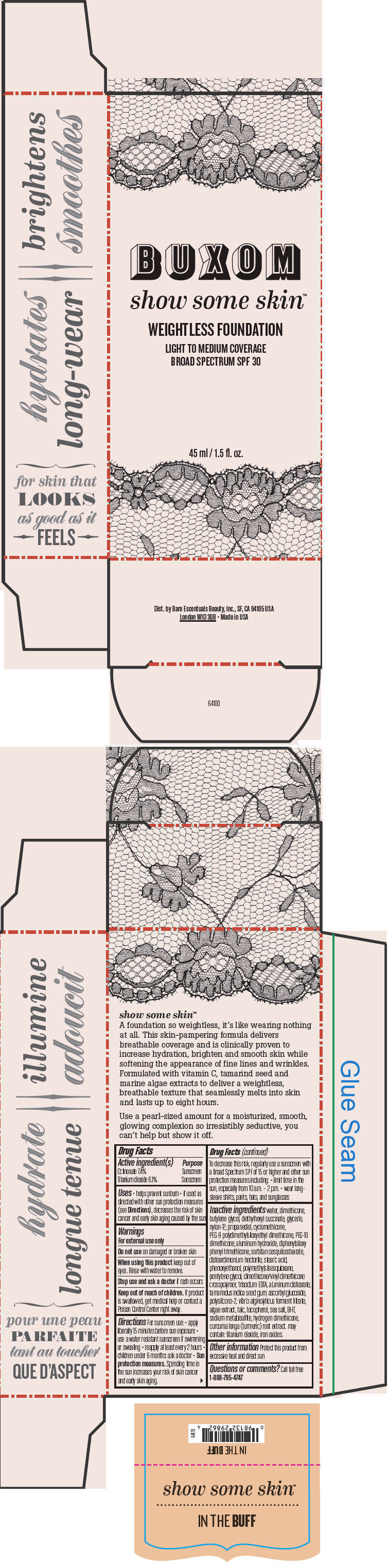
BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 IN THE BUFF- octinoxate and titanium dioxide liquid
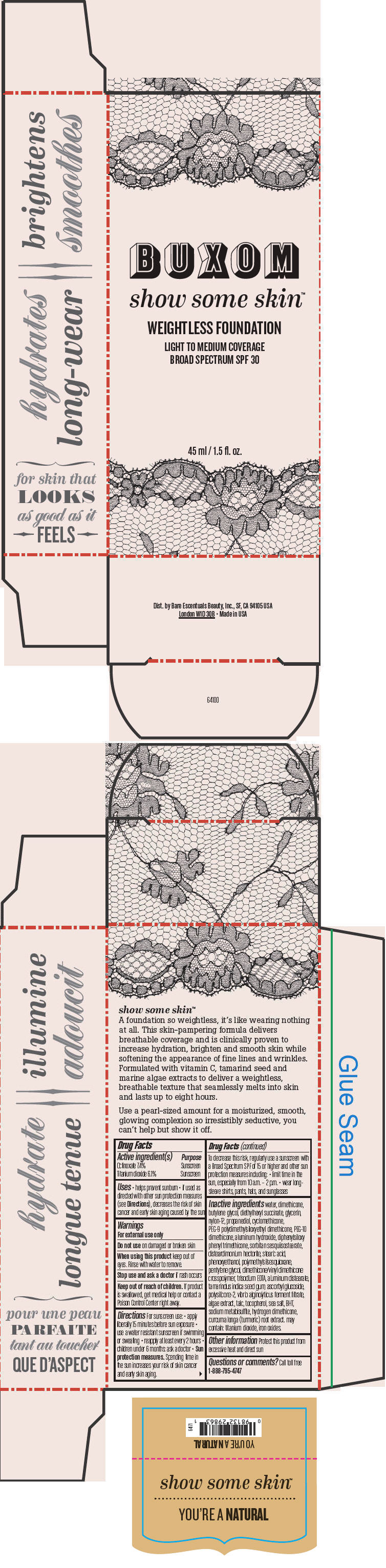
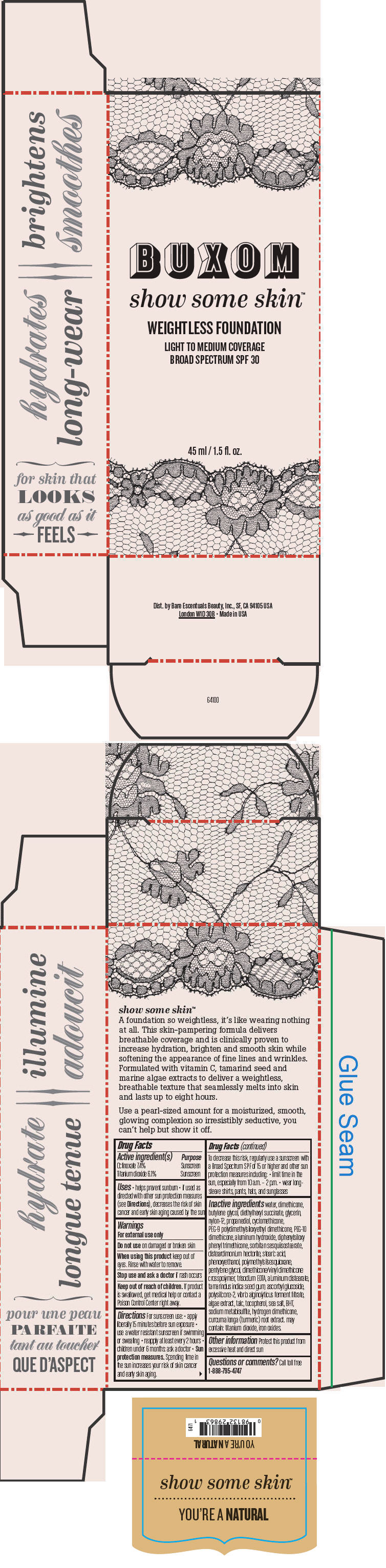
BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 YOURE A NATURAL- octinoxate and titanium dioxide liquid
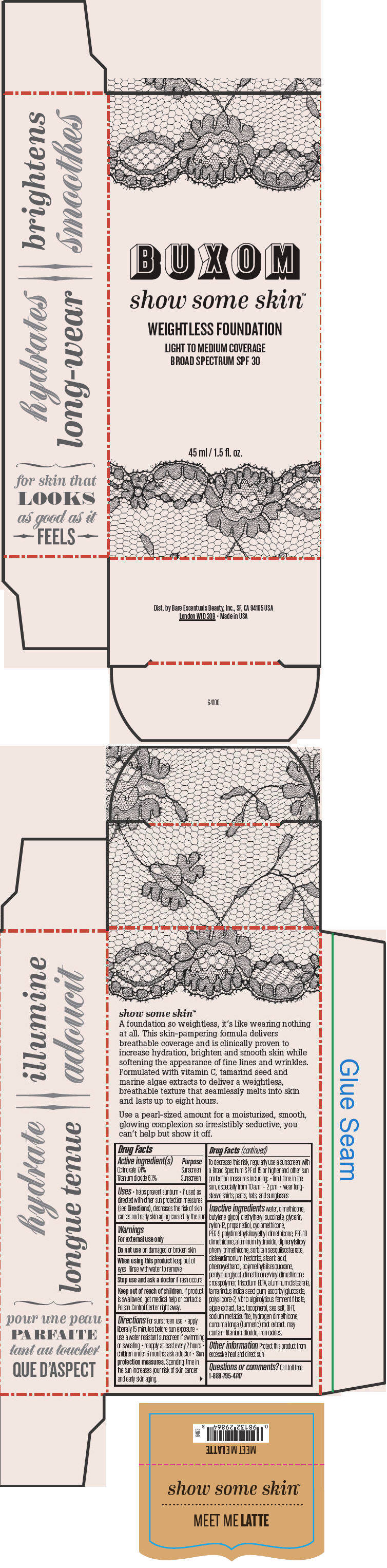
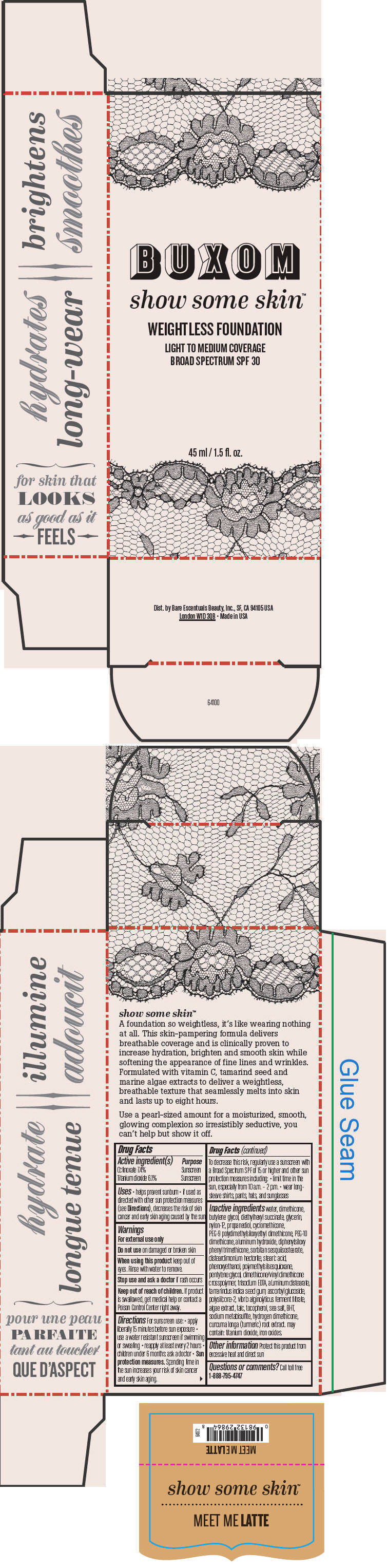
BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 MEET ME LATTE- octinoxate and titanium dioxide liquid
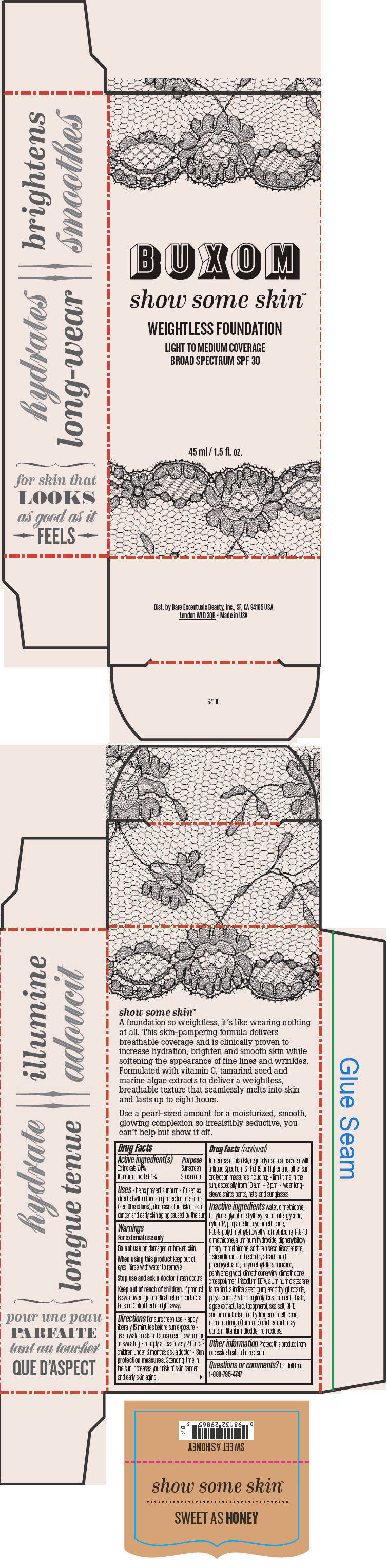
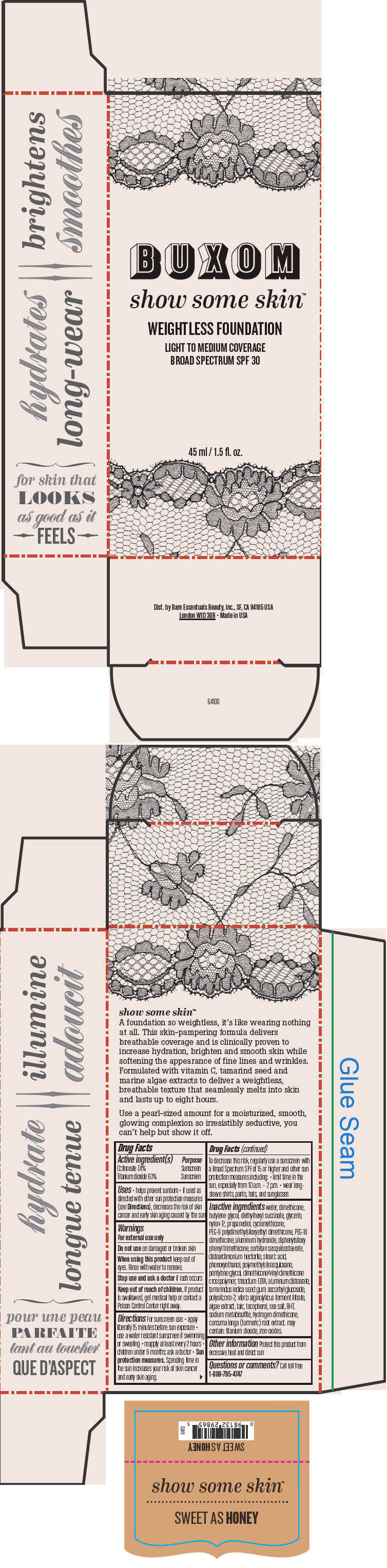
BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 SWEET AS HONEY- octinoxate and titanium dioxide liquid
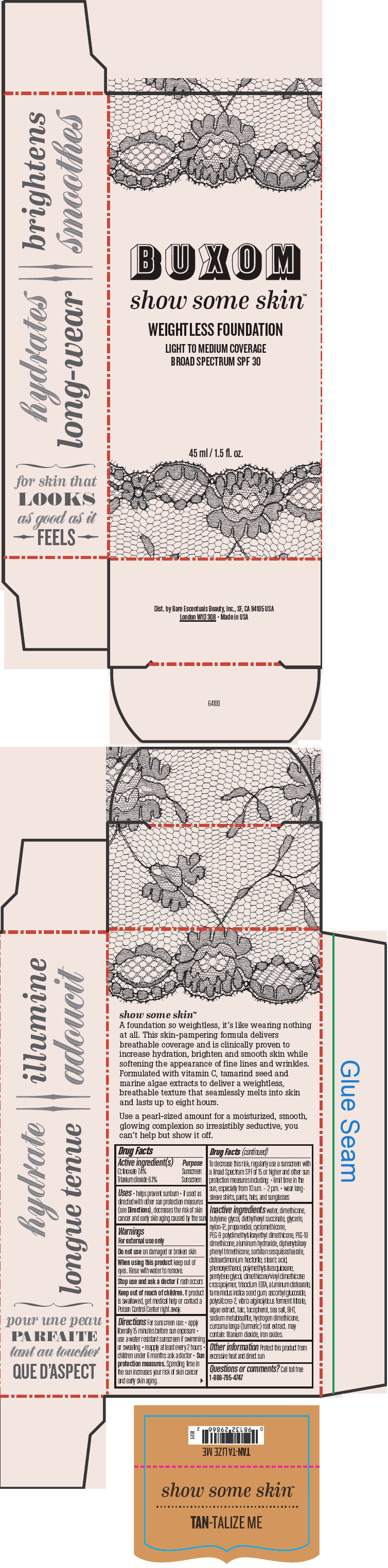
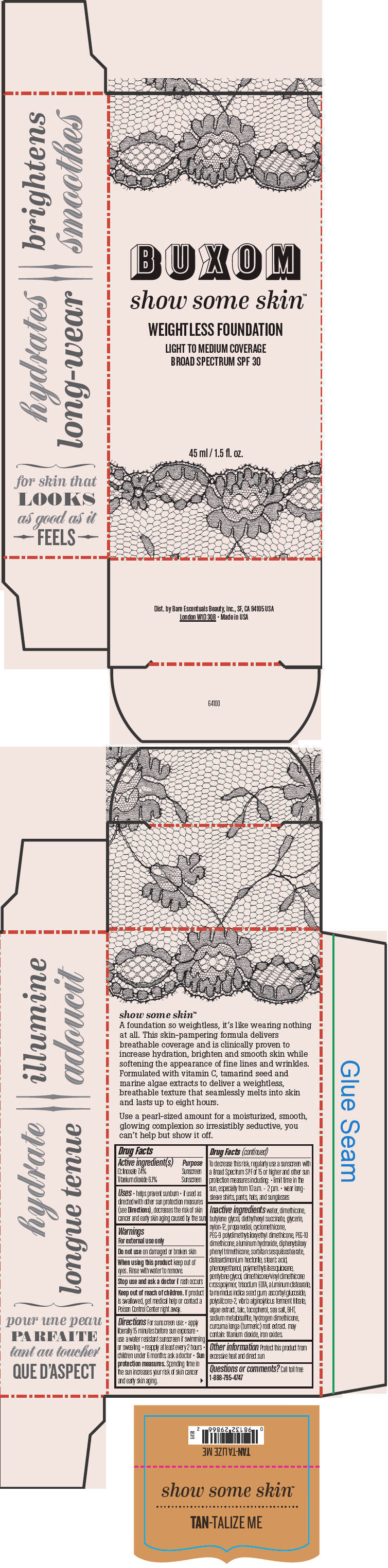
BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 TAN-TALIZE ME- octinoxate and titanium dioxide liquid
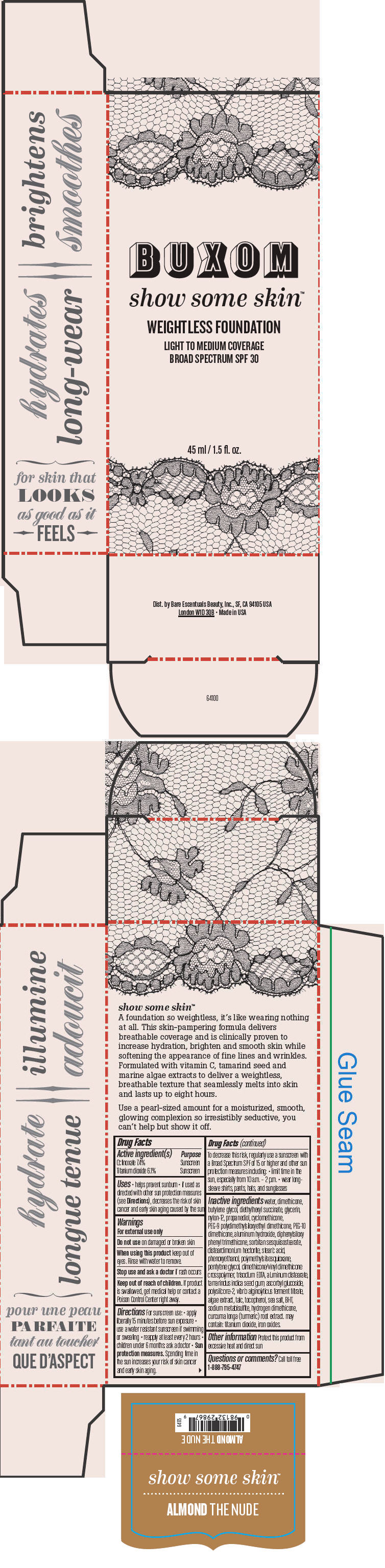
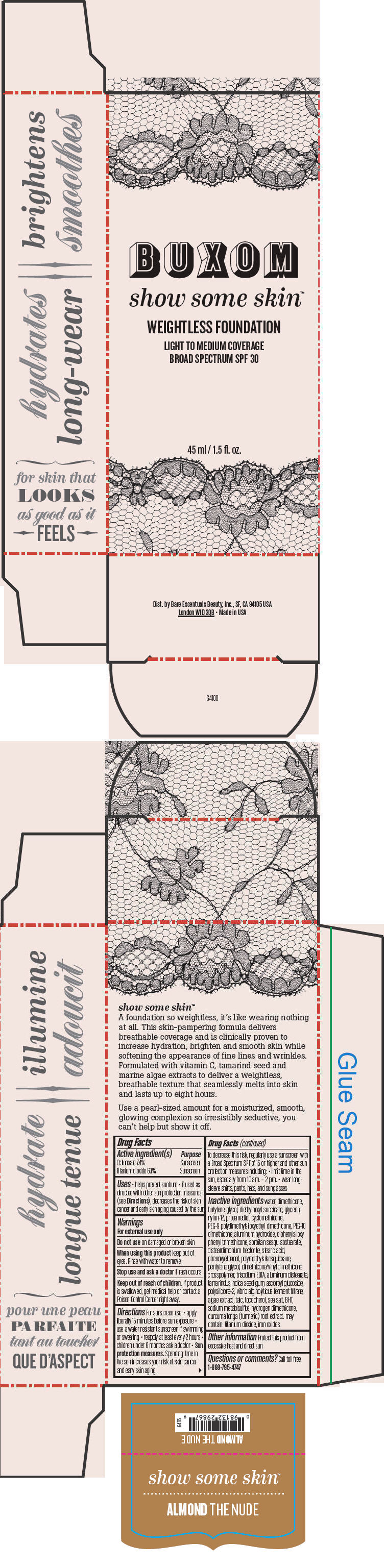
BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 ALMOND THE NUDE- octinoxate and titanium dioxide liquid
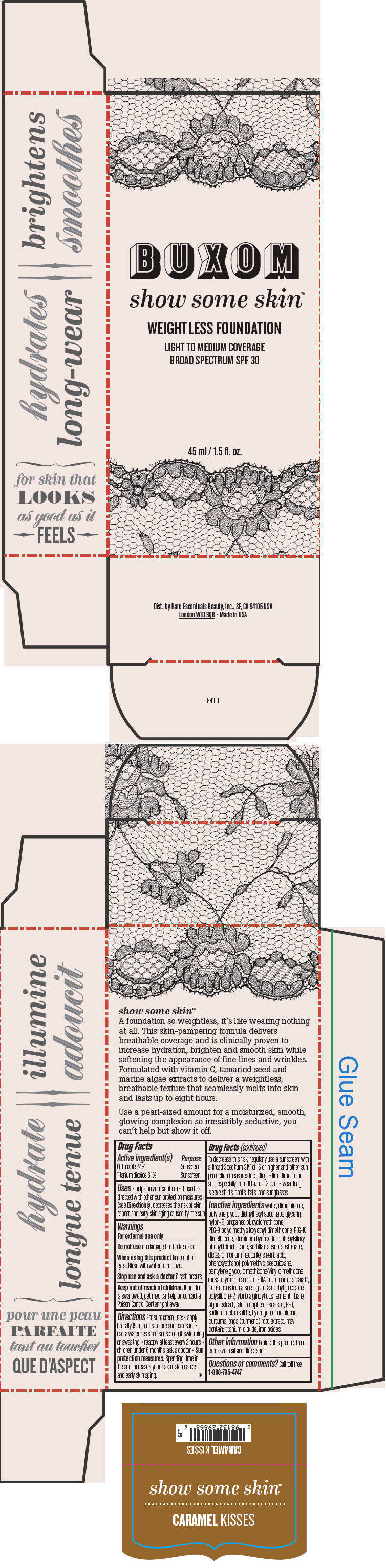
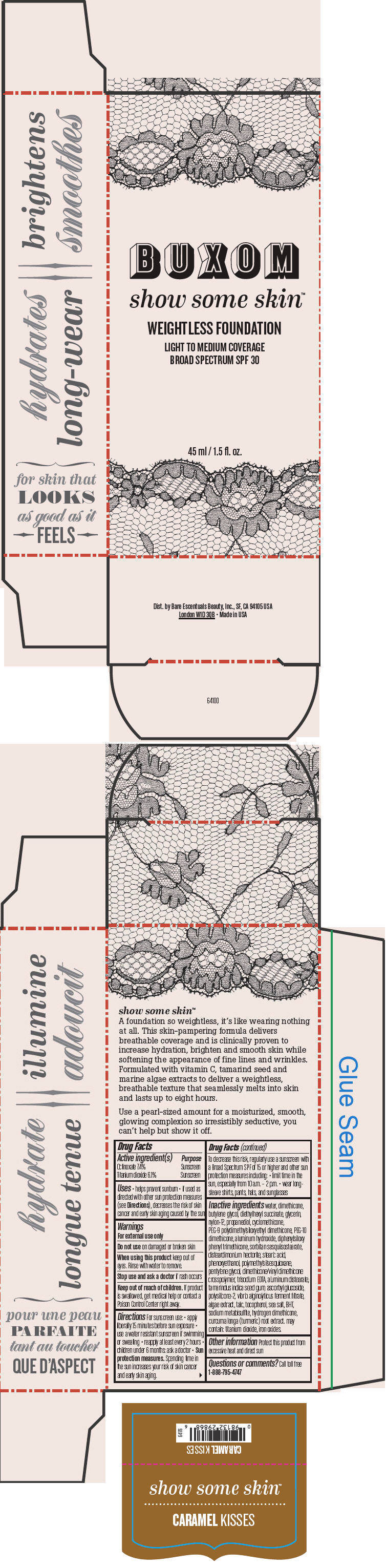
BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 CARAMEL KISSES- octinoxate and titanium dioxide liquid
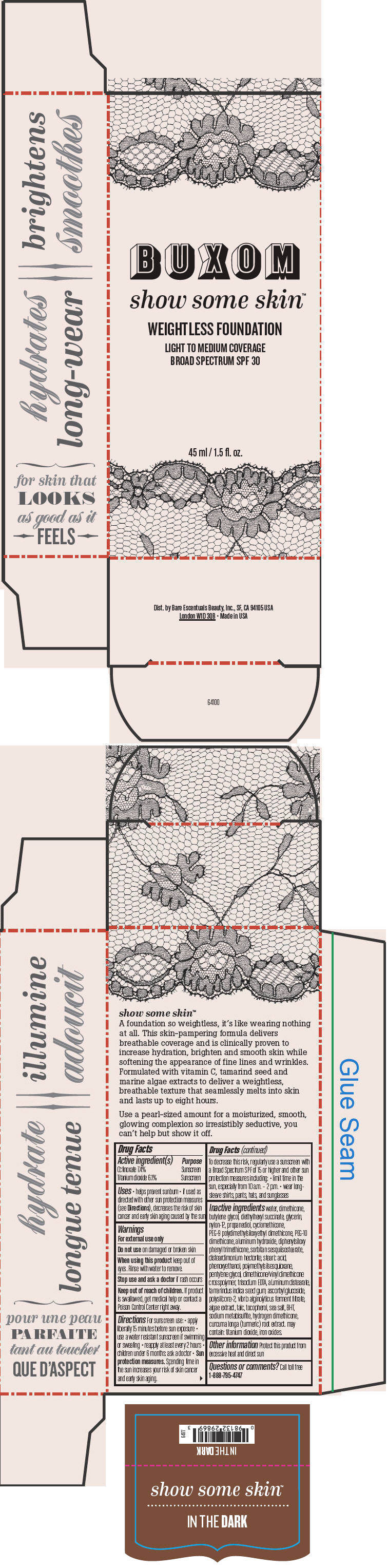
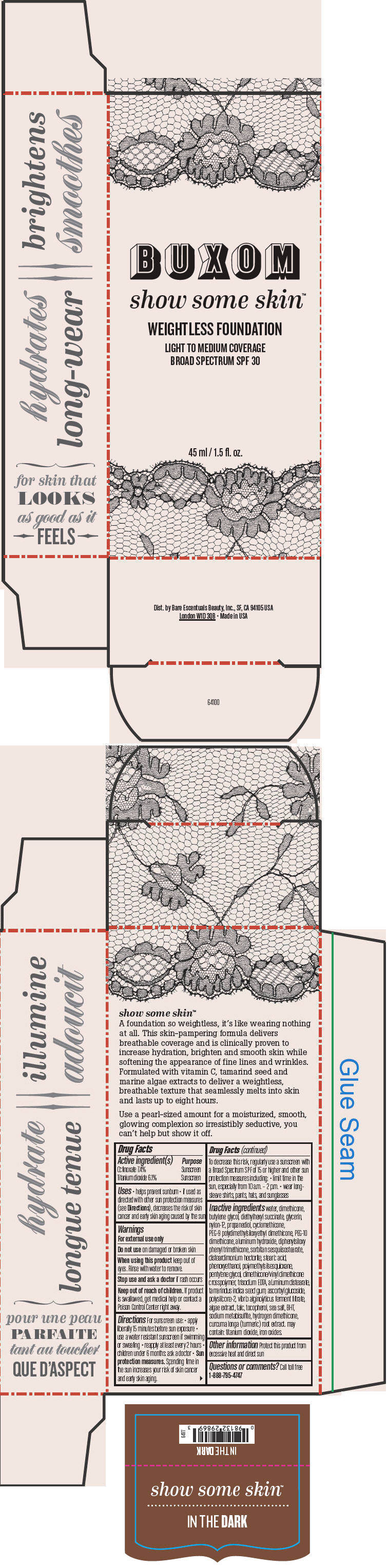
BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 IN THE DARK- octinoxate and titanium dioxide liquid
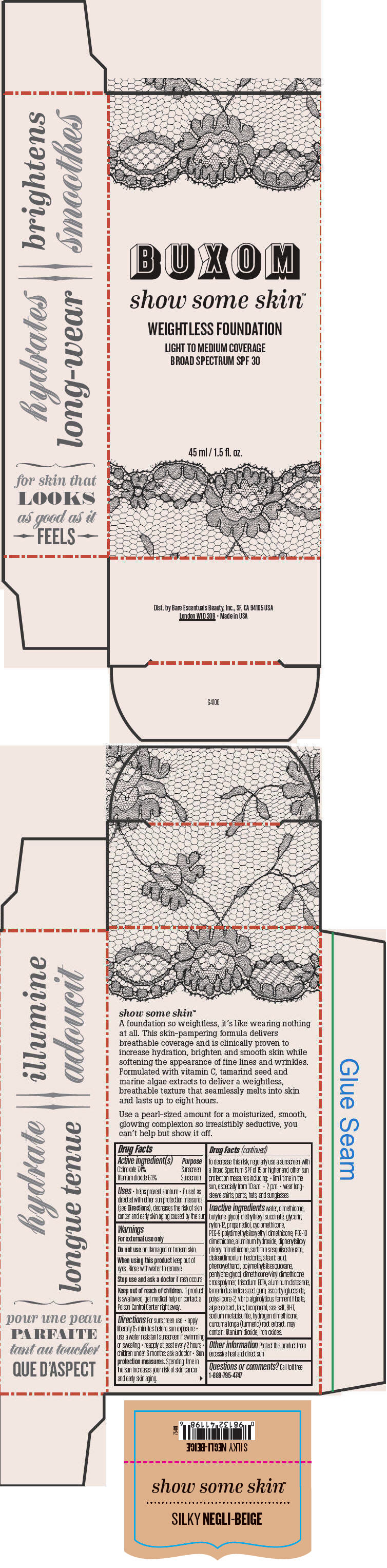
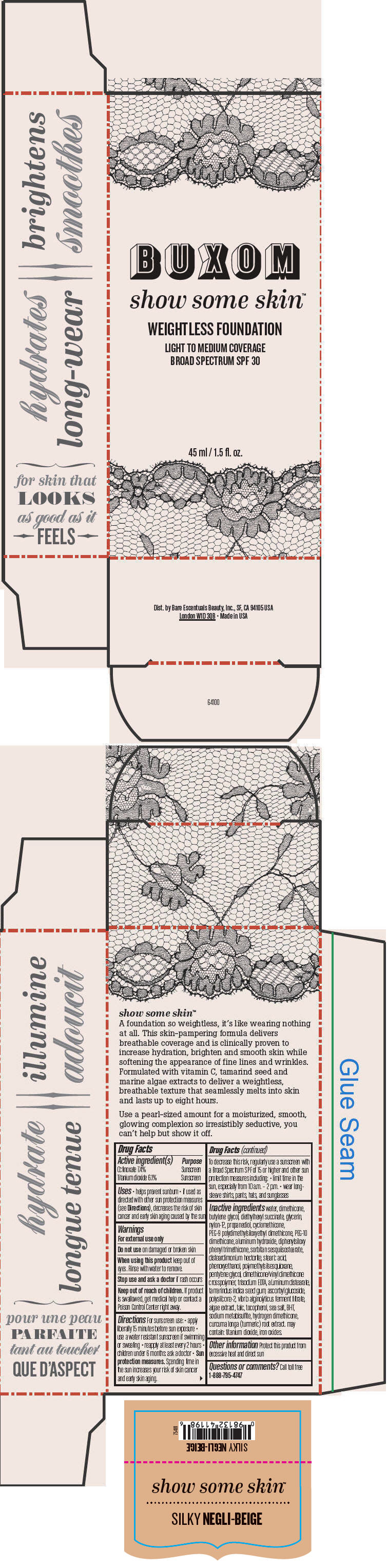
BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 SILKY NEGLI-BEIGE- octinoxate and titanium dioxide liquid
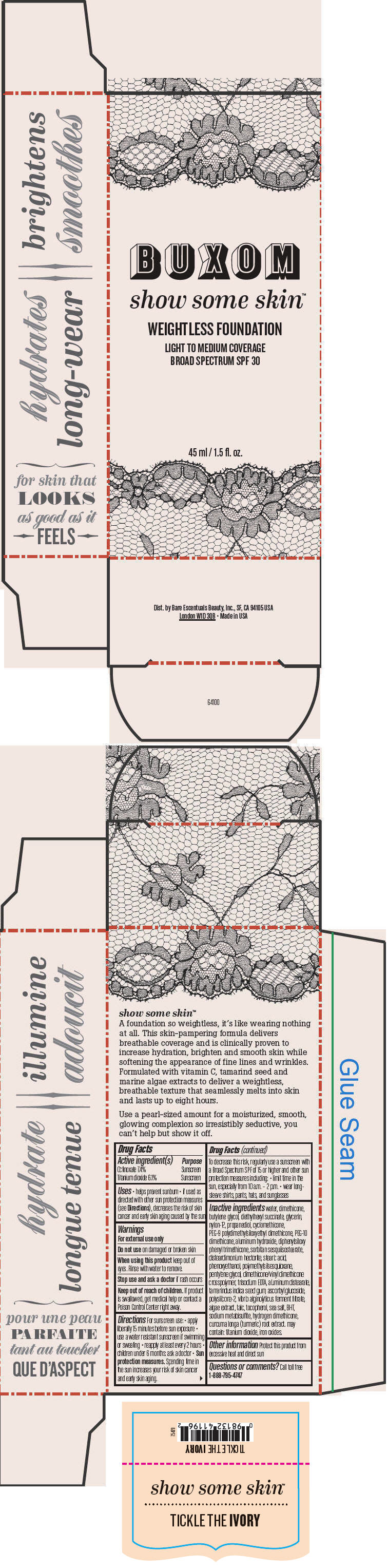
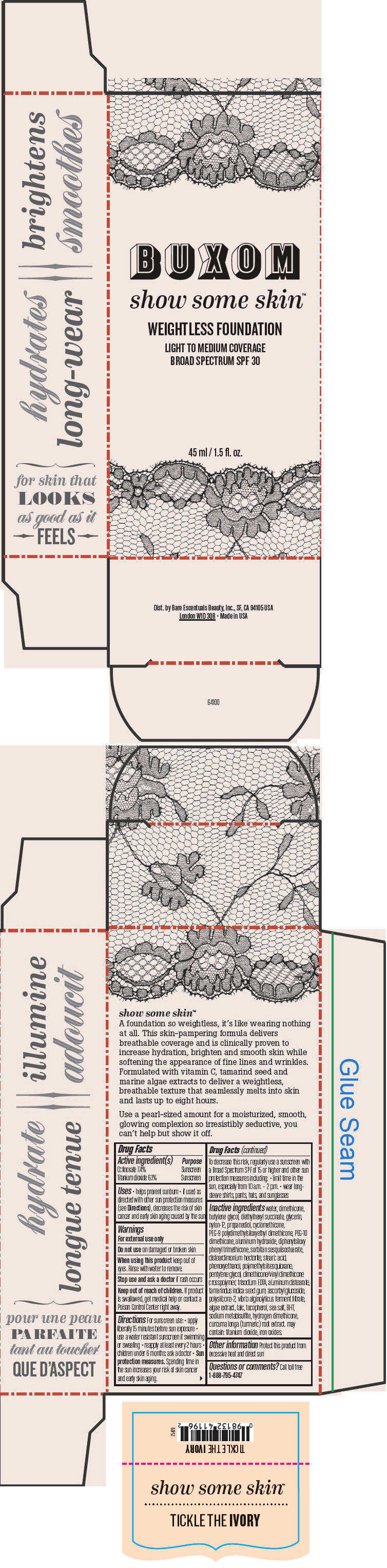
BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 TICKLE THE IVORY- octinoxate and titanium dioxide liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 98132-710-01, 98132-711-01, 98132-712-01, 98132-713-01, view more98132-714-01, 98132-715-01, 98132-716-01, 98132-717-01, 98132-718-01, 98132-719-01, 98132-762-01, 98132-763-01 - Packager: Bare Escentuals Beauty, Inc.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated June 5, 2015
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
-
Uses
- helps prevent sunburn
- if used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun
- Warnings
-
Directions
For sunscreen use:
- apply liberally 15 minutes before sun exposure
- use a water resistant sunscreen if swimming or sweating
- reapply at least every 2 hours
- children under 6 months: ask a doctor
- Sun protection measures. Spending time in the sun increases your risk of skin cancer and early skin aging.
To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. – 2 p.m.
- wear long-sleeve shirts, pants, hats, and sunglasses
-
Inactive ingredients
water, dimethicone, butylene glycol, diethylhexyl succinate, glycerin, nylon-12, propanediol, cyclomethicone, PEG-9 polydimethylsiloxyethyl dimethicone, PEG-10 dimethicone, aluminum hydroxide, diphenylsiloxy phenyl trimethicone, sorbitan sesquiisostearate, disteardimonium hectorite, stearic acid, phenoxyethanol, polymethylsilsesquioxane, pentylene glycol, dimethicone/vinyl dimethicone crosspolymer, trisodium EDTA, aluminum distearate, tamarindus indica seed gum, ascorbyl glucoside, polysilicone-2, vibrio alginolyticus ferment filtrate, algae extract, talc, tocopherol, sea salt, BHT, sodium metabisulfite, hydrogen dimethicone, curcuma longa (turmeric) root extract. may contain: titanium dioxide, iron oxides.
- Other information
- Questions or comments?
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 45 ml Tube Box - Fair Game
- PRINCIPAL DISPLAY PANEL - 45 ml Tube Box - Dim The Light
- PRINCIPAL DISPLAY PANEL - 45 ml Tube Box - In The Buff
- PRINCIPAL DISPLAY PANEL - 45 ml Tube Box - You're A Natural
- PRINCIPAL DISPLAY PANEL - 45 ml Tube Box - Meet Me Latte
- PRINCIPAL DISPLAY PANEL - 45 ml Tube Box - Sweet As Honey
- PRINCIPAL DISPLAY PANEL - 45 ml Tube Box - Tan-Talize Me
- PRINCIPAL DISPLAY PANEL - 45 ml Tube Box - Almond The Nude
- PRINCIPAL DISPLAY PANEL - 45 ml Tube Box - Caramel Kisses
- PRINCIPAL DISPLAY PANEL - 45 ml Tube Box - In The Dark
- PRINCIPAL DISPLAY PANEL - 45 ml Tube Box - Silky Negli-Beige
- PRINCIPAL DISPLAY PANEL - 45 ml Tube Box - Tickle The Ivory
-
INGREDIENTS AND APPEARANCE
BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 FAIR GAME
octinoxate and titanium dioxide liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:98132-710 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Octinoxate (UNII: 4Y5P7MUD51) (Octinoxate - UNII:4Y5P7MUD51) Octinoxate 74 mg in 1 mL Titanium Dioxide (UNII: 15FIX9V2JP) (Titanium Dioxide - UNII:15FIX9V2JP) Titanium Dioxide 61 mg in 1 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) dimethicone (UNII: 92RU3N3Y1O) butylene glycol (UNII: 3XUS85K0RA) diethylhexyl succinate (UNII: 69W9UMG3P8) glycerin (UNII: PDC6A3C0OX) nylon-12 (UNII: 446U8J075B) propanediol (UNII: 5965N8W85T) cyclomethicone (UNII: NMQ347994Z) PEG-9 polydimethylsiloxyethyl dimethicone (UNII: TYP81E471F) aluminum hydroxide (UNII: 5QB0T2IUN0) diphenylsiloxy phenyl trimethicone (UNII: I445L28B12) disteardimonium hectorite (UNII: X687XDK09L) stearic acid (UNII: 4ELV7Z65AP) phenoxyethanol (UNII: HIE492ZZ3T) pentylene glycol (UNII: 50C1307PZG) edetate trisodium (UNII: 420IP921MB) aluminum distearate (UNII: 7P1HP1B9UI) tamarind seed (UNII: 6AHP8A7OML) ascorbyl glucoside (UNII: 2V52R0NHXW) talc (UNII: 7SEV7J4R1U) tocopherol (UNII: R0ZB2556P8) sea salt (UNII: 87GE52P74G) butylated hydroxytoluene (UNII: 1P9D0Z171K) sodium metabisulfite (UNII: 4VON5FNS3C) turmeric (UNII: 856YO1Z64F) ferric oxide red (UNII: 1K09F3G675) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:98132-710-01 1 in 1 BOX 1 45 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part352 01/30/2014 BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 DIM THE LIGHT
octinoxate and titanium dioxide liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:98132-711 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Octinoxate (UNII: 4Y5P7MUD51) (Octinoxate - UNII:4Y5P7MUD51) Octinoxate 74 mg in 1 mL Titanium Dioxide (UNII: 15FIX9V2JP) (Titanium Dioxide - UNII:15FIX9V2JP) Titanium Dioxide 61 mg in 1 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) dimethicone (UNII: 92RU3N3Y1O) butylene glycol (UNII: 3XUS85K0RA) diethylhexyl succinate (UNII: 69W9UMG3P8) glycerin (UNII: PDC6A3C0OX) nylon-12 (UNII: 446U8J075B) propanediol (UNII: 5965N8W85T) cyclomethicone (UNII: NMQ347994Z) PEG-9 polydimethylsiloxyethyl dimethicone (UNII: TYP81E471F) aluminum hydroxide (UNII: 5QB0T2IUN0) diphenylsiloxy phenyl trimethicone (UNII: I445L28B12) disteardimonium hectorite (UNII: X687XDK09L) stearic acid (UNII: 4ELV7Z65AP) phenoxyethanol (UNII: HIE492ZZ3T) pentylene glycol (UNII: 50C1307PZG) edetate trisodium (UNII: 420IP921MB) aluminum distearate (UNII: 7P1HP1B9UI) tamarind seed (UNII: 6AHP8A7OML) ascorbyl glucoside (UNII: 2V52R0NHXW) talc (UNII: 7SEV7J4R1U) tocopherol (UNII: R0ZB2556P8) sea salt (UNII: 87GE52P74G) butylated hydroxytoluene (UNII: 1P9D0Z171K) sodium metabisulfite (UNII: 4VON5FNS3C) turmeric (UNII: 856YO1Z64F) ferric oxide red (UNII: 1K09F3G675) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:98132-711-01 1 in 1 BOX 1 45 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part352 01/30/2014 BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 IN THE BUFF
octinoxate and titanium dioxide liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:98132-712 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Octinoxate (UNII: 4Y5P7MUD51) (Octinoxate - UNII:4Y5P7MUD51) Octinoxate 74 mg in 1 mL Titanium Dioxide (UNII: 15FIX9V2JP) (Titanium Dioxide - UNII:15FIX9V2JP) Titanium Dioxide 61 mg in 1 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) dimethicone (UNII: 92RU3N3Y1O) butylene glycol (UNII: 3XUS85K0RA) diethylhexyl succinate (UNII: 69W9UMG3P8) glycerin (UNII: PDC6A3C0OX) nylon-12 (UNII: 446U8J075B) propanediol (UNII: 5965N8W85T) cyclomethicone (UNII: NMQ347994Z) PEG-9 polydimethylsiloxyethyl dimethicone (UNII: TYP81E471F) aluminum hydroxide (UNII: 5QB0T2IUN0) diphenylsiloxy phenyl trimethicone (UNII: I445L28B12) disteardimonium hectorite (UNII: X687XDK09L) stearic acid (UNII: 4ELV7Z65AP) phenoxyethanol (UNII: HIE492ZZ3T) pentylene glycol (UNII: 50C1307PZG) edetate trisodium (UNII: 420IP921MB) aluminum distearate (UNII: 7P1HP1B9UI) tamarind seed (UNII: 6AHP8A7OML) ascorbyl glucoside (UNII: 2V52R0NHXW) talc (UNII: 7SEV7J4R1U) tocopherol (UNII: R0ZB2556P8) sea salt (UNII: 87GE52P74G) butylated hydroxytoluene (UNII: 1P9D0Z171K) sodium metabisulfite (UNII: 4VON5FNS3C) turmeric (UNII: 856YO1Z64F) ferric oxide red (UNII: 1K09F3G675) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:98132-712-01 1 in 1 BOX 1 45 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part352 01/30/2014 BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 YOURE A NATURAL
octinoxate and titanium dioxide liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:98132-713 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Octinoxate (UNII: 4Y5P7MUD51) (Octinoxate - UNII:4Y5P7MUD51) Octinoxate 74 mg in 1 mL Titanium Dioxide (UNII: 15FIX9V2JP) (Titanium Dioxide - UNII:15FIX9V2JP) Titanium Dioxide 61 mg in 1 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) dimethicone (UNII: 92RU3N3Y1O) butylene glycol (UNII: 3XUS85K0RA) diethylhexyl succinate (UNII: 69W9UMG3P8) glycerin (UNII: PDC6A3C0OX) nylon-12 (UNII: 446U8J075B) propanediol (UNII: 5965N8W85T) cyclomethicone (UNII: NMQ347994Z) PEG-9 polydimethylsiloxyethyl dimethicone (UNII: TYP81E471F) aluminum hydroxide (UNII: 5QB0T2IUN0) diphenylsiloxy phenyl trimethicone (UNII: I445L28B12) disteardimonium hectorite (UNII: X687XDK09L) stearic acid (UNII: 4ELV7Z65AP) phenoxyethanol (UNII: HIE492ZZ3T) pentylene glycol (UNII: 50C1307PZG) edetate trisodium (UNII: 420IP921MB) aluminum distearate (UNII: 7P1HP1B9UI) tamarind seed (UNII: 6AHP8A7OML) ascorbyl glucoside (UNII: 2V52R0NHXW) talc (UNII: 7SEV7J4R1U) tocopherol (UNII: R0ZB2556P8) sea salt (UNII: 87GE52P74G) butylated hydroxytoluene (UNII: 1P9D0Z171K) sodium metabisulfite (UNII: 4VON5FNS3C) turmeric (UNII: 856YO1Z64F) ferric oxide red (UNII: 1K09F3G675) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:98132-713-01 1 in 1 BOX 1 45 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part352 01/30/2014 BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 MEET ME LATTE
octinoxate and titanium dioxide liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:98132-714 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Octinoxate (UNII: 4Y5P7MUD51) (Octinoxate - UNII:4Y5P7MUD51) Octinoxate 74 mg in 1 mL Titanium Dioxide (UNII: 15FIX9V2JP) (Titanium Dioxide - UNII:15FIX9V2JP) Titanium Dioxide 61 mg in 1 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) dimethicone (UNII: 92RU3N3Y1O) butylene glycol (UNII: 3XUS85K0RA) diethylhexyl succinate (UNII: 69W9UMG3P8) glycerin (UNII: PDC6A3C0OX) nylon-12 (UNII: 446U8J075B) propanediol (UNII: 5965N8W85T) cyclomethicone (UNII: NMQ347994Z) PEG-9 polydimethylsiloxyethyl dimethicone (UNII: TYP81E471F) aluminum hydroxide (UNII: 5QB0T2IUN0) diphenylsiloxy phenyl trimethicone (UNII: I445L28B12) disteardimonium hectorite (UNII: X687XDK09L) stearic acid (UNII: 4ELV7Z65AP) phenoxyethanol (UNII: HIE492ZZ3T) pentylene glycol (UNII: 50C1307PZG) edetate trisodium (UNII: 420IP921MB) aluminum distearate (UNII: 7P1HP1B9UI) tamarind seed (UNII: 6AHP8A7OML) ascorbyl glucoside (UNII: 2V52R0NHXW) talc (UNII: 7SEV7J4R1U) tocopherol (UNII: R0ZB2556P8) sea salt (UNII: 87GE52P74G) butylated hydroxytoluene (UNII: 1P9D0Z171K) sodium metabisulfite (UNII: 4VON5FNS3C) turmeric (UNII: 856YO1Z64F) ferric oxide red (UNII: 1K09F3G675) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:98132-714-01 1 in 1 BOX 1 45 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part352 01/30/2014 BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 SWEET AS HONEY
octinoxate and titanium dioxide liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:98132-715 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Octinoxate (UNII: 4Y5P7MUD51) (Octinoxate - UNII:4Y5P7MUD51) Octinoxate 74 mg in 1 mL Titanium Dioxide (UNII: 15FIX9V2JP) (Titanium Dioxide - UNII:15FIX9V2JP) Titanium Dioxide 61 mg in 1 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) dimethicone (UNII: 92RU3N3Y1O) butylene glycol (UNII: 3XUS85K0RA) diethylhexyl succinate (UNII: 69W9UMG3P8) glycerin (UNII: PDC6A3C0OX) nylon-12 (UNII: 446U8J075B) propanediol (UNII: 5965N8W85T) cyclomethicone (UNII: NMQ347994Z) PEG-9 polydimethylsiloxyethyl dimethicone (UNII: TYP81E471F) aluminum hydroxide (UNII: 5QB0T2IUN0) diphenylsiloxy phenyl trimethicone (UNII: I445L28B12) disteardimonium hectorite (UNII: X687XDK09L) stearic acid (UNII: 4ELV7Z65AP) phenoxyethanol (UNII: HIE492ZZ3T) pentylene glycol (UNII: 50C1307PZG) edetate trisodium (UNII: 420IP921MB) aluminum distearate (UNII: 7P1HP1B9UI) tamarind seed (UNII: 6AHP8A7OML) ascorbyl glucoside (UNII: 2V52R0NHXW) talc (UNII: 7SEV7J4R1U) tocopherol (UNII: R0ZB2556P8) sea salt (UNII: 87GE52P74G) butylated hydroxytoluene (UNII: 1P9D0Z171K) sodium metabisulfite (UNII: 4VON5FNS3C) turmeric (UNII: 856YO1Z64F) ferric oxide red (UNII: 1K09F3G675) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:98132-715-01 1 in 1 BOX 1 45 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part352 01/30/2014 BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 TAN-TALIZE ME
octinoxate and titanium dioxide liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:98132-716 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Octinoxate (UNII: 4Y5P7MUD51) (Octinoxate - UNII:4Y5P7MUD51) Octinoxate 74 mg in 1 mL Titanium Dioxide (UNII: 15FIX9V2JP) (Titanium Dioxide - UNII:15FIX9V2JP) Titanium Dioxide 61 mg in 1 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) dimethicone (UNII: 92RU3N3Y1O) butylene glycol (UNII: 3XUS85K0RA) diethylhexyl succinate (UNII: 69W9UMG3P8) glycerin (UNII: PDC6A3C0OX) nylon-12 (UNII: 446U8J075B) propanediol (UNII: 5965N8W85T) cyclomethicone (UNII: NMQ347994Z) PEG-9 polydimethylsiloxyethyl dimethicone (UNII: TYP81E471F) aluminum hydroxide (UNII: 5QB0T2IUN0) diphenylsiloxy phenyl trimethicone (UNII: I445L28B12) disteardimonium hectorite (UNII: X687XDK09L) stearic acid (UNII: 4ELV7Z65AP) phenoxyethanol (UNII: HIE492ZZ3T) pentylene glycol (UNII: 50C1307PZG) edetate trisodium (UNII: 420IP921MB) aluminum distearate (UNII: 7P1HP1B9UI) tamarind seed (UNII: 6AHP8A7OML) ascorbyl glucoside (UNII: 2V52R0NHXW) talc (UNII: 7SEV7J4R1U) tocopherol (UNII: R0ZB2556P8) sea salt (UNII: 87GE52P74G) butylated hydroxytoluene (UNII: 1P9D0Z171K) sodium metabisulfite (UNII: 4VON5FNS3C) turmeric (UNII: 856YO1Z64F) ferric oxide red (UNII: 1K09F3G675) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:98132-716-01 1 in 1 BOX 1 45 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part352 01/30/2014 BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 ALMOND THE NUDE
octinoxate and titanium dioxide liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:98132-717 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Octinoxate (UNII: 4Y5P7MUD51) (Octinoxate - UNII:4Y5P7MUD51) Octinoxate 74 mg in 1 mL Titanium Dioxide (UNII: 15FIX9V2JP) (Titanium Dioxide - UNII:15FIX9V2JP) Titanium Dioxide 61 mg in 1 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) dimethicone (UNII: 92RU3N3Y1O) butylene glycol (UNII: 3XUS85K0RA) diethylhexyl succinate (UNII: 69W9UMG3P8) glycerin (UNII: PDC6A3C0OX) nylon-12 (UNII: 446U8J075B) propanediol (UNII: 5965N8W85T) cyclomethicone (UNII: NMQ347994Z) PEG-9 polydimethylsiloxyethyl dimethicone (UNII: TYP81E471F) aluminum hydroxide (UNII: 5QB0T2IUN0) diphenylsiloxy phenyl trimethicone (UNII: I445L28B12) disteardimonium hectorite (UNII: X687XDK09L) stearic acid (UNII: 4ELV7Z65AP) phenoxyethanol (UNII: HIE492ZZ3T) pentylene glycol (UNII: 50C1307PZG) edetate trisodium (UNII: 420IP921MB) aluminum distearate (UNII: 7P1HP1B9UI) tamarind seed (UNII: 6AHP8A7OML) ascorbyl glucoside (UNII: 2V52R0NHXW) talc (UNII: 7SEV7J4R1U) tocopherol (UNII: R0ZB2556P8) sea salt (UNII: 87GE52P74G) butylated hydroxytoluene (UNII: 1P9D0Z171K) sodium metabisulfite (UNII: 4VON5FNS3C) turmeric (UNII: 856YO1Z64F) ferric oxide red (UNII: 1K09F3G675) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:98132-717-01 1 in 1 BOX 1 45 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part352 01/30/2014 BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 CARAMEL KISSES
octinoxate and titanium dioxide liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:98132-718 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Octinoxate (UNII: 4Y5P7MUD51) (Octinoxate - UNII:4Y5P7MUD51) Octinoxate 74 mg in 1 mL Titanium Dioxide (UNII: 15FIX9V2JP) (Titanium Dioxide - UNII:15FIX9V2JP) Titanium Dioxide 61 mg in 1 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) dimethicone (UNII: 92RU3N3Y1O) butylene glycol (UNII: 3XUS85K0RA) diethylhexyl succinate (UNII: 69W9UMG3P8) glycerin (UNII: PDC6A3C0OX) nylon-12 (UNII: 446U8J075B) propanediol (UNII: 5965N8W85T) cyclomethicone (UNII: NMQ347994Z) PEG-9 polydimethylsiloxyethyl dimethicone (UNII: TYP81E471F) aluminum hydroxide (UNII: 5QB0T2IUN0) diphenylsiloxy phenyl trimethicone (UNII: I445L28B12) disteardimonium hectorite (UNII: X687XDK09L) stearic acid (UNII: 4ELV7Z65AP) phenoxyethanol (UNII: HIE492ZZ3T) pentylene glycol (UNII: 50C1307PZG) edetate trisodium (UNII: 420IP921MB) aluminum distearate (UNII: 7P1HP1B9UI) tamarind seed (UNII: 6AHP8A7OML) ascorbyl glucoside (UNII: 2V52R0NHXW) talc (UNII: 7SEV7J4R1U) tocopherol (UNII: R0ZB2556P8) sea salt (UNII: 87GE52P74G) butylated hydroxytoluene (UNII: 1P9D0Z171K) sodium metabisulfite (UNII: 4VON5FNS3C) turmeric (UNII: 856YO1Z64F) ferric oxide red (UNII: 1K09F3G675) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:98132-718-01 1 in 1 BOX 1 45 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part352 01/30/2014 BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 IN THE DARK
octinoxate and titanium dioxide liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:98132-719 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Octinoxate (UNII: 4Y5P7MUD51) (Octinoxate - UNII:4Y5P7MUD51) Octinoxate 74 mg in 1 mL Titanium Dioxide (UNII: 15FIX9V2JP) (Titanium Dioxide - UNII:15FIX9V2JP) Titanium Dioxide 61 mg in 1 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) dimethicone (UNII: 92RU3N3Y1O) butylene glycol (UNII: 3XUS85K0RA) diethylhexyl succinate (UNII: 69W9UMG3P8) glycerin (UNII: PDC6A3C0OX) nylon-12 (UNII: 446U8J075B) propanediol (UNII: 5965N8W85T) cyclomethicone (UNII: NMQ347994Z) PEG-9 polydimethylsiloxyethyl dimethicone (UNII: TYP81E471F) aluminum hydroxide (UNII: 5QB0T2IUN0) diphenylsiloxy phenyl trimethicone (UNII: I445L28B12) disteardimonium hectorite (UNII: X687XDK09L) stearic acid (UNII: 4ELV7Z65AP) phenoxyethanol (UNII: HIE492ZZ3T) pentylene glycol (UNII: 50C1307PZG) edetate trisodium (UNII: 420IP921MB) aluminum distearate (UNII: 7P1HP1B9UI) tamarind seed (UNII: 6AHP8A7OML) ascorbyl glucoside (UNII: 2V52R0NHXW) talc (UNII: 7SEV7J4R1U) tocopherol (UNII: R0ZB2556P8) sea salt (UNII: 87GE52P74G) butylated hydroxytoluene (UNII: 1P9D0Z171K) sodium metabisulfite (UNII: 4VON5FNS3C) turmeric (UNII: 856YO1Z64F) ferric oxide red (UNII: 1K09F3G675) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:98132-719-01 1 in 1 BOX 1 45 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part352 01/30/2014 BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 SILKY NEGLI-BEIGE
octinoxate and titanium dioxide liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:98132-762 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Octinoxate (UNII: 4Y5P7MUD51) (Octinoxate - UNII:4Y5P7MUD51) Octinoxate 74 mg in 1 mL Titanium Dioxide (UNII: 15FIX9V2JP) (Titanium Dioxide - UNII:15FIX9V2JP) Titanium Dioxide 61 mg in 1 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) dimethicone (UNII: 92RU3N3Y1O) butylene glycol (UNII: 3XUS85K0RA) diethylhexyl succinate (UNII: 69W9UMG3P8) glycerin (UNII: PDC6A3C0OX) nylon-12 (UNII: 446U8J075B) propanediol (UNII: 5965N8W85T) cyclomethicone (UNII: NMQ347994Z) PEG-9 polydimethylsiloxyethyl dimethicone (UNII: TYP81E471F) aluminum hydroxide (UNII: 5QB0T2IUN0) diphenylsiloxy phenyl trimethicone (UNII: I445L28B12) disteardimonium hectorite (UNII: X687XDK09L) stearic acid (UNII: 4ELV7Z65AP) phenoxyethanol (UNII: HIE492ZZ3T) pentylene glycol (UNII: 50C1307PZG) edetate trisodium (UNII: 420IP921MB) aluminum distearate (UNII: 7P1HP1B9UI) tamarind seed (UNII: 6AHP8A7OML) ascorbyl glucoside (UNII: 2V52R0NHXW) talc (UNII: 7SEV7J4R1U) tocopherol (UNII: R0ZB2556P8) sea salt (UNII: 87GE52P74G) butylated hydroxytoluene (UNII: 1P9D0Z171K) sodium metabisulfite (UNII: 4VON5FNS3C) turmeric (UNII: 856YO1Z64F) ferric oxide red (UNII: 1K09F3G675) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:98132-762-01 1 in 1 BOX 1 45 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part352 06/01/2015 BUXOM SHOW SOME SKIN WEIGHTLESS FOUNDATION BROAD SPECTRUM SPF 30 TICKLE THE IVORY
octinoxate and titanium dioxide liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:98132-763 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Octinoxate (UNII: 4Y5P7MUD51) (Octinoxate - UNII:4Y5P7MUD51) Octinoxate 74 mg in 1 mL Titanium Dioxide (UNII: 15FIX9V2JP) (Titanium Dioxide - UNII:15FIX9V2JP) Titanium Dioxide 61 mg in 1 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) dimethicone (UNII: 92RU3N3Y1O) butylene glycol (UNII: 3XUS85K0RA) diethylhexyl succinate (UNII: 69W9UMG3P8) glycerin (UNII: PDC6A3C0OX) nylon-12 (UNII: 446U8J075B) propanediol (UNII: 5965N8W85T) cyclomethicone (UNII: NMQ347994Z) PEG-9 polydimethylsiloxyethyl dimethicone (UNII: TYP81E471F) aluminum hydroxide (UNII: 5QB0T2IUN0) diphenylsiloxy phenyl trimethicone (UNII: I445L28B12) disteardimonium hectorite (UNII: X687XDK09L) stearic acid (UNII: 4ELV7Z65AP) phenoxyethanol (UNII: HIE492ZZ3T) pentylene glycol (UNII: 50C1307PZG) edetate trisodium (UNII: 420IP921MB) aluminum distearate (UNII: 7P1HP1B9UI) tamarind seed (UNII: 6AHP8A7OML) ascorbyl glucoside (UNII: 2V52R0NHXW) talc (UNII: 7SEV7J4R1U) tocopherol (UNII: R0ZB2556P8) sea salt (UNII: 87GE52P74G) butylated hydroxytoluene (UNII: 1P9D0Z171K) sodium metabisulfite (UNII: 4VON5FNS3C) turmeric (UNII: 856YO1Z64F) ferric oxide red (UNII: 1K09F3G675) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:98132-763-01 1 in 1 BOX 1 45 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part352 06/01/2015 Labeler - Bare Escentuals Beauty, Inc. (087008363)