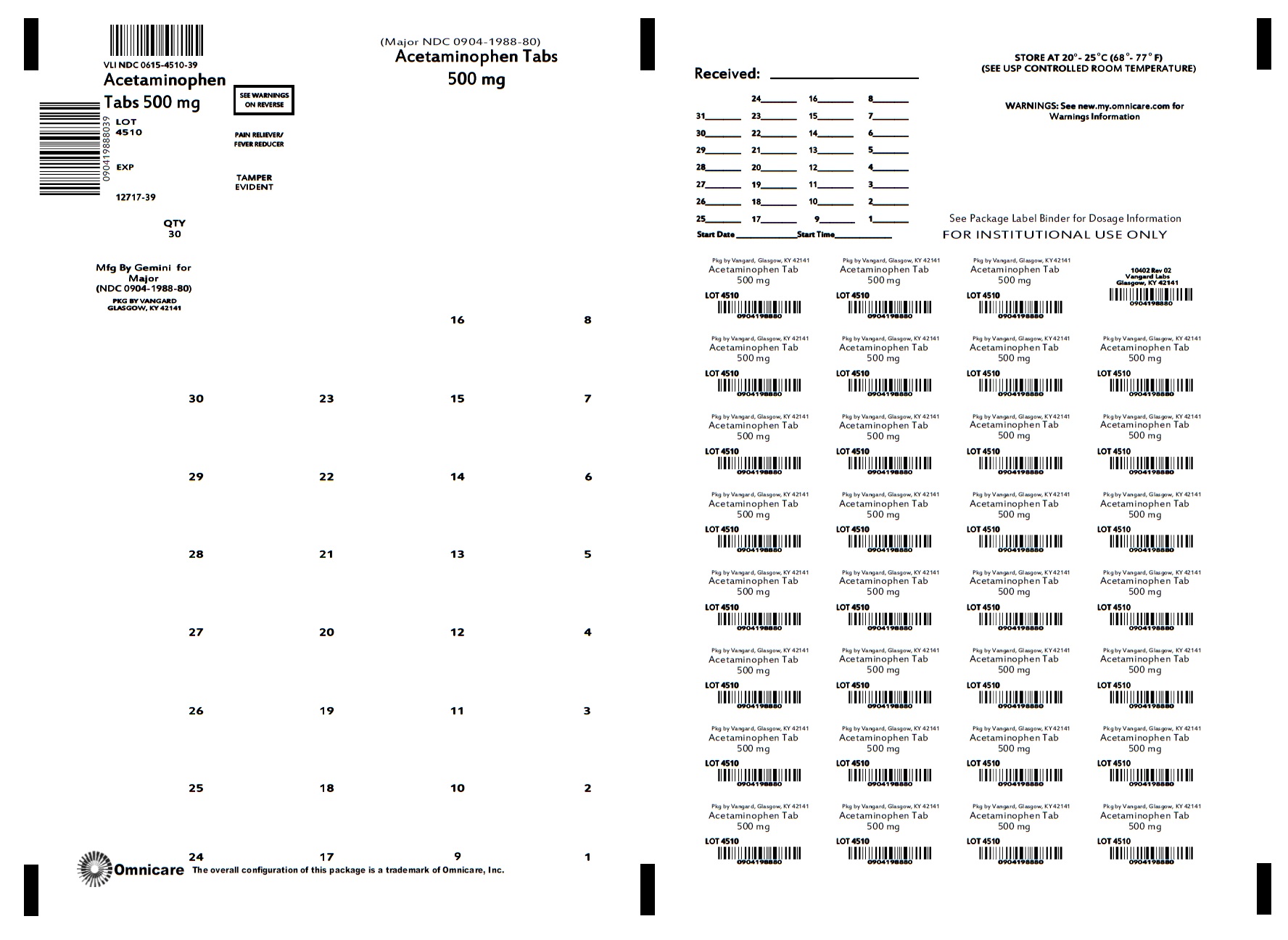
ACETAMINOPHEN- acetaminophen tablet
NCS HealthCare of KY, Inc dba Vangard Labs
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Acetaminophen Tablet 500mg
Uses
for the temporary relief of minor aches and pains due to:
- headache
- muscular aches
- backache
- minor pain of arthritis
- the common cold
- toothache
- premenstrual and menstrual cramps
- temporarily reduces fever
Warnings
Liver Warning: This product contains acetaminophen. Severe liver damage may occur if you take
- more than 4,000 mg of acetaminophen in 24 hours
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert: acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are allergic to acetaminophen or any of the inactive ingredients in this product
Directions
- do not take more than directed (see overdose warning)
adults and children 12 years and over:
- take 2 tablets every 6 hours while symptoms last.
- do not take more than 6 tablets in 24 hours unless directed by a doctor
- do not take for more than 10 days unless directed by a doctor
children under 12 years: ask a doctor
| ACETAMINOPHEN
acetaminophen tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - NCS HealthCare of KY, Inc dba Vangard Labs (050052943) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| NCS HealthCare of KY, Inc dba Vangard Labs | 050052943 | REPACK(0615-4510) | |
Revised: 1/2018
Document Id: 8b0928f1-c468-41c5-a1cc-fc2702e888a2
Set id: 5a42628d-c45d-4339-b1b8-c9b07b89cfae
Version: 16
Effective Time: 20180115
NCS HealthCare of KY, Inc dba Vangard Labs