Label: PHENDIMETRAZINE TARTRATE capsule, extended release
-
Contains inactivated NDC Code(s)
NDC Code(s): 12634-910-00, 12634-910-01, 12634-910-09, 12634-910-40, view more12634-910-42, 12634-910-45, 12634-910-50, 12634-910-52, 12634-910-54, 12634-910-57, 12634-910-58, 12634-910-59, 12634-910-60, 12634-910-61, 12634-910-63, 12634-910-66, 12634-910-67, 12634-910-69, 12634-910-71, 12634-910-74, 12634-910-78, 12634-910-79, 12634-910-80, 12634-910-81, 12634-910-82, 12634-910-84, 12634-910-85, 12634-910-90, 12634-910-91, 12634-910-92, 12634-910-93, 12634-910-94, 12634-910-95, 12634-910-96, 12634-910-97, 12634-910-98, 12634-910-99 - Packager: Apotheca Inc.
- This is a repackaged label.
- Source NDC Code(s): 0185-5254
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: CIII
- Marketing Status: New Drug Application
Drug Label Information
Updated June 29, 2016
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
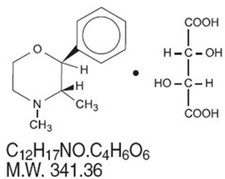
Phendimetrazine tartrate, as the dextro isomer, has the chemical name of (2S,3S)-3,4-dimethyl-2-phenylmorpholine L-(+)-tartrate (1:1).
The structural formula is as follows:
Phendimetrazine tartrate is a white, odorless crystalline powder.
It is freely soluble in water; sparingly soluble in warm alcohol, insoluble in chloroform, acetone, ether and benzene. Each capsule, for oral administration, contains 105 mg phendimetrazine tartrate manufactured in a special base designed for prolonged release. Inactive ingredients: FD&C Blue No. 1, FD&C Red No. 40, gelatin, pharmaceutical glaze, povidone, silica gel, sodium lauryl sulfate, corn starch, sucrose, talc and trace amounts of red imprinting ink.
-
CLINICAL PHARMACOLOGY
Phendimetrazine tartrate is a phenylalkylamine sympathomimetic amine with pharmacological activity similar to the prototype drugs of this class used in obesity, the amphetamines. Actions include central nervous system stimulation and elevation of blood pressure. Tachyphylaxis and tolerance have been demonstrated with all drugs of this class in which these phenomena have been looked for.
Drugs of this class used in obesity are commonly known as "anorectics" or "anorexigenics". It has not been established, however, that the action of such drugs in treating obesity is primarily one of appetite suppression. Other central nervous system actions or metabolic effects, may be involved, for example. Adult obese subjects instructed in dietary management and treated with anorectic drugs, lose more weight on the average than those treated with placebo and diet, as determined in relatively short term clinical trials.
The magnitude of increased weight loss of drug-treated patients over placebo-treated patients is only a fraction of a pound a week. The rate of weight loss is greatest in the first weeks of therapy for both drug and placebo subjects and tends to decrease in succeeding weeks. The possible origins of the increased weight loss due to the various drug effects are not established. The amount of weight loss associated with the use of an anorectic drug varies from trial to trial, and the increased weight loss appears to be related in part to variables other than the drug prescribed, such as the physician investigator, the population treated, and the diet prescribed. Studies do not permit conclusions as to the relative importance of the drug and non-drug factors on weight loss.
The natural history of obesity is measured in years, whereas the studies cited are restricted to a few weeks duration, thus, the total impact of drug-induced weight loss over that of diet alone must be considered clinically limited.
The active drug, 105 mg of phendimetrazine tartrate in each capsule of this special extended-release dosage form approximates the action of three 35 mg immediate release doses taken at four hour intervals.
The major route of elimination is via the kidneys where most of the drug and metabolites are excreted. Some of the drug is metabolized to phenmetrazine and also Phendimetrazine-N-oxide.
The average half-life of elimination when studied under controlled conditions is about 3.7 hours for both the extended-release and immediate release forms. The absorption half-life of the drug from the immediate release 35 mg phendimetrazine tablets is appreciably more rapid than the absorption rate of the drug from the extended-release formulation.
-
INDICATIONS AND USAGE
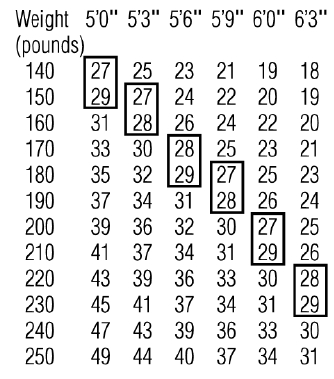
Phendimetrazine tartrate extended-release capsules are indicated in the management of exogenous obesity as a short term adjunct (a few weeks) in a regimen of weight reduction based on caloric restriction in patients with an initial body mass index (BMI) of greater than or equal to 30 kg/m2 or greater than or equal to 27 kg/m2 in the presence of other risk factors (e.g., controlled hypertension, diabetes, hyperlipidemia) who have not responded to appropriate weight reducing regimen (diet and/or exercise) alone.
Below is a chart of Body Mass Index (BMI) based on various heights and weights.
BMI is calculated by taking the patient's weight, in kilograms (kg), divided by the patient's height, in meters (m), squared. Metric conversions are as follows: pounds ÷ 2.2 = kg; inches × 0.0254 = meters
The usefulness of agents of this class (see CLINICAL PHARMACOLOGY) should be measured against possible risk factors inherent in their use such as those described below.
Phendimetrazine tartrate is indicated for use as monotherapy only.
-
CONTRAINDICATIONS
- History of cardiovascular disease (e.g., coronary artery disease, stroke, arrhythmias,
- congestive heart failure, uncontrolled hypertension, pulmonary hypertension)
- During or within 14 days following the administration of monoamine oxidase inhibitors
- Hyperthyroidism
- Glaucoma
- Agitated states
- History of drug abuse
- Pregnancy (see PRECAUTIONS, Pregnancy)
- Nursing
- Use in combination with other anorectic agents or CNS stimulants
- Known hypersensitivity or idiosyncratic reactions to sympathomimetics.
-
WARNINGS
Phendimetrazine tartrate should not be used in combination with other anorectic agents, including prescribed drugs, over-the-counter preparations and herbal products.
In a case-control epidemiological study, the use of anorectic agents, including phendimetrazine tartrate, was associated with an increased risk of developing pulmonary hypertension, a rare, but often fatal disorder. The use of anorectic agents for longer than three months was associated with a 23-fold increase in the risk of developing pulmonary hypertension. Increased risk of pulmonary hypertension with repeated courses of therapy cannot be excluded.
The onset or aggravation of exertional dyspnea, or unexplained symptoms of angina pectoris, syncope, or lower extremity edema suggest the possibility of occurrence of pulmonary hypertension. Under these circumstances, phendimetrazine tartrate should be immediately discontinued, and the patient should be evaluated for the possible presence of pulmonary hypertension.
Valvular heart disease associated with the use of some anorectic agents such as fenfluramine and dexfenfluramine has been reported. Possible contributing factors include use for extended periods of time, higher than recommended dose, and/or use in combination with other anorectic drugs.
The potential risk of possible serious adverse effects such as valvular heart disease and pulmonary hypertension should be assessed carefully against the potential benefit of weight loss. Baseline cardiac evaluation should be considered to detect preexisting valvular heart diseases or pulmonary hypertension prior to initiation of phendimetrazine treatment. Phendimetrazine tartrate is not recommended in patients with known heart murmur or valvular heart disease. Echocardiogram during and after treatment could be useful for detecting any valvular disorders which may occur.
Tolerance to the anorectic effect of phendimetrazine develops within a few weeks. When this occurs, its use should be discontinued; the maximum recommended dose should not be exceeded. Use of phendimetrazine tartrate within 14 days following the administration of monoamine oxidase inhibitors may result in a hypertensive crisis. Abrupt cessation of administration following prolonged high dosage results in extreme fatigue and depression. Because of the effect on the central nervous system, phendimetrazine may impair the ability of the patient to engage in potentially hazardous activities such as operating machinery or driving a motor vehicle; the patient should therefore be cautioned accordingly.
Phendimetrazine tartrate is not recommended for patients who used any anorectic agents within the prior year.
-
PRECAUTIONS
General
Caution is to be exercised in prescribing phendimetrazine tartrate for patients with even mild hypertension.
Insulin or oral hypoglycemic medication requirements in diabetes mellitus may be altered in association with the use of phendimetrazine and the concomitant dietary regimen.
Phendimetrazine may decrease the hypotensive effect of guanethidine.
The least amount feasible should be prescribed or dispensed at one time in order to minimize the possibility of overdosage.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies with phendimetrazine tartrate sustained release have not been performed to evaluate carcinogenic potential, mutagenic potential or effects on fertility.
Pregnancy
Pregnancy Category X
Phendimetrazine tartrate is contraindicated during pregnancy because weight loss offers no potential benefit to a pregnant woman and may result in fetal harm. A minimum weight gain, and no weight loss, is currently recommended for all pregnant women, including those who are already overweight or obese, due to obligatory weight gain that occurs in maternal tissues during pregnancy. Phendimetrazine tartrate, a phenylalkylamine sympathomimetic amine has pharmacological activity similar to amphetamines (see CLINICAL PHARMACOLOGY). Animal reproduction studies have not been conducted in phendimetrazine tartrate. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to a fetus.
Nursing Mothers
It is not known if phendimetrazine tartrate is excreted in human milk. Phendimetrazine tartrate, a phenylalkylamine sympathomimetic amine, has pharmacological activity similar to the amphetamines (see CLINICAL PHARMACOLOGY), and other amphetamines are present in human milk. Because of the potential for serious adverse reactions in nursing infants, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Drug Interactions
Monoamine Oxidase Inhibitors
Use of phendimetrazine tartrate is contraindicated during or within 14 days following the administration of monoamine oxidase inhibitors because of the risk of hypertensive crisis.
Alcohol
Concomitant use of alcohol with phendimetrazine tartrate may result in an adverse drug reaction.
Insulin and Oral Hypoglycemic Medications
Requirements may be altered
Adrenergic Neuron Blocking Drugs
phendimetrazine tartrate may decrease the hypotensive effect of adrenergic neuron blocking drugs.
Pediatric Use
Safety and effectiveness in pediatric patients have not been established. Because pediatric obesity is a chronic condition requiring long-term treatment, the use of phendimetrazine tartrate ER approved for short-term therapy, is not recommended in patients less that 17 years of age.
Renal Impairment
Phendimetrazine tartrate extended-release capsules were not studied in patients with renal impairment. As phendimetrazine tartrate is excreted in urine, exposure increases can be expected in patients with renal impairment. Use caution when administering phendimetrazine tartrate to patients with renal impairment.
Geriatric Use
In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
The major route of elimination is via the kidney where most of the drug and metabolites are excreted. The risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
-
ADVERSE REACTIONS
The following adverse reactions are described, or described in greater detail, in other sections:
- Primary pulmonary hypertension (see Warnings)
- Valvular heart disease (see Warnings)
-Effect on the ability to engage in potentially hazardous tasks (see Warnings)
-Withdrawal effects following prolonged high dosage administration (see Drug Abuse and Dependence)
The following adverse reactions to phendimetrazine have been identified:
Cardiovascular
Primary pulmonary hypertension and/or regurgitant cardiac valvular disease, palpitation, tachycardia, elevated blood pressure, ischemic events.
-
DRUG ABUSE AND DEPENDENCE
Controlled Substance
Phendimetrazine tartrate extended-release capsules are defined by the Drug Enforcement Administration as a Schedule III controlled substance.
Abuse
Phendimetrazine tartrate is related chemically and pharmacologically to the amphetamines. Amphetamines and related stimulant drugs have been extensively abused, and the possibility of abuse of phendimetrazine should be kept in mind when evaluating the desirability of including a drug as part of a weight reduction program.
Dependence
Abuse of amphetamines and related drugs may be associated with intense psychological dependence and severe social dysfunction. There are reports of patients who have increased the dosage to many times that recommended. Abrupt cessation following prolonged high dosage administration results in extreme fatigue and mental depression; changes are also noted on the sleep EEG. Manifestations of chronic intoxication with anorectic drugs include severe dermatoses, marked insomnia, irritability, hyperactivity and personality changes. The most severe manifestation of chronic intoxications is psychosis, often clinically indistinguishable from schizophrenia.
-
OVERDOSAGE
The least amount feasible should be prescribed or dispensed at one time in order to minimize the possibility of overdosage.
Acute Overdosage
Acute overdosage with phendimetrazine tartrate may manifest itself by the following signs and symptoms: unusual restlessness, confusion, belligerence, hallucinations, and panic states. Fatigue and depression usually follow the central stimulation. Cardiovascular effects include tachycardia, arrhythmias, hypertension, or hypotension and circulatory collapse. Gastrointestinal symptoms include nausea, vomiting, diarrhea, and abdominal cramps. Poisoning may result in convulsions, coma, and death.
The management of acute overdosage is largely symptomatic. It includes lavage and sedation with a barbiturate. If hypertension is marked, the use of a nitrate or rapid-acting alpha receptor-blocking agent should be considered. Experience with hemodialysis or peritoneal dialysis is inadequate to permit recommendations for its use.
Chronic Intoxication
Manifestations of chronic intoxication with anorectic drugs include sever dermatoses, marked insomnia, irritability, hyperactivity and personality changes. The most server manifestation of chronic intoxications is psychosis, often clinically indistinguishable from schizophrenia. See Drug Abuse and Dependence.
-
DOSAGE AND ADMINISTRATION
Extended-release capsule
Since the product is an extended-release dosage form, limit to one extended-release capsule (105 mg phendimetrazine tartrate) in the morning (30 to 60 minutes before morning meal).
Each extended-release capsule contains 105 mg phendimetrazine tartrate in a Brown/Clear capsule imprinted E 5254.
-
HOW SUPPLIED
NDC 12634-910-00 Bottles of 10
NDC 12634-910-01 Bottles of 100
NDC 12634-910-09 Bottles of 35
NDC 12634-910-40 Bottles of 40
NDC 12634-910-42 Bottles of 42
NDC 12634-910-45 Bottles of 45
NDC 12634-910-50 Bottles of 50
NDC 12634-910-52 Blister Pack of 12
NDC 12634-910-54 Blister Pack of 14
NDC 12634-910-57 Blister Pack of 20
NDC 12634-910-58 Blister Pack of 28
NDC 12634-910-59 Blister Pack of 30
NDC 12634-910-60 Bottles of 60
NDC 12634-910-61 Blister Pack of 10
NDC 12634-910-63 Blister Pack of 3
NDC 12634-910-66 Blister Pack of 6
NDC 12634-910-67 Blister Pack of 7
NDC 12634-910-69 Blister Pack of 9
NDC 12634-910-71 Bottles of 30
NDC 12634-910-74 Bottles of 24
NDC 12634-910-78 Bottles of 28
NDC 12634-910-79 Bottles of 25
NDC 12634-910-80 Bottles of 20
NDC 12634-910-81 Bottles of 21
NDC 12634-910-82 Bottles of 12
NDC 12634-910-84 Bottles of 14
NDC 12634-910-85 Bottles of 15
NDC 12634-910-90 Bottles of 90
NDC 12634-910-91 Blister Pack of 1
NDC 12634-910-92 Bottles of 2
NDC 12634-910-93 Bottles of 3
NDC 12634-910-94 Bottles of 4
NDC 12634-910-95 Bottles of 5
NDC 12634-910-96 Bottles of 6
NDC 12634-910-97 Bottles of 7
NDC 12634-910-98 Bottles of 8
NDC 12634-910-99 Bottles of 9
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Protect from moisture.
DISPENSE IN A TIGHT CONTAINER AS DEFINED IN THE USP.
To report SUSPECTED ADVERSE REACTIONS, contact FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Manufactured by Epic Pharma, LLC
Laurelton, NY 11413
Repackaged and Distributed by:
Apotheca Inc.
Phoenix, AZ 85006
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
PHENDIMETRAZINE TARTRATE
phendimetrazine tartrate capsule, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:12634-910(NDC:0185-5254) Route of Administration ORAL DEA Schedule CIII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHENDIMETRAZINE TARTRATE (UNII: 6985IP0T80) (PHENDIMETRAZINE - UNII:AB2794W8KV) PHENDIMETRAZINE TARTRATE 105 mg Inactive Ingredients Ingredient Name Strength FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 40 (UNII: WZB9127XOA) GELATIN (UNII: 2G86QN327L) POVIDONE (UNII: FZ989GH94E) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM LAURYL SULFATE (UNII: 368GB5141J) STARCH, CORN (UNII: O8232NY3SJ) SUCROSE (UNII: C151H8M554) TALC (UNII: 7SEV7J4R1U) SHELLAC (UNII: 46N107B71O) Product Characteristics Color brown (Brown/Clear) Score no score Shape CAPSULE (CAPSULE) Size 19mm Flavor Imprint Code E;5254 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:12634-910-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 2 NDC:12634-910-00 10 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 3 NDC:12634-910-09 35 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 4 NDC:12634-910-40 40 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 5 NDC:12634-910-42 42 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 6 NDC:12634-910-45 45 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 7 NDC:12634-910-50 50 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 8 NDC:12634-910-52 12 in 1 BLISTER PACK; Type 0: Not a Combination Product 09/06/1977 9 NDC:12634-910-54 14 in 1 BLISTER PACK; Type 0: Not a Combination Product 09/06/1977 10 NDC:12634-910-57 20 in 1 BLISTER PACK; Type 0: Not a Combination Product 09/06/1977 11 NDC:12634-910-58 28 in 1 BLISTER PACK; Type 0: Not a Combination Product 09/06/1977 12 NDC:12634-910-59 30 in 1 BLISTER PACK; Type 0: Not a Combination Product 09/06/1977 13 NDC:12634-910-60 60 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 14 NDC:12634-910-61 10 in 1 BLISTER PACK; Type 0: Not a Combination Product 09/06/1977 15 NDC:12634-910-63 3 in 1 BLISTER PACK; Type 0: Not a Combination Product 09/06/1977 16 NDC:12634-910-66 6 in 1 BLISTER PACK; Type 0: Not a Combination Product 09/06/1977 17 NDC:12634-910-67 7 in 1 BLISTER PACK; Type 0: Not a Combination Product 09/06/1977 18 NDC:12634-910-69 9 in 1 BLISTER PACK; Type 0: Not a Combination Product 09/06/1977 19 NDC:12634-910-71 30 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 20 NDC:12634-910-74 24 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 21 NDC:12634-910-78 28 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 22 NDC:12634-910-79 25 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 23 NDC:12634-910-80 20 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 24 NDC:12634-910-81 21 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 25 NDC:12634-910-82 12 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 26 NDC:12634-910-84 14 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 27 NDC:12634-910-85 15 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 28 NDC:12634-910-90 90 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 29 NDC:12634-910-91 1 in 1 BLISTER PACK; Type 0: Not a Combination Product 09/06/1977 30 NDC:12634-910-92 2 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 31 NDC:12634-910-93 3 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 32 NDC:12634-910-94 4 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 33 NDC:12634-910-95 5 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 34 NDC:12634-910-96 6 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 35 NDC:12634-910-97 7 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 36 NDC:12634-910-98 8 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 37 NDC:12634-910-99 9 in 1 BOTTLE; Type 0: Not a Combination Product 09/06/1977 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA018074 09/06/1977 Labeler - Apotheca Inc. (051457844) Establishment Name Address ID/FEI Business Operations Apotheca Inc. 051457844 relabel(12634-910) , repack(12634-910)