Label: ACL11CH NERVE BLOCK- kit
- NHRIC Code(s): 55553-482-02
- NDC Code(s): 54365-400-11
- Packager: Smiths Medical ASD, Inc.
- Category: MEDICAL DEVICE
Drug Label Information
Updated April 14, 2014
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
-
WARNINGS
Warnings
For external use only. Flammable, keep away from fire or flame.
To reduce the risk of fire, PREP CAREFULLY:
•
solution contains alcohol and gives off flammable vapors
•
avoid getting solution into hairy areas. Hair may take up to 1 hour to dry. Wet hair is flammable.
•
do not drape or use ignition source (e.g., cautery, laser) until solution is completely dry (minimum of 3 minutes on hairless skin; up to 1 hour in hair).
•
do not allow solution to pool
•
remove wet materials from prep area - DO NOT USE
- WHEN USING
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
-
WHEN USING
Directions
•
use with care in premature infants or infants under 2 months of age. These products may cause irritation or chemical burns.
•
use in a well ventilated area
•
maximal treatment area for one applicator is approximately 4 in. x 5 in. (130 cm2)
•
remove applicator from package; do not touch sponge
•
hold the applicator with the sponge down. Pinch wings only once to activate the ampule and release the antiseptic.
•
wet the sponge by pressing and releasing the sponge against the treatment area until liquid is visible on the skin
•
completely wet the treatment area with antiseptic
•
dry surgical sites (e.g., abdomen or arm): use gentle repeated back-and-forth strokes for 30 seconds
•
moist surgical sites (e.g., inguinal fold): use gentle repeated back-and-forth strokes for 2 minutes
•
allow the solution to completely dry (minimum of 3 minutes on hairless skin; up to 1 hour in hair). Do not blot or wipe away.
•
discard the applicator after a single use along with any portion of the solution not required to cover the prep area. It is not necessary to use the entire amount available. - STORAGE AND HANDLING
- INACTIVE INGREDIENT
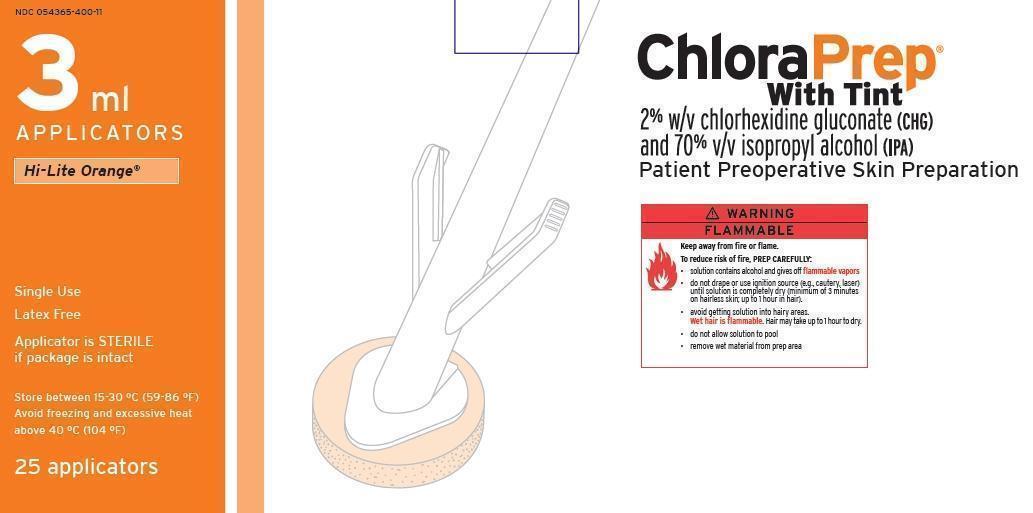
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ACL11CH NERVE BLOCK
regional anesthesia kit kitProduct Information Product Type MEDICAL DEVICE Item Code (Source) NHRIC:55553-482 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:55553-482-02 30 in 1 CASE 1 1 in 1 PACKAGE, COMBINATION Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 APPLICATOR 3 mL Part 1 of 1 CHLORAPREP ONE-STEP
chlorhexidine gluconate and isopropyl alcohol solutionProduct Information Item Code (Source) NDC:54365-400 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CHLORHEXIDINE GLUCONATE (UNII: MOR84MUD8E) (CHLORHEXIDINE - UNII:R4KO0DY52L) CHLORHEXIDINE GLUCONATE 20 mg in 1 mL ISOPROPYL ALCOHOL (UNII: ND2M416302) (ISOPROPYL ALCOHOL - UNII:ND2M416302) ISOPROPYL ALCOHOL 0.70 mL in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54365-400-11 3 mL in 1 APPLICATOR Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA020832 08/18/2006 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date premarket notification K965017 01/14/2014 Labeler - Smiths Medical ASD, Inc. (137835299) Establishment Name Address ID/FEI Business Operations Smiths Medical ASD, Inc. 137835299 manufacture Establishment Name Address ID/FEI Business Operations CareFusion 213 LLC 830430778 manufacture