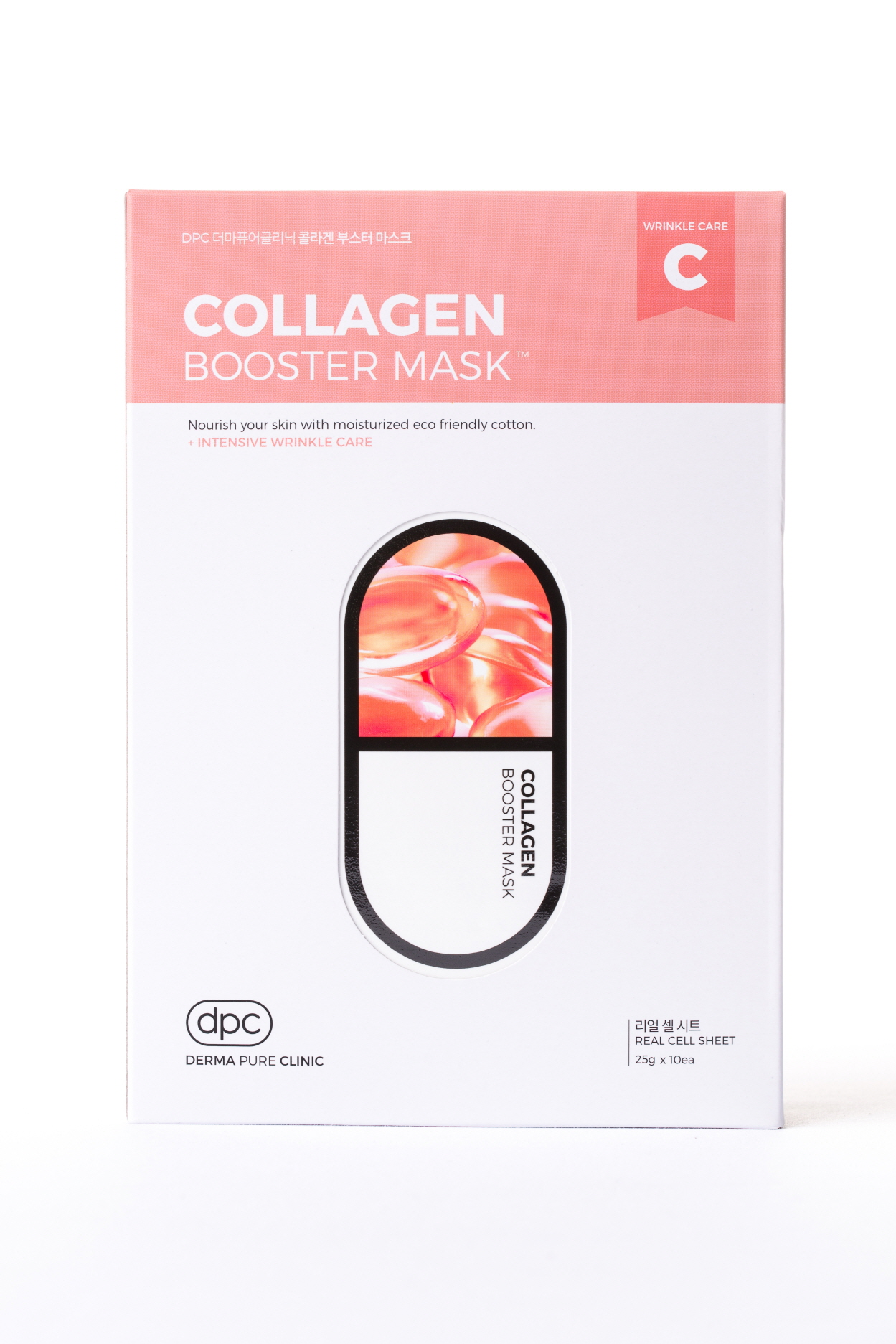
Label: DPC COLLAGEN BOOSTER MASK- adenosine liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 71673-0002-1 - Packager: MSCO
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated August 23, 2017
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
-
INACTIVE INGREDIENT
Water
Propanediol
Glycerin
Erythritol
Butylene Glycol
Caprylic/Capric Triglyceride
Dipotassium Glycyrrhizate
Disodium EDTA
Carbomer
Xanthan Gum
Hydroxyethylcellulose
Chlorphenesin
Sodium Hyaluronate
Squalane
Hydrolyzed Collagen
Simmondsia Chinensis (Jojoba) Seed Oil
Glycine Soja (Soybean) Seed Extract
Phaseolus radiatus seed extract
Thioctic Acid
Glutathione
Polyglutamic Acid
Methylpropanediol
Phenoxyethanol
Polyacrylate-13
Polyisobutene
Polysorbate 20
Sorbitan isostearate
Glyceryl Stearate
Polysorbate 60
Arginine
Citrus Grandis (Grapefruit) Seed Extract
Acorus Calamus Root Extract
Perilla Ocymoides Leaf Extract
1,2-Hexanediol
Caprylyl Glycol
Glycyrrhiza Glabra (Licorice) Root Extract
Zingiber Officinale (Ginger) Root Extract
Schizandra Chinensis Fruit Extract
Coptis Japonica Root Extract
Camellia Sinensis Leaf Extract
Fragrance - PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- WARNINGS
- DOSAGE & ADMINISTRATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DPC COLLAGEN BOOSTER MASK
adenosine liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71673-0002 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ADENOSINE (UNII: K72T3FS567) (ADENOSINE - UNII:K72T3FS567) ADENOSINE 0.04 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) HYALURONATE SODIUM (UNII: YSE9PPT4TH) GLYCERIN (UNII: PDC6A3C0OX) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71673-0002-1 5 in 1 PACKAGE 08/23/2017 1 25 g in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 08/23/2017 Labeler - MSCO (689039838) Registrant - MSCO (689039838) Establishment Name Address ID/FEI Business Operations MSCO 689039838 label(71673-0002) Establishment Name Address ID/FEI Business Operations CNF CO.,LTD. 689852175 manufacture(71673-0002)