Label: CAPSAICIN cream
- NDC Code(s): 69420-6025-1, 69420-6025-5
- Packager: SA3, LLC
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated May 17, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
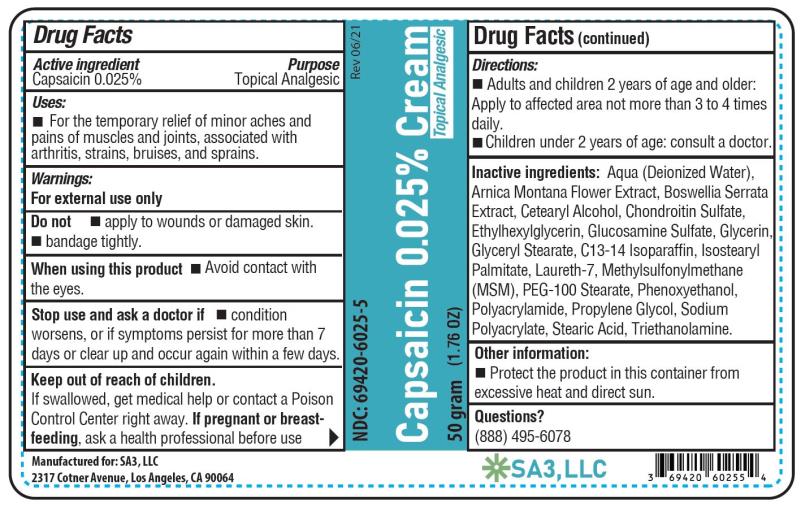
- Active ingredient
- Purpose
- Uses
-
Warnings
For external use only
Read all warnings and directions before use. Test first on small area of skin.
Do not
• Apply to wounds or damaged skin
• Bandage tightly
• If you are allergic to capsicum or chili peppers
When using this product
• You may experience a burning sensation. The intensity of this reaction varies among individuals and may be severe. With regular use, this sensation generally disappears after several days.
• Avoid contact with the eyes, lips, nose and mucous membranes
• Do not tightly wrap or bandage the treated area
• Do not apply heat to the treated area immediately before or after use
- Directions
- Other information
-
Inactive ingredients
Aqua (Deionized Water), Arnica Montana Flower Extract, Boswellia Serrata Extract, Cetearyl Alcohol, Chondroitin Sulfate, Ethylhexylglycerin, Glucosamine Sulfate, Glycerin, Glyceryl Stearate, C13-14 Isoparaffin, Isostearyl Palmitate, Laureth-7, Methylsufonylmethane (MSM), PEG-100 Stearate, Phenoxyethanol, Polyacrylamide, Propylene Glycol, Sodium Polyacrylate, Stearic Acid, and Triethanolamine
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CAPSAICIN
capsaicin creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69420-6025 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAPSAICIN (UNII: S07O44R1ZM) (CAPSAICIN - UNII:S07O44R1ZM) CAPSAICIN 0.025 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ARNICA MONTANA FLOWER (UNII: OZ0E5Y15PZ) INDIAN FRANKINCENSE (UNII: 4PW41QCO2M) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) GLUCOSAMINE SULFATE (UNII: 1FW7WLR731) GLYCERIN (UNII: PDC6A3C0OX) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) C13-14 ISOPARAFFIN (UNII: E4F12ROE70) ISOSTEARYL PALMITATE (UNII: 9EHU0R7ER1) LAURETH-7 (UNII: Z95S6G8201) DIMETHYL SULFONE (UNII: 9H4PO4Z4FT) PEG-100 STEARATE (UNII: YD01N1999R) PHENOXYETHANOL (UNII: HIE492ZZ3T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) STEARIC ACID (UNII: 4ELV7Z65AP) TROLAMINE (UNII: 9O3K93S3TK) CHONDROITIN SULFATE (BOVINE) (UNII: 6IC1M3OG5Z) POLYACRYLAMIDE (10000 MW) (UNII: E2KR9C9V2I) SODIUM POLYACRYLATE (2500000 MW) (UNII: 05I15JNI2J) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69420-6025-5 50 g in 1 TUBE; Type 0: Not a Combination Product 06/11/2021 2 NDC:69420-6025-1 120 g in 1 TUBE; Type 0: Not a Combination Product 06/11/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 06/11/2021 Labeler - SA3, LLC (079627454)