Label: SODIUM NITRITE injection, solution
- NDC Code(s): 60267-311-10
- Packager: Hope Pharmaceuticals
- Category: HUMAN PRESCRIPTION DRUG LABEL
Drug Label Information
Updated February 14, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use Sodium Nitrite Injection safely and effectively. See full prescribing information for Sodium Nitrite Injection.
Sodium Nitrite Injection, USP
Initial U.S. Approval: 2011WARNING: LIFE-THREATENING HYPOTENSION AND METHEMOGLOBIN FORMATION
See full prescribing information for complete boxed warning.
Sodium nitrite can cause serious adverse reactions and death from:
Patients should be closely monitored to ensure adequate perfusion and oxygenation during treatment with sodium nitrite.
INDICATIONS AND USAGE
DOSAGE AND ADMINISTRATION
- If clinical suspicion of cyanide poisoning is high, administer Sodium Nitrite Injection without delay and in conjunction with appropriate airway, ventilatory, and circulatory support. (2.1)
- The expert advice of a regional poison control center may be obtained by calling 1-800-222-1222. (2.1)
Dosing:
Age Intravenous Dose of Sodium Nitrite and Sodium Thiosulfate Adults - 1.)
- Sodium Nitrite -10 mL of sodium nitrite at the rate of 2.5 to 5 mL/minute
- 2.)
- Sodium Thiosulfate - 50 mL of sodium thiosulfate immediately following administration of sodium nitrite.
Children - 1.)
- Sodium Nitrite - 0.2 mL/kg (6 mg/kg or 6-8 mL/m2 BSA) of sodium nitrite at the rate of 2.5 to 5 mL/minute not to exceed 10 mL
- 2.)
- Sodium Thiosulfate - 1 mL/kg of body weight (250 mg/kg or approximately 30-40 mL/m2 of BSA) not to exceed 50 mL total dose immediately following administration of sodium nitrite.
- Redosing: If signs of cyanide poisoning reappear, repeat treatment using one-half the original dose of both sodium nitrite and sodium thiosulfate. (2.2)
- Monitoring: Blood pressure must be monitored during treatment. (2.2)
- Sodium nitrite is chemically incompatible with hydroxocobalamin and should not be administered via the same intravenous line. (2.4)
DOSAGE FORMS AND STRENGTHS
Sodium Nitrite Injection consists of:
- one vial of sodium nitrite injection, USP 300 mg/10 mL (30 mg/mL) (3)
CONTRAINDICATIONS
- None. (4)
WARNINGS AND PRECAUTIONS
- Methemoglobinemia: Sodium nitrite reacts with hemoglobin to form methemoglobin and should be used with caution in patients known to have anemia. Monitor oxyhemoglobin and methemoglobin levels by pulse co-oximetry or other measurements. Optimally, the sodium nitrite dose should be reduced in proportion to the oxygen carrying capacity. (5.2)
- Smoke inhalation: Carbon monoxide contained in smoke can result in the formation of carboxyhemoglobin that can reduce the oxygen carrying capacity of the blood. Sodium nitrite should be used with caution in patients with smoke inhalation injury because of the potential for worsening hypoxia due to methemoglobin formation.
Carboxyhemoglobin and oxyhemoglobin levels should be monitored by pulse oximetry or other measurements in patients that present with evidence of smoke inhalation. Optimally, the sodium nitrite dose should be reduced in proportion to the oxygen carrying capacity. (5.4)
ADVERSE REACTIONS
Most common adverse reactions are:
- Syncope, hypotension, tachycardia, palpitations, dysrhythmia, methemoglobinemia, headache, dizziness, blurred vision, seizures, confusion, coma (6)
To report SUSPECTED ADVERSE REACTIONS, contact Hope Pharmaceuticals at 1-800-755-9595 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 1/2021
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: LIFE THREATENING HYPOTENSION AND METHEMOGLOBIN FORMATION
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Important Dosage and Administration Instructions
2.2 Recommended Dosing
2.3 Recommended Monitoring
2.4 Incompatibility Information
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Hypotension
5.2 Methemoglobinemia
5.3 Anemia
5.4 Smoke Inhalation Injury
5.5 Neonates and Infants
5.6 G6PD Deficiency
5.7 Use with Other Drugs
6 ADVERSE REACTIONS
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: LIFE THREATENING HYPOTENSION AND METHEMOGLOBIN FORMATION
Sodium nitrite can cause serious adverse reactions and death in humans, even at doses less than twice the recommended therapeutic dose. Sodium nitrite causes hypotension and methemoglobin formation, which diminishes oxygen carrying capacity. Hypotension and methemoglobin formation can occur concurrently or separately. Because of these risks, sodium nitrite should be used to treat acute life-threatening cyanide poisoning and be used with caution in patients where the diagnosis of cyanide poisoning is uncertain.
Patients should be closely monitored to ensure adequate perfusion and oxygenation during treatment with sodium nitrite. Alternative therapeutic approaches should be considered in patients known to have diminished oxygen or cardiovascular reserve (e.g., smoke inhalation victims, pre-existing anemia, cardiac or respiratory compromise), and those at higher risk of developing methemoglobinemia (e.g., congenital methemoglobin reductase deficiency) as they are at greater risk for potentially life-threatening adverse events related to the use of sodium nitrite. [see Warnings and Precautions (5.1 and 5.2)]
-
1 INDICATIONS AND USAGE
Sodium Nitrite Injection, an antidote, is indicated for sequential use with sodium thiosulfate for the treatment of acute cyanide poisoning that is judged to be serious or life-threatening. When the diagnosis of cyanide poisoning is uncertain, the potentially life-threatening risks associated with Sodium Nitrite Injection should be carefully weighed against the potential benefits, especially if the patient is not in extremis.
-
2 DOSAGE AND ADMINISTRATION
2.1 Important Dosage and Administration Instructions
- If clinical suspicion of cyanide poisoning is high, administer Sodium Nitrite Injection without delay.
- Comprehensive treatment of acute cyanide intoxication requires support of vital functions. Administration of sodium nitrite and sodium thiosulfate should be considered adjunctive to appropriate supportive therapies. Airway, ventilatory and circulatory support, and oxygen administration should not be delayed in order to administer sodium nitrite and sodium thiosulfate [see Warnings and Precautions (5.1)].
- The expert advice of a regional poison control center may be obtained by calling 1-800-222-1222.
Identifying Patients with Cyanide Poisoning
Cyanide poisoning may result from inhalation, ingestion, or dermal exposure to various cyanide-containing compounds, including smoke from closed-space fires. Sources of cyanide poisoning include hydrogen cyanide and its salts, cyanogenic plants, aliphatic nitriles, and prolonged exposure to sodium nitroprusside..
The presence and extent of cyanide poisoning are often initially unknown. There is no widely available, rapid, confirmatory cyanide blood test. Treatment decisions must be made on the basis of clinical history and signs and symptoms of cyanide intoxication
Table 1. Common Signs and Symptoms of Cyanide Poisoning Symptoms - Headache
- Confusion
- Dyspnea
- Chest Tightness
- Nausea
Signs - Altered Mental Status
(e.g., confusion, disorientation) - Seizures or Coma
- Mydriasis
- Tachypnea/Hyperpnea (early)
- Bradypnea/Apnea (late)
- Hypertension (early)/ Hypotension (late)
- Cardiovascular Collapse
- Vomiting
- Plasma Lactate Concentration ≥ 8 mmol/L
In some settings, panic symptoms including tachypnea and vomiting may mimic early cyanide poisoning signs. The presence of altered mental status (e.g., confusion and disorientation) and/or mydriasis is suggestive of true cyanide poisoning although these signs can occur with other toxic exposures as well.
Smoke Inhalation
Not all smoke inhalation victims will have cyanide poisoning and may present with burns, trauma, and exposure to other toxic substances making a diagnosis of cyanide poisoning particularly difficult. Prior to administration of Sodium Nitrite Injection, smoke-inhalation victims should be assessed for the following:
- Exposure to fire or smoke in an enclosed area
- Presence of soot around the mouth, nose, or oropharynx
- Altered mental status
Although hypotension is highly suggestive of cyanide poisoning, it is only present in a small percentage of cyanide-poisoned smoke inhalation victims. Also indicative of cyanide poisoning is a plasma lactate concentration greater than or equal to 10 mmol/L (a value higher than that typically listed in the table of signs and symptoms of isolated cyanide poisoning because carbon monoxide associated with smoke inhalation also contributes to lactic acidemia). If cyanide poisoning is suspected, treatment should not be delayed in order to obtain a plasma lactate concentration.
Use with Other Cyanide Antidotes
The safety of administering other cyanide antidotes simultaneously with Sodium Nitrite Injection has not been established. If a decision is made to administer another cyanide antidote with Sodium Nitrite Injection, these drugs should not be administered concurrently in the same intravenous (IV) line. [see Dosage and Administration (2.2)]
2.2 Recommended Dosing
Sodium Nitrite Injection and Sodium Thiosulfate Injection are administered by slow intravenous injection. They should be given as early as possible after a diagnosis of acute life-threatening cyanide poisoning has been established. Sodium nitrite should be administered first, followed immediately by sodium thiosulfate. Blood pressure must be monitored during infusion in both adults and children. The rate of infusion should be decreased if significant hypotension is noted.
Age Intravenous Dose of Sodium Nitrite and Sodium Thiosulfate Adults - 1.)
- Sodium Nitrite -10 mL of sodium nitrite at the rate of 2.5 to 5 mL/minute
- 2.)
- Sodium Thiosulfate - 50 mL of sodium thiosulfate immediately following administration of sodium nitrite.
Children - 1.)
- Sodium Nitrite -0.2 mL/kg (6 mg/kg or 6-8 mL/m2 BSA) of sodium nitrite at the rate of 2.5 to 5 mL/minute not to exceed 10 mL
- 2.)
- Sodium Thiosulfate - 1 mL/kg of body weight (250 mg/kg or approximately 30-40 mL/m2 of BSA) not to exceed 50 mL total dose immediately following administration of sodium nitrite.
NOTE: If signs of poisoning reappear, repeat treatment using one-half the original dose of both sodium nitrite and sodium thiosulfate.
In adult and pediatric patients with known anemia, it is recommended that the dosage of sodium nitrite should be reduced proportionately to the hemoglobin concentration. [see Warnings and Precautions (5.2)]
All parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
2.3 Recommended Monitoring
Monitor patients for at least 24-48 hours after Sodium Nitrite Injection administration for adequacy of oxygenation and perfusion and for recurrent signs and symptoms of cyanide toxicity. When possible, obtain hemoglobin/hematocrit when treatment is initiated. Measurements of oxygen saturation using standard pulse oximetry and calculated oxygen saturation values based on measured PO2 are unreliable in the presence of methemoglobinemia.
Methemoglobin level: Administrations of sodium nitrite solely to achieve an arbitrary level of methemoglobinemia may be unnecessary and potentially hazardous. The therapeutic effects of sodium nitrite do not appear to be mediated by methemoglobin formation alone [see Clinical Pharmacology (12)] and clinical responses to sodium nitrite administration have been reported in association with methemoglobin levels of less than 10%. Administration of sodium nitrite beyond the initial dose should be guided primarily by clinical response to treatment (i.e., a second dose should be considered only if there is inadequate clinical response to the first dose). It is generally recommended that methemoglobin concentrations be closely monitored and kept below 30%. Monitor serum methemoglobin levels during treatment using co-oximetry, and discontinue administration of sodium nitrite when methemoglobin levels exceed 30%. Intravenous methylene blue and exchange transfusion have been reported in the literature as treatments for life-threatening methemoglobinemia.
2.4 Incompatibility Information
Chemical incompatibility has been reported between sodium nitrite and hydroxocobalamin and these drugs should not be administered simultaneously through the same IV line. No chemical incompatibility has been reported between sodium thiosulfate and sodium nitrite, when administered sequentially through the same IV line as described in Dosage and Administration.
Simultaneous administration of sodium nitrite and blood products (whole blood, packed red cells, platelet concentrate and/or fresh frozen plasma) through the same intravenous line is not recommended. However, blood products and sodium nitrite can be administered simultaneously using separate intravenous lines (preferably on contralateral extremities, if peripheral lines are being used).
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Hypotension
Sodium nitrite has been associated with severe hypotension, methemoglobinemia, and death at doses less than twice recommended therapeutic doses. Hypotension may occur concurrently or separately. Sodium nitrite should be used to treat life-threatening cyanide poisoning. When the diagnosis of cyanide poisoning is uncertain and/or the patient is not in extremis, special consideration should be given to administration of sodium nitrite if the patient is known or suspected to have diminished oxygen or cardiovascular reserve (e.g., smoke inhalation victims, pre-existing anemia, substantial blood loss, cardiac or respiratory compromise) or to be at higher risk of developing methemoglobinemia (e.g., congenital methemoglobin reductase deficiency).
5.2 Methemoglobinemia
Supportive care alone may be sufficient treatment without administration of antidotes for many cases of cyanide intoxication, particularly in conscious patients without signs of severe toxicity. Monitor patients closely to ensure adequate perfusion and oxygenation during treatment with sodium nitrite.
Monitor methemoglobin levels and administer oxygen during treatment with sodium nitrite whenever possible. When sodium nitrite is administered to humans a wide range of methemoglobin concentrations occur. Methemoglobin concentrations as high as 58% have been reported after two 300-mg doses of sodium nitrite administered to an adult. Sodium nitrite should be used with caution in the presence of other drugs that may cause methemoglobinemia such as procaine and nitroprusside. Sodium nitrite should be used with caution in patients who may be particularly susceptible to injury from vasodilation and its related hemodynamic sequelae. Hemodynamics should be monitored closely during and after administration of sodium nitrite, and infusion rates should be slowed if hypotension occurs.
5.3 Anemia
Use sodium nitrite with caution in patients with known anemia. Patients with anemia will form more methemoglobin (as a percentage of total hemoglobin) than persons with normal red blood cell (RBC) volumes. Optimally, these patients should receive a sodium nitrite dose that is reduced in proportion to their oxygen carrying capacity.
5.4 Smoke Inhalation Injury
Use sodium nitrite should be used with caution in persons with smoke inhalation injury or carbon monoxide poisoning because of the potential for worsening hypoxia due to methemoglobin formation.
5.5 Neonates and Infants
Neonates and infants may be more susceptible than adults and older pediatric patients to severe methemoglobinemia when sodium nitrite is administered. Follow reduced dosing guidelines in pediatric patients.
5.6 G6PD Deficiency
Because patients with G6PD deficiency are at increased risk of a hemolytic crisis with sodium nitrite administration, consider alternative therapeutic approaches in these patients. Monitor patients with known or suspected G6PD deficiency for an acute drop in hematocrit. Exchange transfusion may be needed for patients with G6PD deficiency who receive sodium nitrite.
-
6 ADVERSE REACTIONS
There have been no controlled clinical trials conducted to systematically assess the adverse events profile of sodium nitrite.
The medical literature has reported the following adverse events in association with sodium nitrite administration. These adverse events were not reported in the context of controlled trials or with consistent monitoring and reporting methodologies for adverse events. Therefore, frequency of occurrence of these adverse events cannot be assessed.
Cardiovascular system: syncope, hypotension, tachycardia, methemoglobinemia, palpitations, dysrhythmia
Hematological: methemoglobinemia
Central nervous system: headache, dizziness, blurred vision, seizures, confusion, coma
Gastrointestinal system: nausea, vomiting, abdominal pain
Respiratory system: tachypnea, dyspnea
Body as a Whole: anxiety, diaphoresis, lightheadedness, injection site tingling, cyanosis, acidosis, fatigue, weakness, urticaria, generalized numbness and tingling
Severe hypotension, methemoglobinemia, cardiac dysrhythmias, coma and death have been reported in patients without life-threatening cyanide poisoning but who were treated with injection of sodium nitrite at doses less than twice those recommended for the treatment of cyanide poisoning.
- 7 DRUG INTERACTIONS
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Life-sustaining therapy should not be withheld. Cyanide poisoning is a medical emergency in pregnancy, which can be fatal for the pregnant woman and fetus if left untreated (see Clinical Considerations). Therefore, if a pregnant woman has known or suspected cyanide poisoning, Sodium Nitrite Injection for sequential use with Sodium Thiosulfate Injection is recommended [see Indications and Usage (1)]. There are no available data on Sodium Nitrite Injection use in pregnant women to establish a drug-associated risk for major birth defects, miscarriage, or adverse maternal or fetal outcomes. If available, consider alternative therapies not associated with methemoglobinemia.
There are no intravenous animal studies to evaluate the effect of sodium nitrite on embryofetal development. In published animal studies, fetal mortality was reported when pregnant guinea pigs were subcutaneously administered sodium nitrite at 1.7 times the maximum recommended human dose (MRHD) of 450 mg sodium nitrite when maternal and fetal methemoglobin concentrations were at their peak. In other published studies, no evidence of malformations were reported in guinea pigs, mice, or rats; however, severe anemia, reduced growth, and increased pup mortality was reported when pregnant rats were treated with 4.7 times the MRHD of sodium nitrite via drinking water during gestation and throughout lactation (see Data).
Clinical Considerations
Disease-associated maternal and/or embryo/fetal risk
Cyanide readily crosses the placenta. Cyanide poisoning is a medical emergency in pregnancy, which can be fatal for the pregnant woman and fetus if left untreated. Treatment for cyanide poisoning should not be withheld because of potential concerns regarding the effects of Sodium Nitrite Injection on the fetus.
Fetal/neonatal adverse reactions
Sodium nitrite produces methemoglobin. Fetal hemoglobin is oxidized to methemoglobin more easily than adult hemoglobin. In addition, the fetus has lower levels of methemoglobin reductase than adults [see Use in Specific Populations (8.4)]. Based on animal studies, prenatal exposure to sodium nitrite resulted in impaired neural development likely the result of prenatal hypoxia (see Data). If available, consider alternative therapy not known to be associated with methemoglobinemia.
Data
Animal Data
There are no intravenous toxicology studies of sodium nitrite that evaluate the potential for reproductive and developmental toxicity. In a published study, sodium nitrite treatment of pregnant guinea pigs with 60 or 70 mg/kg/day resulted in abortion of the litters within 1-4 days of treatment. All animals treated subcutaneously with 70 mg/kg, sodium nitrite died within 60 minutes of treatment. Further studies demonstrated that a dose of 60 mg/kg resulted in measurable blood levels of methemoglobin in the dams and their fetuses for up to 6 hours post treatment. Maternal methemoglobin levels were higher than the levels in the offspring at all times measured. Based on a body surface area comparison, a 60 mg/kg dose in the guinea pig that resulted in death was only 1.7 times higher than the highest clinical dose of sodium nitrite that would be used to treat cyanide poisoning (based on a body surface area comparison).
In a published study, treatment of pregnant rats via drinking water with sodium nitrite at concentrations of either 2000 or 3000 mg/L during gestation and throughout lactation resulted in severe anemia, reduced growth and increased mortality in the offspring. This exposure regimen in the rat model would result in dosing of approximately 220 and 300 mg/kg/day (4.7 and 6.5 times the highest clinical dose of sodium nitrite that would be used to treat cyanide poisoning, based on a body surface area comparison).
In a published study, treatment of pregnant mice with approximately 243 mg/kg sodium nitrite daily (2.6 times the maximum recommended daily dose of 450 mg/day based on body surface are) via the drinking water (1000 mg/L) from Gestation Day 7-18 did not result in evidence of malformations or embryofetal toxicity or maternal toxicity.
Behavioral and neurodevelopmental studies in rats suggest persistent effects of prenatal exposure to sodium nitrite that were detectable postnatally. Specifically, animals that were exposed prenatally to sodium nitrite (2000 mg/L via drinking water) demonstrated impaired discrimination learning behavior (both auditory and visual) and reduced long-term retention of the passive-avoidance response compared to control animals. Additional studies demonstrated a delay in the development of AchE and 5-HT positive fiber ingrowth into the hippocampal dentate gyrus and parietal neocortex during the first week of life of prenatal nitrite treated pups. These changes have been attributed to prenatal hypoxia following nitrite exposure.
8.2 Lactation
Risk Summary
There are no data on the presence of sodium nitrite in human or animal milk, the effects on the breastfed infant, or the effects on milk production. Cyanide is present in human milk. Because of the potential for serious adverse reactions in the breastfed infant, breastfeeding is not recommended during treatment with Sodium Nitrite Injection. There are no data to determine when breastfeeding may be safely restarted following the administration of Sodium Nitrite Injection.
8.4 Pediatric Use
There are case reports in the medical literature of sodium nitrite in conjunction with sodium thiosulfate being administered to pediatric patients with cyanide poisoning; however, there have been no clinical studies to evaluate the safety or efficacy of sodium nitrite in the pediatric population. As for adult patients, dosing recommendations for pediatric patients have been based on theoretical calculations of antidote detoxifying potential, extrapolation from animal experiments, and a small number of human case reports.
Sodium nitrite must be used with caution in patients less than 6 months of age because they may be at higher risk of developing severe methemoglobinemia compared to older children and adults. The presence of fetal hemoglobin, which is oxidized to methemoglobin more easily than adult hemoglobin, and lower methemoglobin reductase levels compared to older children and adults may contribute to risk.
Mortality attributed to sodium nitrite was reported following administration of an adult dose (300 mg IV followed by a second dose of 150 mg) to a 17-month old child. [see Dosage and Administration (2), Warnings and Precautions, (5), Adverse Reactions (6)]
8.5 Geriatric Use
Sodium nitrite is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
8.6 Renal Impairment
Sodium nitrite is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
-
10 OVERDOSAGE
Large doses of sodium nitrite result in severe hypotension and toxic levels of methemoglobin which may lead to cardiovascular collapse.
Sodium nitrite administration has been reported to cause or significantly contribute to mortality in adults at oral doses as low as 1 g and intravenous doses as low as 600 mg. A death attributed to sodium nitrite has been reported following administration of an adult dose (300 mg IV followed by a second dose of 150 mg) to a 17-month old child.
Cyanosis may become apparent at a methemoglobin level of 10-20%. Other clinical signs and symptoms of sodium nitrite toxicity (anxiety, dyspnea, nausea, and tachycardia) can be apparent at methemoglobin levels as low as 15%. More serious signs and symptoms, including cardiac dysrhythmias, circulatory failure, and central nervous system depression are seen as methemoglobin levels increase, and levels above 70% are usually fatal.
Treatment of overdose involves supplemental oxygen and supportive measures such as exchange transfusion. Treatment of severe methemoglobinemia with intravenous methylene blue has been described in the medical literature; however, this may also cause release of cyanide bound to methemoglobin. Because hypotension appears to be mediated primarily by an increase in venous capacitance, measures to increase venous return may be most appropriate to treat hypotension.
-
11 DESCRIPTION
Sodium nitrite has the chemical name nitrous acid sodium salt. The chemical formula is NaNO2 and the molecular weight is 69.0. The structural formula is:
Structure of Sodium Nitrite
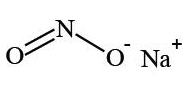
Sodium Nitrite Injection is a cyanide antidote which contains one 10 mL glass vial of a 3% solution of sodium nitrite injection.
Sodium nitrite injection is a sterile aqueous solution and is intended for intravenous injection. Each vial contains 300 mg of sodium nitrite in 10 mL solution (30 mg/mL). Sodium nitrite injection is a clear solution with a pH between 7.0 and 9.0.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Exposure to a high dose of cyanide can result in death within minutes due to the inhibition of cytochrome oxidase resulting in arrest of cellular respiration. Specifically, cyanide binds rapidly with cytochrome a3, a component of the cytochrome c oxidase complex in mitochondria. Inhibition of cytochrome a3 prevents the cell from using oxygen and forces anaerobic metabolism, resulting in lactate production, cellular hypoxia and metabolic acidosis. In massive acute cyanide poisoning, the mechanism of toxicity may involve other enzyme systems as well.
The synergy resulting from treatment of cyanide poisoning with the combination of sodium nitrite and sodium thiosulfate is the result of differences in their primary mechanisms of action as antidotes for cyanide poisoning.
Sodium Nitrite
Sodium nitrite is thought to exert its therapeutic effect by reacting with hemoglobin to form methemoglobin, an oxidized form of hemoglobin incapable of oxygen transport but with high affinity for cyanide. Cyanide preferentially binds to methemoglobin over cytochrome a3, forming the nontoxic cyanomethemoglobin. Methemoglobin displaces cyanide from cytochrome oxidase, allowing resumption of aerobic metabolism. The chemical reaction is as follows:
NaNO2 + Hemoglobin → Methemoglobin
HCN + Methemoglobin → CyanomethemoglobinVasodilation has also been cited to account for at least part of the therapeutic effect of sodium nitrite. It has been suggested that sodium nitrite-induced methemoglobinemia may be more efficacious against cyanide poisoning than comparable levels of methemoglobinemia induced by other oxidants. Also, sodium nitrite appears to retain some efficacy even when the formation of methemoglobin is inhibited by methylene blue.
Sodium Thiosulfate
The primary route of endogenous cyanide detoxification is by enzymatic transulfuration to thiocyanate (SCN-), which is relatively nontoxic and readily excreted in the urine. Sodium thiosulfate is thought to serve as a sulfur donor in the reaction catalyzed by the enzyme rhodanese, thus enhancing the endogenous detoxification of cyanide in the following chemical reaction:
Rhodanese
Na2S2O3 + CN- → SCN- + Na2SO3.12.2 Pharmacodynamics
Sodium Nitrite
When 4 mg/kg sodium nitrite was administered intravenously to six healthy human volunteers, the mean peak methemoglobin concentration was 7%, achieved at 30-60 minutes after injection, consistent with reports in cyanide poisoning victims. Supine systolic and diastolic blood pressures dropped approximately 20% within 10 minutes, a drop which was sustained throughout the 40 minutes of testing. This was associated with a 20 beat per minute increase in pulse rate that returned to baseline in 10 minutes. Five of these subjects were unable to withstand orthostatic testing due to fainting. One additional subject, who received a 12 mg/kg dose of sodium nitrite, experienced severe cardiovascular effects and achieved a peak methemoglobin concentration of 30% at 60 minutes following injection.
Oral doses of 120 to 180 mg of sodium nitrite administered to healthy volunteers caused minimal cardiovascular changes when subjects were maintained in the horizontal position. However, minutes after being placed in the upright position subjects exhibited tachycardia and hypotension with syncope.
The half life for conversion of methemoglobin to normal hemoglobin in a cyanide poisoning victim who has been administered sodium nitrite is estimated to be 55 minutes.
12.3 Pharmacokinetics
Sodium Nitrite
Sodium nitrite is a strong oxidant, and reacts rapidly with hemoglobin to form methemoglobin. The pharmacokinetics of free sodium nitrite in humans have not been well studied. It has been reported that approximately 40% of sodium nitrite is excreted unchanged in the urine while the remaining 60% is metabolized to ammonia and related small molecules.
Cyanide
The apparent terminal elimination half life and volume of distribution of cyanide, in a patient treated for an acute cyanide poisoning with sodium nitrite and sodium thiosulfate administration, have been reported to be 19 hours and 0.41 L/kg, respectively. Additionally, an initial elimination half life of cyanide has been reported to be approximately 1-3 hours.
Thiocyanate
After detoxification, in healthy subjects, thiocyanate is excreted mainly in the urine at a rate inversely proportional to creatinine clearance. In healthy subjects, the elimination half-life and volume of distribution of thiocyanate have been reported to be 2.7 days and 0.25 L/kg, respectively. However, in subjects with renal insufficiency the reported elimination half life is approximately 9 days.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis:
The potential benefit of an acute exposure to sodium nitrite as part of a cyanide antidote outweighs concerns raised by the equivocal findings in chronic rodent studies. Sodium nitrite (0, 750, 1500, or 3000 ppm equivalent to average daily doses of approximately 0, 35, 70, or 130 mg/kg for males and 0, 40, 80, or 150 mg/kg for females) was orally administered to rats (Fischer 344 strain) for 2 years via drinking water. There were no significant increases in the incidence of tumor in either male or female rats. Sodium nitrite (0, 750, 1500, or 3000 ppm equivalent to average daily doses of approximately 0, 60, 120, or 220 mg/kg for males and 0, 45, 90, or 165 mg/kg for females) was administered to B6C3F1 mice for 2 years via the drinking water. Equivocal results were obtained in female mice. Specifically, there was a positive trend toward an increase in the incidence of squamous cell papilloma or carcinoma in the forestomach of female mice. Although the incidence of hyperplasia of the glandular stomach epithelium was significantly greater in the high-dose male mice compared to controls, there were no significant increases in tumors in the male mice. Numerous reports in the published literature indicate that sodium nitrite may react in vivo with secondary amines to form carcinogenic nitrosamines in the stomach. Concurrent exposure to sodium nitrite and secondary amines in feed or drinking water resulted in an increase in the incidence of tumors in rodents.
Mutagenesis:
Sodium nitrite is mutagenic in S. typhimurium strains TA100, TA1530, TA1535 with and without metabolic activation; however, it was negative in strain TA98, TA102, DJ460 and E. coli strain WP2UVRA/PKM101. Sodium nitrite has been reported to be genotoxic to V79 hamster cells in vitro and in the mouse lymphoma assay, both assays conducted in the absence of metabolic activation. Sodium nitrite was negative in the in vitro chromosomal aberrations assay using human peripheral blood lymphocytes. Acute administration of sodium nitrite to male rats or male mice did not produce an increased incidence of micronuclei in bone marrow. Likewise, sodium nitrite administration to mice for 14-weeks did not result in an increase in the incidence of micronuclei in the peripheral blood.
Impairment of Fertility:
Multigenerational fertility and reproduction studies conducted by the National Toxicology Program did not detect any evidence of an effect of sodium nitrite (0.0, 0.06, 0.12, and 0.24% weight/volume) on either fertility or any reproductive parameter in Swiss CD-1 mice. This treatment protocol resulted in approximate doses of 125, 260, and 425 mg/kg/day. The highest exposure in this mouse study is 4.6 times greater than the highest clinical dose of sodium nitrite that would be used to treat cyanide poisoning (based on a body surface area comparison).
-
14 CLINICAL STUDIES
Human Data
The human data supporting the use of sodium thiosulfate for cyanide poisoning consists primarily of published case reports. There are no randomized controlled clinical trials. Nearly all the human data describing the use of sodium thiosulfate report its use in conjunction with sodium nitrite. Dosing recommendations for humans have been based on theoretical calculations of antidote detoxifying potential, extrapolation from animal experiments, and a small number of human case reports.
There have been no human studies to prospectively and systematically evaluate the safety of sodium thiosulfate or sodium nitrite in humans. Available human safety information is based largely on anecdotal case reports and case series of limited scope.
Animal Data (Cyanide Poisoning)
THE EFFECTIVENESS OF SODIUM THIOSULFATE AND SODIUM NITRITE FOR THE TREATMENT OF ACUTE CYANIDE POISONING HAS NOT BEEN STUDIED IN HUMANS IN ADEQUATE AND WELL-CONTROLLED CLINICAL TRIALS BECAUSE INDUCING THE CONDITION IN HUMANS TO STUDY THE DRUG'S EFFICACY IS NOT ETHICAL.
Due to the extreme toxicity of cyanide, experimental evaluation of treatment efficacy has predominantly been completed in animal models. The efficacy of sodium thiosulfate treatment alone to counteract the toxicity of cyanide was initially reported in 1895 by Lang. The efficacy of amyl nitrite treatment in cyanide poisoning of the dog model was first reported in 1888 by Pedigo. Further studies in the dog model, which demonstrated the utility of sodium nitrite as a therapeutic intervention, were reported in 1929 by Mladoveanu and Gheorghiu. However, Hugs and Chen et al. independently reported upon the superior efficacy of the combination of sodium nitrite and sodium thiosulfate in 1932-1933. Treatment consisted of intravenously administered 22.5 mg/kg (half the lethal dose) sodium nitrite or 1 g/kg sodium thiosulfate alone or in sequence immediately after subcutaneous injection of sodium cyanide into dogs over a range of doses. Subsequent doses of 10 mg/kg sodium nitrite and/or 0.5 g/kg sodium thiosulfate were administered when clinical signs or symptoms of poisoning persisted or reappeared. Either therapy administered alone increased the dose of sodium cyanide required to cause death, and when administered together, sodium nitrite and sodium thiosulfate resulted in a synergistic effect in raising the lethal dose of sodium cyanide. The combined therapy appeared to have reduced efficacy when therapy was delayed until signs of poisoning (e.g. convulsions) appeared; however, other investigators have reported survival in dogs that were administered antidotal treatment after respiratory arrest had occurred.
Animal studies conducted in other species (e.g., rat, guinea pig, sheep, pigeon and cat) have also supported a synergistic effect of intravenous sodium nitrite and sodium thiosulfate in the treatment of cyanide poisoning.
While intravenous injection of sodium nitrite and sodium thiosulfate was effective in reversing the effects of lethal doses of cyanide in dogs, intramuscular injection of sodium nitrite, with or without sodium thiosulfate, was found not to be effective in the same setting.
- 16 HOW SUPPLIED/STORAGE AND HANDLING
-
17 PATIENT COUNSELING INFORMATION
Sodium Nitrite Injection is indicated for cyanide poisoning and in this setting, patients will likely be unresponsive or may have difficulty in comprehending counseling information.
Hypotension and Methemoglobin Formation
When feasible, patients should be informed of the possibility of life-threatening hypotension and methemoglobin formation.
Monitoring
Where feasible, patients should be informed of the need for close monitoring of blood pressure and oxygenation.
Lactation
Advise women that breastfeeding is not recommended during treatment with Sodium Nitrite Injection [see Use in Specific Populations (8.2)].
- SPL UNCLASSIFIED SECTION
-
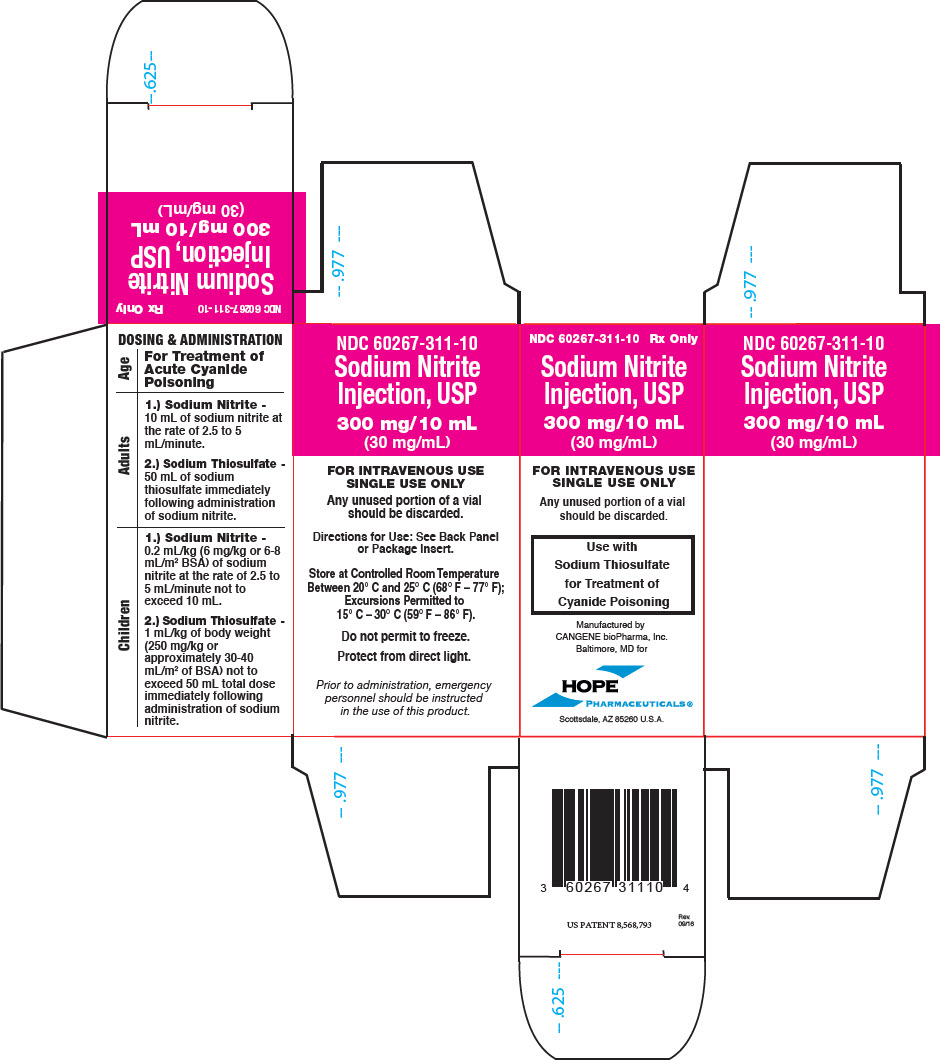
PRINCIPAL DISPLAY PANEL - 10 mL Vial Carton
NDC 60267-311-10
Rx OnlySodium Nitrite
Injection, USP
300 mg/10 mL
(30 mg/mL)FOR INTRAVENOUS USE
SINGLE USE ONLYAny unused portion of a vial
should be discarded.Use with
Sodium Thiosulfate
for Treatment of
Cyanide PoisoningManufactured by
CANGENE bioPharma, Inc.
Baltimore, MD forHOPE
PHARMACEUTICALS ®Scottsdale, AZ 85260 U.S.A.
-
INGREDIENTS AND APPEARANCE
SODIUM NITRITE
sodium nitrite injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:60267-311 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Sodium Nitrite (UNII: M0KG633D4F) (Nitrite Ion - UNII:J39976L608) Sodium Nitrite 30 mg in 1 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Nitrogen (UNII: N762921K75) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:60267-311-10 10 mL in 1 VIAL, SINGLE-USE; Type 0: Not a Combination Product 02/14/2012 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA203922 02/14/2012 Labeler - Hope Pharmaceuticals (015227945) Establishment Name Address ID/FEI Business Operations Curia Wisconsin, Inc. 004670501 API MANUFACTURE(60267-311) Establishment Name Address ID/FEI Business Operations Cangene Biopharma, Inc. 050783398 MANUFACTURE(60267-311)