PHYTONADIONE- phytonadione tablet
Oceanside Pharmaceuticals
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use PHYTONADIONE TABLETS safely and effectively. See full prescribing information for PHYTONADIONE TABLETS.
PHYTONADIONE tablets, for oral use Initial U.S. Approval: 1995 INDICATIONS AND USAGEPhytonadione tablets are a vitamin K replacement indicated for the treatment of adults with the following coagulation disorders which are due to faulty formation of factors II, VII, IX and X when caused by vitamin K deficiency or interference with vitamin K activity:
DOSAGE AND ADMINISTRATIONDOSAGE FORMS AND STRENGTHSTablets: 5 mg (3) CONTRAINDICATIONS
ADVERSE REACTIONSMost common adverse reactions are transient “flushing sensations”, “peculiar” sensations of taste and instances of dizziness, rapid and weak pulse, profuse sweating, brief hypotension, dyspnea, and cyanosis. (6) To report SUSPECTED ADVERSE REACTIONS, contact Oceanside Pharmaceuticals at 1-800-321-4576 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. DRUG INTERACTIONS
See 17 for PATIENT COUNSELING INFORMATION. Revised: 7/2021 |
FULL PRESCRIBING INFORMATION: CONTENTS*1 INDICATIONS AND USAGE2 DOSAGE AND ADMINISTRATION2.1 Dosing Considerations2.2 Recommended Dosage3 DOSAGE FORMS AND STRENGTHS4 CONTRAINDICATIONS6 ADVERSE REACTIONS7 DRUG INTERACTIONS8 USE IN SPECIFIC POPULATIONS8.1 Pregnancy8.2 Lactation8.4 Pediatric Use8.5 Geriatric Use11 DESCRIPTION12 CLINICAL PHARMACOLOGY12.1 Mechanism of Action12.2 Pharmacodynamics12.3 Pharmacokinetics13 NONCLINICAL TOXICOLOGY13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility16 HOW SUPPLIED/STORAGE AND HANDLING17 PATIENT COUNSELING INFORMATION
|
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
Phytonadione tablets are indicated for the treatment of adults with the following coagulation disorders which are due to faulty formation of factors II, VII, IX and X when caused by vitamin K deficiency or interference with vitamin K activity.
- •
- anticoagulant-induced hypoprothrombinemia caused by coumarin or indanedione derivatives;
- •
- hypoprothrombinemia secondary to antibacterial therapy;
- •
- hypoprothrombinemia secondary to factors limiting absorption or synthesis of vitamin K, e.g., obstructive jaundice, biliary fistula, sprue, ulcerative colitis, celiac disease, intestinal resection, cystic fibrosis of the pancreas, and regional enteritis;
- •
- Other drug-induced hypoprothrombinemia where it is definitely shown that the result is due to interference with vitamin K metabolism, e.g., salicylates.
2 DOSAGE AND ADMINISTRATION
2.1 Dosing Considerations
Avoid the oral route when the clinical disorder would prevent proper absorption. Bile salts must be given with the tablets when the endogenous supply of bile to the gastrointestinal tract is deficient. The coagulant effects of phytonadione tablets are not immediate; improvement of international normalized ratio (INR) may take 1-8 hours. Interim use of whole blood or component therapy may also be necessary if bleeding is severe.
Phytonadione tablets will not counteract the anticoagulant action of heparin.
When phytonadione tablets are used to correct excessive anticoagulant-induced hypoprothrombinemia, anticoagulant therapy still being indicated, the patient is again faced with the clotting hazards existing prior to starting the anticoagulant therapy. Phytonadione tablets are not a clotting agent, but overzealous therapy with vitamin K1 may restore conditions which originally permitted thromboembolic phenomena. Dosage should be kept as low as possible, and prothrombin time should be checked regularly as clinical conditions indicate.
2.2 Recommended Dosage
Anticoagulant-Induced Prothrombin Deficiency in Adults
The recommended dose to correct excessively prolonged prothrombin times caused by oral anticoagulant therapy is, 2.5 to 10 mg or up to 25 mg initially. In some instances 50 mg may be required. Frequency and amount of subsequent doses should be determined by prothrombin time response or clinical condition. If, in 12 to 48 hours after oral administration, the prothrombin time has not been shortened satisfactorily, repeat the dose.
Repeated large doses of phytonadione tablets are not warranted in liver disease if the response to initial use of the vitamin is unsatisfactory. Failure to respond to phytonadione tablets may indicate a congenital coagulation defect or that the condition being treated is unresponsive to vitamin K.
Hypoprothrombinemia Due to Other Causes in Adults
If possible, discontinuation or reduction of the dosage of drugs interfering with coagulation mechanisms (such as salicylates, antibiotics) is suggested as an alternative to administering concurrent phytonadione tablets. The severity of the coagulation disorder should determine whether the immediate administration of phytonadione tablets is required in addition to discontinuation or reduction of interfering drugs.
The recommended dose is 2.5 to 25 mg or more (sometimes up to 50 mg). Evaluate INR after 6-8 hours, and repeat dose if INR remains prolonged. Modify subsequent dosage (amount and frequency) based upon the INR or clinical condition.
3 DOSAGE FORMS AND STRENGTHS
Tablets: 5 mg, clean, pale yellow, semi-glossy, round, flat, beveled edge, scored and debossed with “VRX” above “405” on one side and debossed with “MEPHYTON” on the other side.
4 CONTRAINDICATIONS
Phytonadione tablets are contraindicated in patients with a history of a hypersensitivity reaction to phytonadione or inactive ingredients [see Description (11)].
6 ADVERSE REACTIONS
The following adverse reactions associated with the use of parenteral phytonadione were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Severe hypersensitivity reactions, including anaphylactoid reactions and deaths, have been reported following parenteral administration. The majority of these reported events occurred following intravenous administration.
Transient “flushing sensations” and “peculiar” sensations of taste have been observed with parenteral phytonadione, as well as instances of dizziness, rapid and weak pulse, profuse sweating, brief hypotension, dyspnea, and cyanosis.
Hyperbilirubinemia has been observed in the newborn following administration of parenteral phytonadione. This has occurred primarily with doses above those recommended.
7 DRUG INTERACTIONS
Anticoagulants
Phytonadione tablets may induce temporary resistance to prothrombin-depressing anticoagulants, especially when larger doses of phytonadione tablets are used. Should this occur, higher doses of anticoagulant therapy may be needed when resuming anticoagulant therapy, or a change in therapy to a different class of anticoagulant may be necessary (i.e., heparin sodium).
Phytonadione tablets do not affect the anticoagulant action of heparin.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Published studies with the use of phytonadione during pregnancy have not reported a clear association with phytonadione and adverse developmental outcomes [see Data]. There are maternal and fetal risks associated with vitamin K deficiency during pregnancy [see Clinical Considerations]. Animal reproduction studies have not been conducted with phytonadione.
The estimated background risk for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Clinical Considerations
Disease-associated maternal and/or embryo/fetal risk
Pregnant women with vitamin K deficiency hypoprothrombinemia may be at increased risk for bleeding diatheses during pregnancy and hemorrhagic events at delivery. Subclinical vitamin K deficiency during pregnancy has been implicated in rare cases of fetal intracranial hemorrhage.
Data
Human Data
Phytonadione has been measured in cord blood of infants whose mothers were treated with phytonadione during pregnancy in concentrations lower than seen in maternal plasma. Administration of vitamin K1 to pregnant women shortly before delivery increased both maternal and cord blood concentrations. Published data do not report a clear association with phytonadione and adverse maternal or fetal outcomes when used during pregnancy. However, these studies cannot definitively establish the absence of any risk because of methodologic limitations including small sample size and lack of blinding.
Animal Data
In pregnant rats receiving vitamin K1 orally, fetal plasma and liver concentrations increased following administration, supporting placental transfer.
8.2 Lactation
Risk Summary
Phytonadione is present in breastmilk. There are no data on the effects of phytonadione tablets on the breastfed child or on milk production. The developmental and health benefits of breastfeeding should be considered along with the clinical need for phytonadione tablets and any potential adverse effects on the breastfed child from phytonadione tablets or from the underlying maternal condition.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established with phytonadione tablets. Hemolysis, jaundice, and hyperbilirubinemia in newborns, particularly in premature infants, have been reported with vitamin K.
8.5 Geriatric Use
Clinical studies of phytonadione tablets did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
11 DESCRIPTION
Phytonadione is a vitamin K replacement, which is a clear, yellow to amber, viscous, and nearly odorless liquid. It is insoluble in water, soluble in chloroform and slightly soluble in ethanol. It has a molecular weight of 450.7.
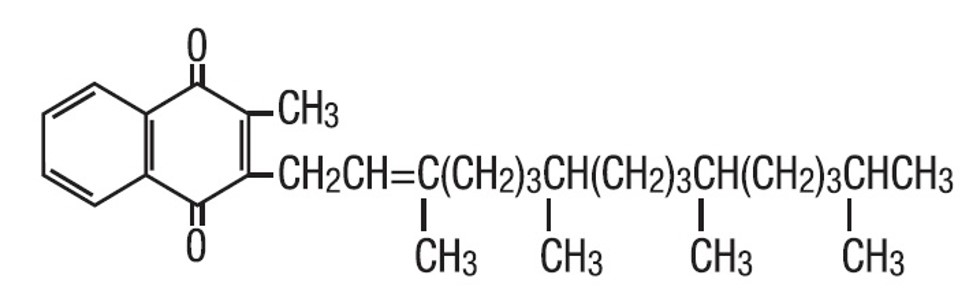
Phytonadione is 2-methyl-3-phytyl-1, 4-naphthoquinone. Its empirical formula is C31H46O2 and its structural formula is:
Phytonadione Tablets for oral administration contain 5 mg of phytonadione and are clean, pale yellow, semi-glossy, round, flat, beveled edge, scored and debossed with “VRX” above “405” on one side and debossed with “MEPHYTON” on the other side. Inactive ingredients are acacia, calcium phosphate, colloidal silicon dioxide, lactose, magnesium stearate, starch, and talc.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Phytonadione tablets possess the same type and degree of activity as does naturally-occurring vitamin K, which is necessary for the production via the liver of active prothrombin (factor II), proconvertin (factor VII), plasma thromboplastin component (factor IX), and Stuart factor (factor X). The prothrombin test is sensitive to the levels of three of these four factors – II, VII, and X. Vitamin K is an essential cofactor for a microsomal enzyme that catalyzes the posttranslational carboxylation of multiple, specific, peptide-bound glutamic acid residues in inactive hepatic precursors of factors II, VII, IX, and X. The resulting gamma-carboxyglutamic acid residues convert the precursors into active coagulation factors that are subsequently secreted by liver cells into the blood.
In normal animals and humans, phytonadione is virtually devoid of pharmacodynamic activity. However, in animals and humans deficient in vitamin K, the pharmacological action of vitamin K is related to its normal physiological function, that is, to promote the hepatic biosynthesis of vitamin K-dependent clotting factors.
12.3 Pharmacokinetics
Absorption
Oral phytonadione is adequately absorbed from the gastrointestinal tract only if bile salts are present.
Distribution
After absorption, phytonadione is initially concentrated in the liver, but the concentration declines rapidly. Very little vitamin K accumulates in tissues.
Elimination
Little is known about the metabolic fate of vitamin K. Almost no free unmetabolized vitamin K appears in bile or urine.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies of carcinogenicity or impairment of fertility have not been performed with phytonadione tablets. Phytonadione tablets at concentrations up to 2,000 mcg/plate, with or without metabolic activation, were negative in the Ames microbial mutagen test.
16 HOW SUPPLIED/STORAGE AND HANDLING
Phytonadione Tablets, 5 mg, are clean, pale yellow, semi-glossy, round, flat, beveled edge, scored and debossed with “VRX” above “405” on one side and debossed with “MEPHYTON” on the other side. They are supplied as follows:
NDC 68682-170-30 bottles of 30.
Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Always protect phytonadione tablets from light. Store in tightly closed original container and carton until contents have been used.
17 PATIENT COUNSELING INFORMATION
Vitamin K1 is fairly rapidly degraded by light; therefore, advise patients to always protect phytonadione tablets from light. Store phytonadione tablets in closed original carton until contents have been used [see How Supplied/Storage and Handling (16)].
Distributed by:
Oceanside Pharmaceuticals, a division of Bausch Health US, LLC
Bridgewater, NJ 08807 USA
Manufactured by:
Bausch Health Companies Inc.
Steinbach, MB R5G 1Z7, Canada
MEPHYTON is a trademark of Bausch Health Companies Inc. or its affiliates.
© 2021 Bausch Health Companies Inc. or its affiliates
9561504
20003221
| PHYTONADIONE
phytonadione tablet |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Oceanside Pharmaceuticals (832011691) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Bausch Health Companies Inc. | 253292734 | MANUFACTURE(68682-170) | |