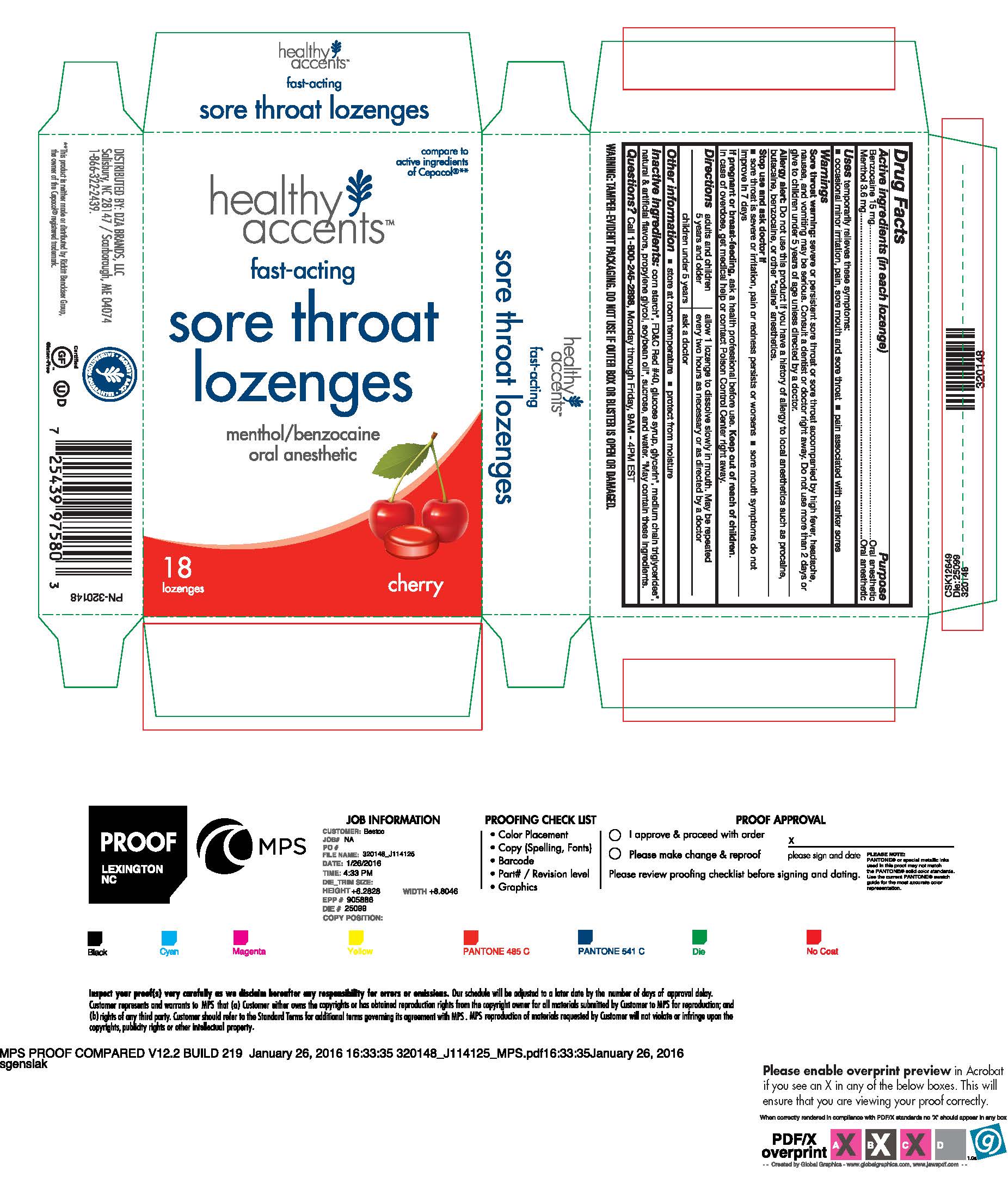
CHERRY SORE THROAT LOZENGES- benzocaine lozenge
DZA Brands LLC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Benzocaine 15 mg
Menthol 3.6 mg
Uses temporarily relieves these symptoms:
occasional minor irritation, pain, sore mouth and sore throat
pain associated with canker sores
Warnings
Sore throat warning: severe of persistent sore throat or sore throat accompanied by high fever, headache, nausea, and vomiting may be serious. Consult a dentist or doctor right away. Do not use more than 2 days or give to children under 5 years of age unless directed by a doctor.
Allergy alert: Do not use this product if you have a history of allergy to local anesthetics such as procaine, butacaine, benzocaine, or other "caine" anesthetics.
Stop use and ask doctor if
sore throat is severe or irritation, pain or redness persists or worsens
sore mouth symptoms do not improve in 7 days
Directions
adults and children 5 years and older allow 1 lozenge to dissolve slowly in mouth. May be repeated every two hours as necessary or as directed by a doctor.
children under 5 years ask a doctor
| CHERRY SORE THROAT LOZENGES
benzocaine lozenge |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
 |
||||||||||||||||||
|
||||||||||||||||||
| Labeler - DZA Brands LLC (090322194) |
| Registrant - BestCo Inc. (002149136) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| BestCo Inc. | 002149136 | manufacture(55316-027) | |