Label: P32 SODIUM PHOSPHATE injection, suspension
-
Contains inactivated NDC Code(s)
NDC Code(s): 51808-127-01 - Packager: AnazaoHealth Corporation
- Category: HUMAN PRESCRIPTION DRUG LABEL
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated June 19, 2012
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- DESCRIPTION
- INDICATIONS AND USAGE
- CONTRAINDICATIONS
- WARNINGS
- DOSAGE AND ADMINISTRATION
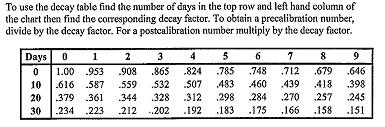
- DECAY CHART
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
P32 SODIUM PHOSPHATE
p32 sodium phosphate injection, suspensionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:51808-127 Route of Administration INTRA-ARTICULAR, INTRAPERITONEAL, INTERSTITIAL, INTRAPLEURAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHOSPHORUS P-32 (UNII: 690284A407) (PHOSPHORUS P-32 - UNII:690284A407) PHOSPHORUS P-32 5 mCi in 1 mL Inactive Ingredients Ingredient Name Strength DEXTROSE (UNII: IY9XDZ35W2) 0.3 g in 1 mL BENZYL ALCOHOL (UNII: LKG8494WBH) 0.02 mL in 1 mL SODIUM ACETATE (UNII: 4550K0SC9B) 1 mg in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51808-127-01 3 mL in 1 VIAL Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Unapproved drug other 06/19/2012 Labeler - AnazaoHealth Corporation (011038762) Establishment Name Address ID/FEI Business Operations AnazaoHealth Corporation 011038762 MANUFACTURE(51808-127)