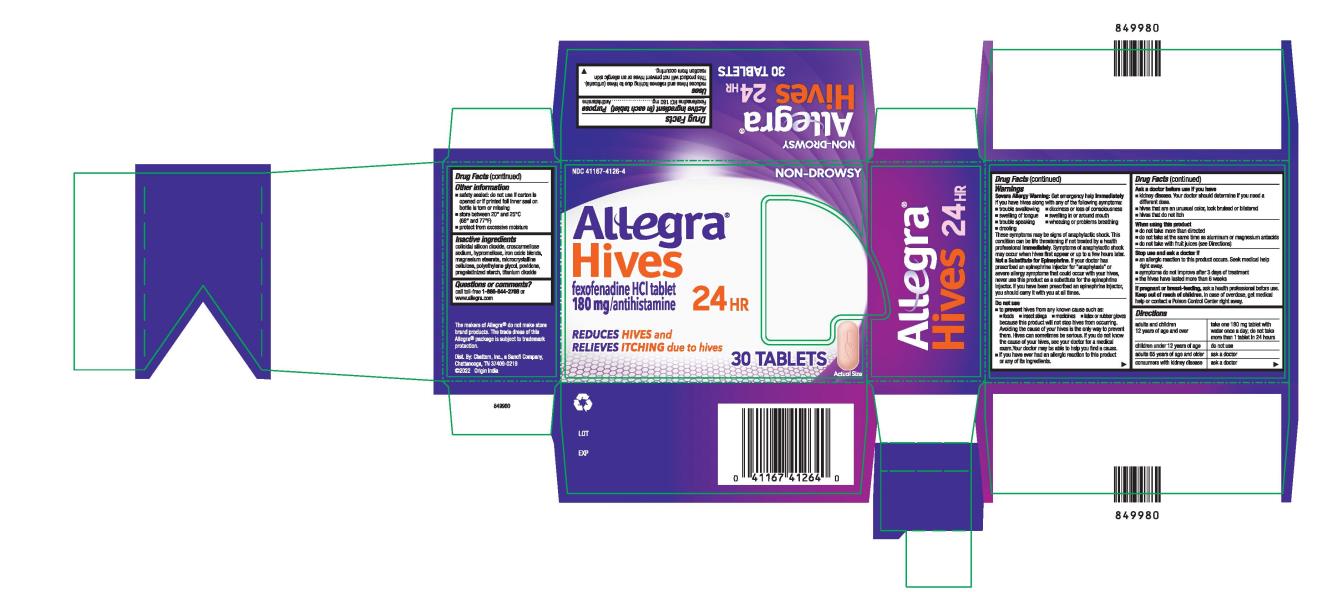
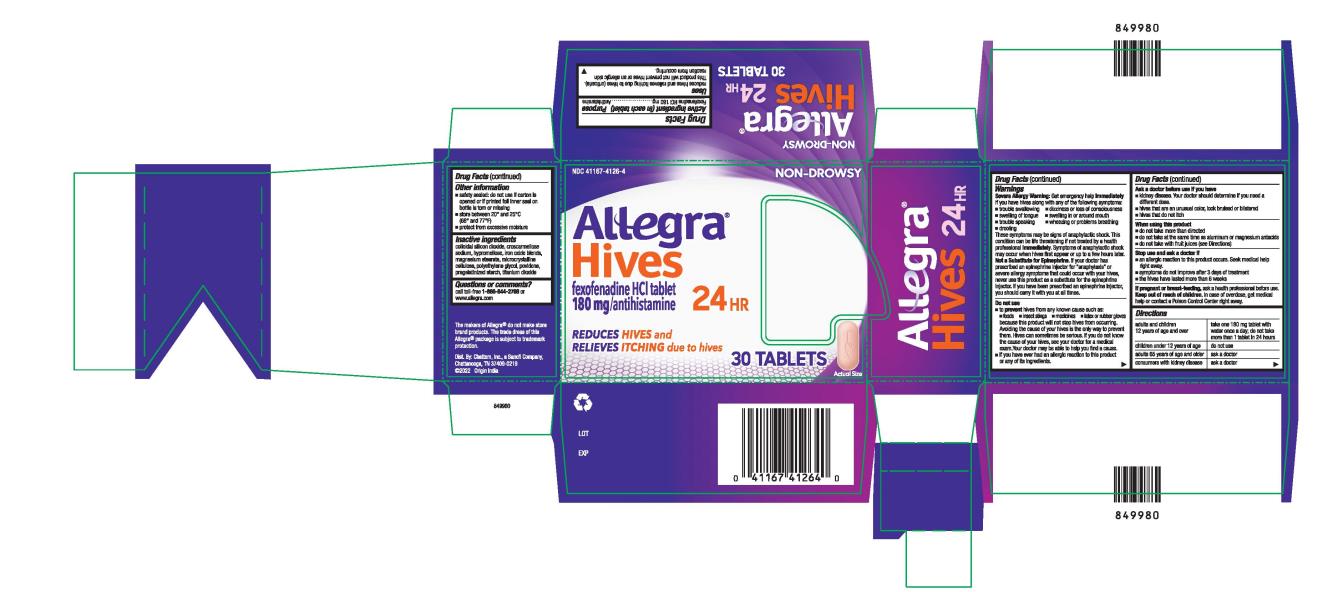
Label: ALLEGRA HIVES 24HR- fexofenadine hydrochloride tablet, film coated
- NDC Code(s): 41167-4126-3, 41167-4126-4, 41167-4126-8
- Packager: Chattem, Inc.
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated April 2, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient (in each tablet)
- Purpose
- Uses
-
Warnings
Severe Allergy Warning: Get emergency help immediately if you have hives along with any of the following symptoms:
■ trouble swallowing ■ dizziness or loss of consciousness ■ swelling of tongue
■ swelling in or around mouth ■ trouble speaking ■ wheezing or problems breathing ■ drooling
These symptoms may be signs of anaphylactic shock. This condition can be life threatening if not treated by a health professional immediately. Symptoms of anaphylactic shock may occur when hives first appear or up to a few hours later.
Not a Substitute for Epinephrine. If your doctor has prescribed an epinephrine injector for “anaphylaxis” or severe allergy symptoms that could occur with your hives, never use this product as a substitute for the epinephrine injector. If you have been prescribed an epinephrine injector, you should carry it with you at all times.
Do not use
■ to prevent hives from any known cause such as:
■ foods ■ insect stings ■ medicines ■ latex or rubber gloves because this product will not stop hives from occurring. Avoiding the cause of your hives is the only way to prevent them. Hives can sometimes be serious. If you do not know the cause of your hives, see your doctor for a medical exam. Your doctor may be able to help you find a cause.
■ if you have ever had an allergic reaction to this product or any of its ingredients.
Ask a doctor before use if you have
■ kidney disease. Your doctor should determine if you need a different dose.
■ hives that are an unusual color, look bruised or blistered
■ hives that do not itch
When using this product
■ do not take more than directed
■ do not take at the same time as aluminum or magnesium antacids
■ do not take with fruit juices (see Directions)
- Directions
-
Other information
■(if blister) safety sealed: do not use if carton is opened or if individual blister units are torn or opened
■(on the carton if bottle) safety sealed: do not use if carton is opened or if printed foil inner seal on bottle is torn or missing
■(if bottle) safety sealed: do not use if carton was opened or if printed foil inner seal on bottle is torn or missing
■ store between 20° and 25°C (68° and 77°F)
■ protect from excessive moisture
- Inactive ingredients
- Questions or comments?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ALLEGRA HIVES 24HR
fexofenadine hydrochloride tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:41167-4126 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FEXOFENADINE HYDROCHLORIDE (UNII: 2S068B75ZU) (FEXOFENADINE - UNII:E6582LOH6V) FEXOFENADINE HYDROCHLORIDE 180 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) BROWN IRON OXIDE (UNII: 1N032N7MFO) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POVIDONE (UNII: FZ989GH94E) STARCH, CORN (UNII: O8232NY3SJ) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color orange (peach) Score no score Shape OVAL Size 17mm Flavor Imprint Code 018;E Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:41167-4126-8 1 in 1 CARTON 06/06/2022 06/07/2022 1 5 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC:41167-4126-3 1 in 1 CARTON 01/31/2024 2 15 in 1 BOTTLE; Type 0: Not a Combination Product 3 NDC:41167-4126-4 1 in 1 CARTON 06/06/2022 3 30 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA020872 06/06/2022 Labeler - Chattem, Inc. (003336013)