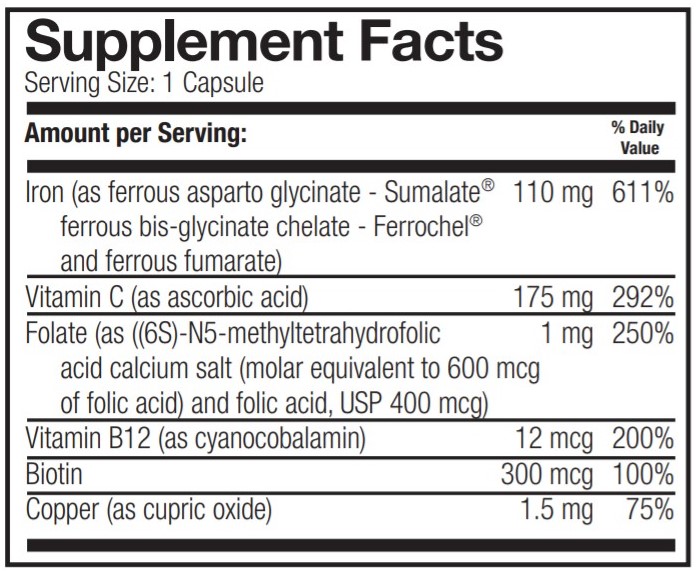
FERIVA FA- ferrous bisglycinate, ferrous asparto glycinate, ferrous fumarate, ascorbic acid, folic acid, cyanocobalamin, biotin, cupric oxide and docusate sodium capsule, gelatin coated
Avion Pharmaceuticals, LLC
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
FeRiva FA ® 75854-0319-30
OTHER INGREDIENTS: Silicon dioxide and magnesium stearate. Gel capsule: (bovine gelatin, titanium dioxide, FD&C blue #1 and FD&C red #40).
INDICATIONS: FeRiva FA ® is a multivitamin/multimineral dietary supplement indicated for use in improving the nutritional status of patients with iron deficiency.
CONTRAINDICATIONS: FeRiva FA ® is contraindicated in patients with a known hypersensitivity to any of the ingredients. Hemochromatosis and hemosiderosis are contraindicated to iron therapy.
WARNING: Accidental overdose of iron-containing products is a leading cause of fatal poisoning in children under 6. Keep this product out of reach of children. In case of accidental overdose, call a doctor or poison control center immediately.
PRECAUTIONS: Folic acid alone is improper therapy in the treatment of pernicious anemia and other megaloblastic anemias where Vitamin B12 is deficient. Folic acid in doses above 1.0 mg daily may obscure pernicious anemia in that hematologic remission can occur while neurological manifestations progress. While prescribing this nutritional supplement for pregnant women, nursing mothers or for women prior to conception, their medical condition and other drugs, herbs and/or supplements consumption should be considered.
ADVERSE REACTIONS: Allergic sensitization has been reported following both oral and parenteral administration of folic acid.
DOSAGE AND ADMINISTRATION: Adults, one (1) capsule daily, with or without food, or as prescribed by your healthcare provider. Do not administer to children under the age of 12.
HOW SUPPLIED: In child-resistant bottles of 30 capsules (75854-319-30). The listed product number is not a National Drug Code, but has instead been formatted to comply with standard industry practice for pharmacy computer systems.
STORAGE: Store at 20° - 25°C (68° - 77°F); excursions permitted to 15° - 30°C (59° - 86°F) [See USP Controlled Room Temperature.]
MANUFACTURED FOR:
Avion Pharmaceuticals, LLC
Alpharetta, GA 30022
1-888-61-AVION
Rev. 0915-01
Sumalate ® is a registered trademark of Albion Laboratories, Inc., covered by one or more claims of U.S. Patent Nos. 6,716,814, 8,007,846, and 8,425,956.
Ferrochel ® is a registered trademark of Albion Laboratories, Inc. covered by U.S. Patents 6,716,814 & 7,838,042.
THESE STATEMENTS HAVE NOT BEEN EVALUATED BY THE FOOD AND DRUG ADMINISTRATION. THIS PRODUCT IS NOT INTENDED TO DIAGNOSE, TREAT, CURE OR PREVENT ANY DISEASE.
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
| FERIVA FA
ferrous bisglycinate, ferrous asparto glycinate, ferrous fumarate, ascorbic acid, folic acid, cyanocobalamin, biotin, cupric oxide and docusate sodium capsule, gelatin coated |
|||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||
| Labeler - Avion Pharmaceuticals, LLC (040348516) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Avion Pharmaceuticals, LLC | 040348516 | manufacture(75854-319) | |