Label: FOLLTROPIN (follicle stimulating hormone- porcine injection, powder, for solution
- NDC Code(s): 17030-102-70
- Packager: Vetoquinol USA, Inc.
- Category: PRESCRIPTION ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Animal Drug Application
Drug Label Information
Updated October 8, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- CAUTION:
-
DESCRIPTION:
FOLLTROPIN® (porcine pituitary-derived follicle stimulating hormone for injection) is a powder for solution containing 700 IU (equivalent to 400 mg NIH-FSH-P1) of follicle stimulating hormone (FSH) per vial. FOLLTROPIN® contains a low amount of luteinizing hormone (less than 1000 μg NIH-LH-S19 per vial). When reconstituted according to label directions the final solution contains 35 IU FSH per mL for intramuscular injection. There are no inactive ingredients in the FSH vial.
- INDICATIONS FOR USE:
-
DOSAGE AND ADMINISTRATION:
For intramuscular injection only.
Reconstitute each vial of FOLLTROPIN® with 20 mL bacteriostatic sodium chloride injection, using strict aseptic technique. Do not use if clumps or particles are visible after gentle mixing. Once reconstituted, FOLLTROPIN® should be stored refrigerated at 2-8°C (36-46°F) for no longer than 4 days.
Regimen: Start injections on day 8 to 10 following observed or induced estrus. Administer 2.5 mL (87.5 IU1) of FOLLTROPIN® intramuscularly, twice daily at 12 hour intervals, for 4 consecutive days. In conjunction with the 6th dose of FOLLTROPIN®, administer an FDA-approved prostaglandin product (cloprostenol sodium or dinoprost tromethamine) for cattle, using the labeled dosage and administration instructions to cause luteolysis and induce estrus.
Inseminate animals at 12 and 24 hours after the onset of estrus or 60 and 72 hours after prostaglandin treatment. Additional inseminations may be conducted at 12 hour intervals, if indicated.
Collection of embryos is normally started on day 7 following insemination.
- 1
- IU = International Units (established by WHO Expert Committee on Biological Standardization in 1986).
-
CONTRAINDICATIONS:
Do not use in pregnant cattle. Laboratory studies with FSH in rats and rabbits have shown evidence of embryotoxicity/fetotoxicity and the safety of the product has not been assessed in pregnant cows. Do not use FOLLTROPIN® in cows that are known to be hypersensitive to the active ingredient, porcine pituitary-derived follicle stimulating hormone.
-
WARNINGS AND PRECAUTIONS:
Withdrawal Period:
Withdrawal Period Neither a withdrawal period nor a milk discard time is required when this product is used according to label directions. User Safety Warnings:
Not for use in humans. Keep out of reach of children. Care should be taken when handling the product to avoid accidental self-injection. Accidental self-injection may cause biological effects in women and, if pregnant, to the unborn child. In the event of accidental self-injection seek medical attention immediately by consulting a physician/health professional, particularly in women who are pregnant, or whose pregnancy status is unknown. To obtain a Safety Data Sheet or to report adverse reactions, contact 1-800-835-9496 and make this number available to the physician/health professional.
Animal Safety Warnings and Precautions:
Management considerations: Successful use of FOLLTROPIN® depends on good reproductive health of the cow and quality herd management procedures. Cattle should be clinically healthy and cycling normally. A comprehensive and ongoing nutritional, reproductive, and herd health program should be in place when using FOLLTROPIN® for superovulation.
The use of this product may result in trim loss of edible tissue at slaughter.
- ADVERSE REACTIONS:
-
TARGET ANIMAL SAFETY:
Existing pharmacology and toxicology information and knowledge gained from the widespread clinical use of FOLLTROPIN® in cattle was sufficient to demonstrate that FOLLTROPIN® is not systemically toxic to the cow and the targets of toxicity in both cattle and other species are limited to the reproductive organs and functions. A field safety study demonstrated that the use of FOLLTROPIN® in accordance with label directions did not produce any reactions at the injection site. A retrospective study of dairy cattle superovulated up to six times at intervals of less than 93 days did not show any adverse effect of repeated superovulation on the yield of transferable embryos. Cows within the superovulation program produced embryos that were successfully transferred into recipient cows and were able to themselves conceive and deliver normal calves. The development of cystic ovaries following treatment with FOLLTROPIN® has been reported; however, these results have not been substantiated by well-controlled studies or through a comprehensive literature review.
-
EFFECTIVENESS:
The effectiveness of FOLLTROPIN® for the induction of superovulation in beef and dairy heifers and cows was substantiated through a systematic review and meta-analysis which included 2685 superovulation records obtained from twenty clinical studies and one retrospective study. The analysis revealed a high level of variability in the number of transferable embryos obtained between treatment groups; such variability is representative of the expected variation for superovulation programs. The estimated mean number of transferable embryos was 4.5 per study animal and the 95% confidence limits of the mean were 3.5 (lower limit) and 5.6 (upper limit).
-
STORAGE, HANDLING, AND DISPOSAL:
Freeze-dried powder vials: Do not store above 25°C (77°F).
Reconstituted solution: Store under refrigeration 2-8°C (36-46°F).
Keep the vials in the outer carton in order to protect from light.
Shelf life following reconstitution according to directions: 4 days
Do not freeze after reconstitution.
Discard any unused portion of the reconstituted FOLLTROPIN® solution.
See FDA's website http://www.fda.gov/safesharpsdisposal for information on safe disposal of needles and other sharps.
Unless otherwise directed, dispose of unused medicine and empty containers in solid waste following applicable Federal, State and local environmental laws and regulations.
-
HOW SUPPLIED:
FOLLTROPIN®DUAL PACK contains two 20 mL vials each containing 700 IU1 FSH as a lyophilized powder.
-
CONTACT INFORMATION:
Contact Vetoquinol USA, Inc. at 1-800-267-5707 for customer service or to obtain product information, including a SDS. After hours or to report adverse reactions, call 1-800-835-9496.
For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888-FDA-VETS or online at http://www.fda.gov/AnimalVeterinary/SafetyHealth.
- SPL UNCLASSIFIED SECTION
-
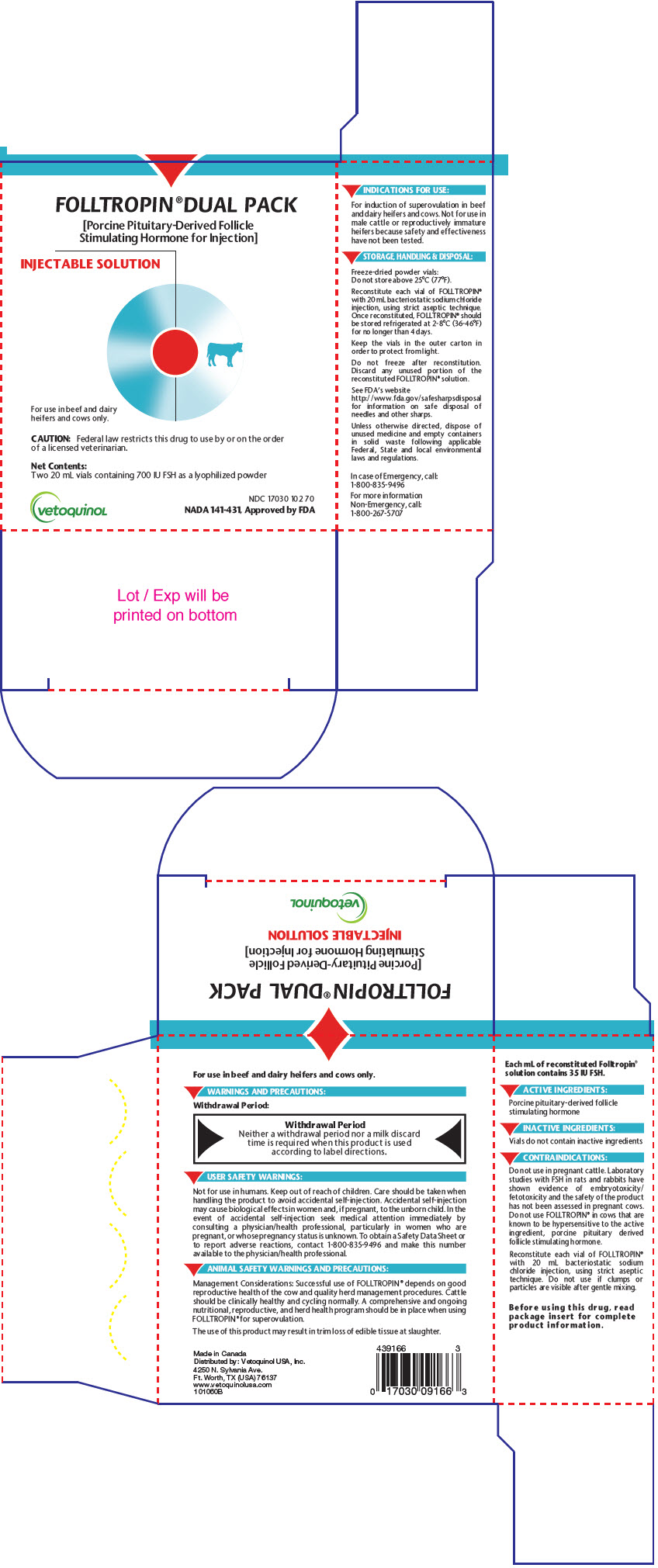
PRINCIPAL DISPLAY PANEL - 20 mL Vial Carton
FOLLTROPIN®DUAL PACK
[Porcine Pituitary-Derived Follicle
Stimulating Hormone for Injection]INJECTABLE SOLUTION
For use in beef and dairy
heifers and cows only.CAUTION: Federal law restricts this drug to use by or on the order
of a licensed veterinarian.Net Contents:
Two 20 mL vials containing 700 IU FSH as a lyophilized powdervetoquinoL
NDC 17030 102 70
NADA 141-431, Approved by FDA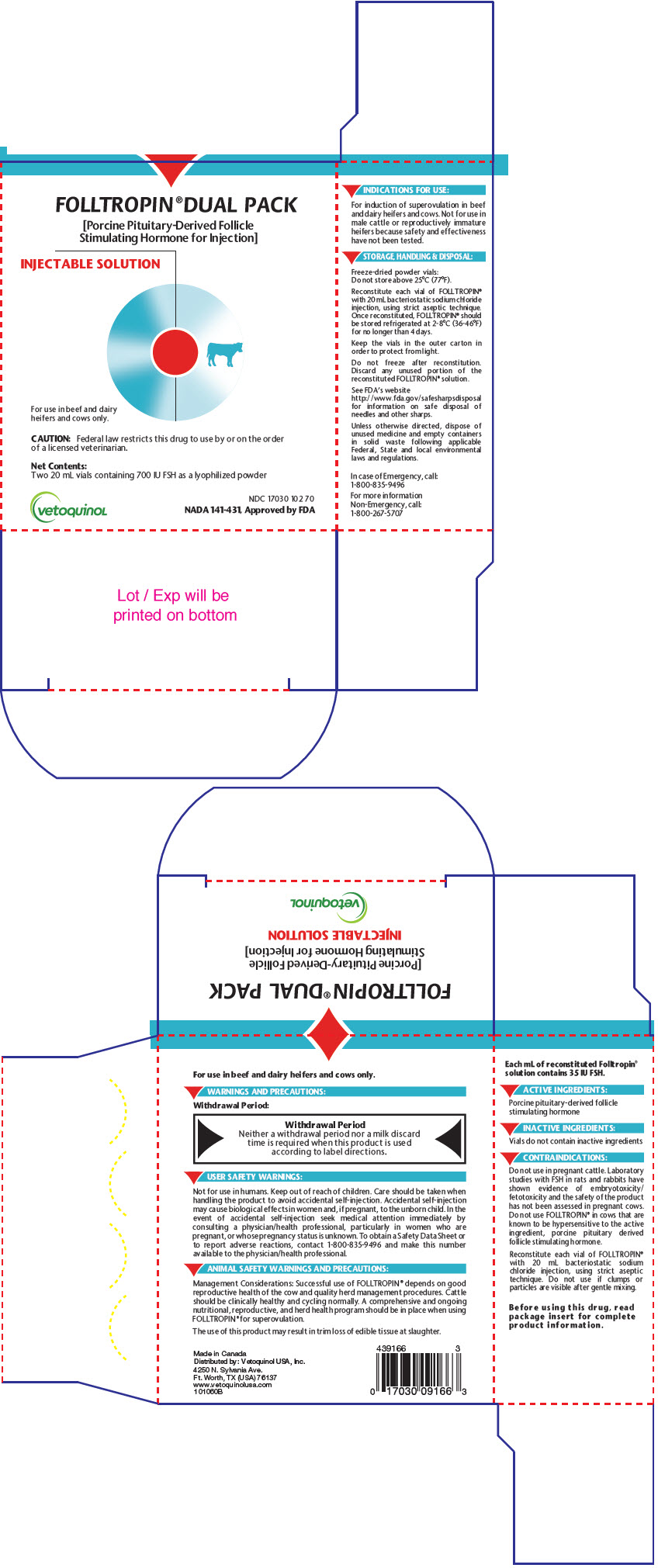
-
INGREDIENTS AND APPEARANCE
FOLLTROPIN
follicle stimulating hormone (porcine) injection, powder, for solutionProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:17030-102 Route of Administration INTRAMUSCULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Follicle Stimulating Hormone (Porcine) (UNII: 8FYM5179QJ) (Follitropin - UNII:076WHW89TW) Follicle Stimulating Hormone (Porcine) 700 [iU] Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:17030-102-70 2 in 1 CARTON 1 1 in 1 VIAL, MULTI-DOSE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141431 01/23/2017 Labeler - Vetoquinol USA, Inc. (106824209) Establishment Name Address ID/FEI Business Operations Vetoquinol N.-A. 202717443 API MANUFACTURE, ANALYSIS, LABEL, PACK Establishment Name Address ID/FEI Business Operations Jubilant HollisterStier General Partnership 246762764 MANUFACTURE, ANALYSIS Establishment Name Address ID/FEI Business Operations Vetoquinol N.-A. INC 202919940 MANUFACTURE, ANALYSIS, LABEL, PACK Establishment Name Address ID/FEI Business Operations Nucro-Technics 241266845 ANALYSIS Establishment Name Address ID/FEI Business Operations Neopharm Labs Inc. 243379372 ANALYSIS