CLARO OTIC SOLUTION- (florfenicol, terbinafine, mometasone furoate) solution
Bayer HealthCare, LLC Animal Health Division
----------
CLARO®
(florfenicol, terbinafine, mometasone furoate)
CAUTION:
Federal (U.S.A.) law restricts this drug to use by or on the order of a licensed veterinarian.
DESCRIPTION:
CLARO® contains 16.6 mg/mL florfenicol, 14.8 mg/mL terbinafine (equivalent to 16.6 mg/mL terbinafine hydrochloride) and 2.2 mg/mL mometasone furoate.
Inactive ingredients include purified water, propylene carbonate, propylene glycol, ethyl alcohol, and polyethylene glycol.
INDICATIONS:
CLARO® is indicated for the treatment of otitis externa in dogs associated with susceptible strains of yeast (Malassezia pachydermatis) and bacteria (Staphylococcus pseudintermedius).
DOSAGE AND ADMINISTRATION:
Shake before use.
CLARO® should be administered by veterinary personnel.
Administer one dose (1 dropperette) per affected ear. The duration of effect should last 30 days.
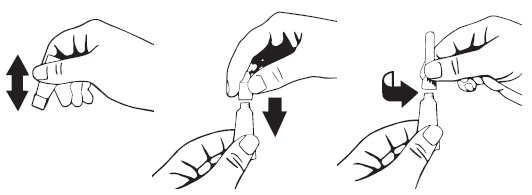
- 1.
- Clean and dry the external ear canal before administering the product.
- 2.
- Verify the tympanic membrane is intact prior to administration.
- 3.
- Remove single dose dropperette from the package.
- 4.
- While holding the dropperette in an upright position, remove the cap from the dropperette.
- 5.
- Turn the cap over and push the other end of the cap onto the tip of the dropperette.
- 6.
- Twist the cap to break the seal and then remove cap from the dropperette.
- 7.
- Screw the applicator nozzle onto the dropperette.
- 8.
- Insert the tapered tip of the dropperette into the affected external ear canal and squeeze to instill the entire contents (1 mL) into the affected ear.
- 9.
- Gently massage the base of the ear to allow distribution of the solution.
- 10.
- Repeat with other ear as prescribed.
Cleaning the ear after dosing may affect product effectiveness.
CONTRAINDICATIONS:
Do not use in dogs with known tympanic membrane perforation (see PRECAUTIONS).
CLARO® is contraindicated in dogs with known or suspected hypersensitivity to florfenicol, terbinafine hydrochloride, or mometasone furoate.
WARNINGS:
Human Warnings: Not for use in humans. Keep this and all drugs out of reach of children. In case of accidental ingestion by humans, contact a physician immediately. In case of accidental skin contact, wash area thoroughly with water. Avoid contact with eyes.
Humans with known hypersensitivity to florfenicol, terbinafine hydrochloride, or mometasone furoate should not handle this product.
PRECAUTIONS:
Do not administer orally.
The use of CLARO® in dogs with perforated tympanic membranes has not been evaluated. The integrity of the tympanic membrane should be confirmed before administering the product. Reevaluate the dog if hearing loss or signs of vestibular dysfunction are observed during treatment.
Use of topical otic corticosteroids has been associated with adrenocortical suppression and iatrogenic hyperadrenocorticism in dogs (see ANIMAL SAFETY).
Use with caution in dogs with impaired hepatic function (see ANIMAL SAFETY).
The safe use of CLARO® in dogs used for breeding purposes, during pregnancy, or in lactating bitches has not been evaluated.
ADVERSE REACTIONS:
In a field study conducted in the United States (see EFFECTIVENESS), there were no directly attributable adverse reactions in 146 dogs administered CLARO®.
To report suspected adverse drug events and/or obtain a copy of the Safety Data Sheet (SDS) or for technical assistance, contact Bayer HealthCare at 1-800-422-9874.
For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888-FDA-VETS or online at http://www.fda.gov/AnimalVeterinary/SafetyHealth.
PHARMACOLOGY:
CLARO® Otic Solution is a fixed combination of three active substances: florfenicol (antibacterial), terbinafine (antifungal), and mometasone furoate (steroidal anti-inflammatory). Florfenicol is a bacteriostatic antibiotic which acts by inhibiting protein synthesis. Terbinafine is an antifungal which selectively inhibits the early synthesis of ergosterol.
Mometasone furoate is a glucocorticosteroid with anti-inflammatory activity.
MICROBIOLOGY:
The compatibility and additive effect of each of the components in CLARO® solution was demonstrated in a component effectiveness and non-interference study. An in vitro study of organisms collected from clinical cases of otitis externa in dogs enrolled in the clinical effectiveness study determined that florfenicol and terbinafine hydrochloride inhibit the growth of bacteria and yeast commonly associated with otitis externa in dogs. No consistent synergistic or antagonistic effect of the two antimicrobials was demonstrated. The addition of mometasone furoate to the combination did not impair antimicrobial activity to any clinically significant extent.
In a field study (see EFFECTIVENESS), at least 10 isolates from successfully treated cases were obtained for S. pseudintermedius and M. pachydermatis.
EFFECTIVENESS:
In a well-controlled, double-masked field study, CLARO® was evaluated against a vehicle control in 221 dogs with otitis externa. One hundred and forty six dogs were treated with CLARO® and 75 dogs were treated with the vehicle control. All dogs were evaluated for safety. Treatment (1 mL) was administered once on Day 0 to the affected ear(s).
Prior to treatment, the ear(s) was cleaned with saline. The dogs were evaluated on Days 0, 7, 14, and 30. Blood work and urinalysis were obtained on Day 0 pre-treatment and Day 30 at study completion. Four clinical signs associated with otitis externa were evaluated: erythema, exudate, swelling, and ulceration. Success was based on clinical improvement at Day 30. Of the 183 dogs included in the effectiveness evaluation, 72.5% of dogs administered CLARO® solution were successfully treated, compared to 11.1% of the dogs in the vehicle-control group (p=0.0001).
ANIMAL SAFETY:
In a target animal safety study, CLARO® was administered aurally to 12-week-old Beagle puppies (4 dogs/sex/group) at 0X, 1X, 3X, and 5X the recommended dose once every 2 weeks for a total dosing period of 28 days (3 times the treatment duration). No clinically relevant treatment-related findings were noted in hearing tests, body weight, weight gain, or food consumption. CLARO® administration was associated with post-treatment ear wetness or clear aural exudate, increased absolute neutrophil count, decreased absolute lymphocyte and eosinophil counts, suppression of the adrenal cortical response to ACTH-stimulation, decreased adrenal weight and atrophy of the adrenal cortex, increased liver weight with hepatocellular enlargement/cytoplasmic change, and decreased thymus weight. Other potentially treatment-related effects included mild changes to AST, total protein, inorganic phosphorus, creatinine, and calcium.
STORAGE INFORMATION:
Store between 20ºC-25ºC (68ºF-77ºF), excursions are permitted 15ºC-30ºC (59ºF-86ºF).
HOW SUPPLIED:
CLARO® solution is supplied in a single-use dropperette in a blister. Each dropperette contains one 1 mL dose.
CLARO® is available in cartons of two, ten, or twenty dropperettes.
Bayer, the Bayer Cross and CLARO are registered trademarks of Bayer.
©2018 Bayer
Manufactured for
Bayer HealthCare LLC
Animal Health Division
P.O. Box 390, Shawnee Mission, Kansas 66201 USA
Made in Germany
NADA 141-440, Approved by FDA.
February, 2018
86384450 LV1802
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL
CLARO®
(florfenicol, terbinafine, mometasone furoate)
Otic Solution
For otic use in dogs only
CAUTION: Federal (U.S.A.) law restricts this drug to use by or on the order of a licensed veterinarian.
NADA 141-440, Approved by FDA
Net Contents: 10 x 1 mL single dose dropperettes
| CLARO
OTIC SOLUTION
(florfenicol, terbinafine, mometasone furoate) solution |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| Labeler - Bayer HealthCare, LLC Animal Health Division (152266193) |