Label: SODIUM FLUORIDE solution/ drops
- NDC Code(s): 58657-322-50
- Packager: Method Pharmaceuticals, LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated December 10, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
Description
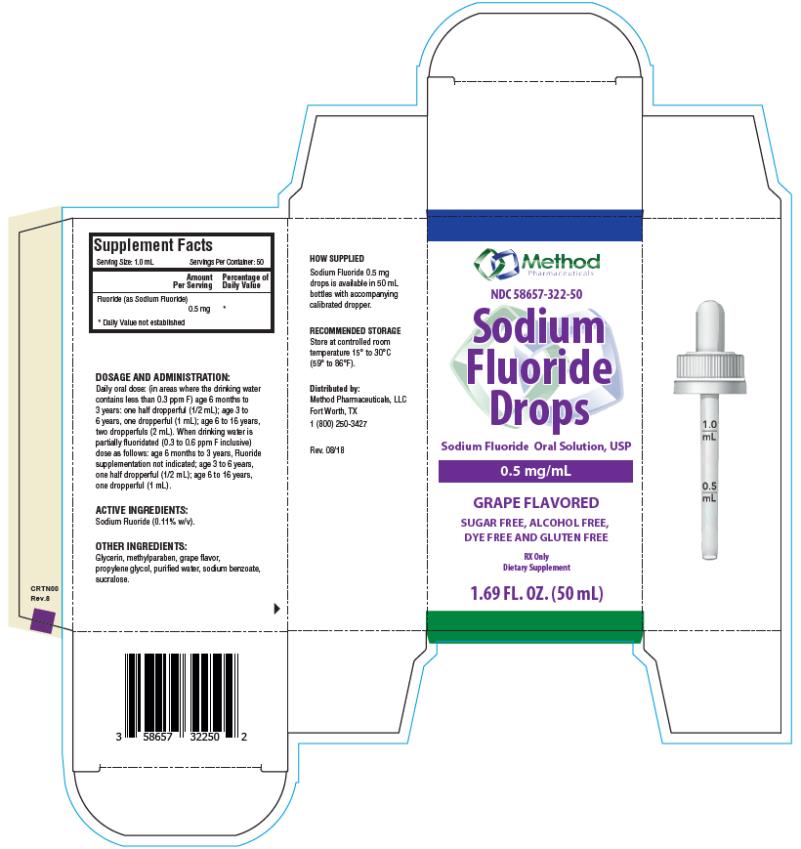
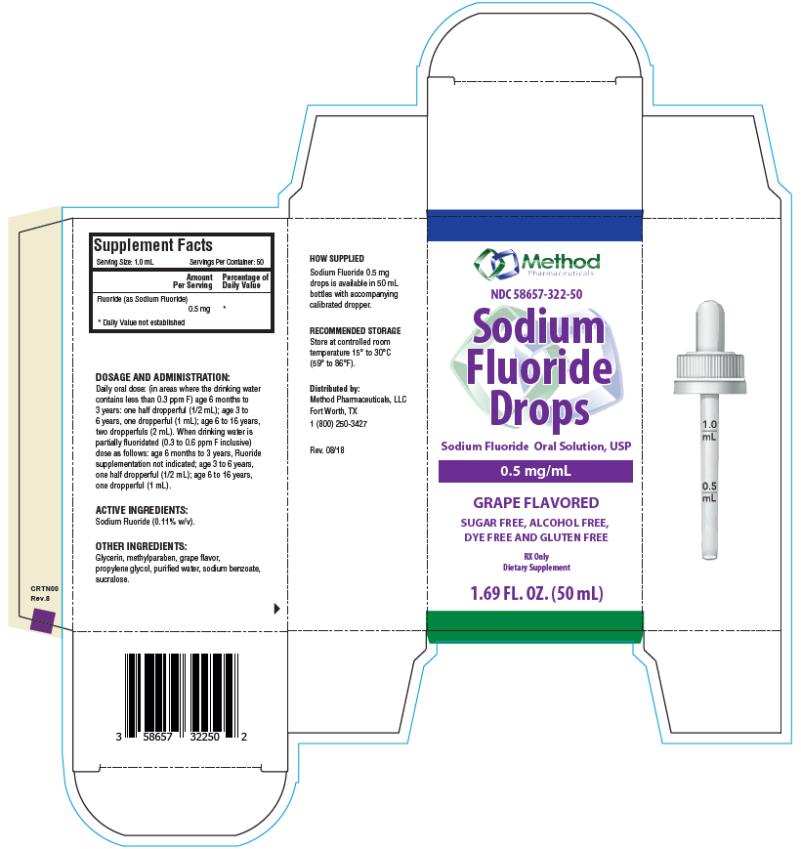
Each mL of Sodium Fluoride Drops contains 0.5 mg Fluoride ion (F) from 1.1 mg Sodium Fluoride (NaF). For use as a dental caries preventive in pediatric patients. Sugar Free, Alcohol Free, Dye Free and Gluten Free.
Supplement Facts
Serving Size: 1 mL
Servings Per Container: 50
Amount per serving % Daily Value
Fluoride (as Sodium Fluoride) 0.5 mg **
** Daily Value not established.
Active Ingredients: Sodium Fluoride (0.11% w/v).
Other Ingredients: Glycerin, methylparaben, grape flavor, propylene glycol, purified water, sodium benzoate, sucralose.
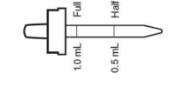
FLUORIDE SUPPLEMENT DOSAGE SCHEDULES AGE Fluoride Ion Level in Drinking Water (ppm)* < 0.3 ppm 0.3 - 0.6 ppm > 0.6 ppm Birth to 6 months None None None 6 months to 3 years Half dropperful 0.25 mg F (1/2 mL) None None 3 to 6 years One dropperful 0.5 mg F (1 mL)† Half dropperful 0.25 mg F (1/2 mL) None 6 to 16 years Two dropperfuls 1 mg F (2 mL) One dropperful 0.5 mg F (1 mL) None * 1.0 ppm = 1 mg/Liter
† 1.1 mg Sodium Fluoride contains 0.5 mg Fluoride ion
Fluoride Supplement Dose Schedule approved by the American Dental Association, American Academy of Pediatrics and American Academy of Pediatric Dentistry.
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
- Clinical Pharmacology
-
Indications and Usage
As a supplemental source of Fluoride. It has been established that ingestion of fluoridated drinking water (1 ppm F) during the period of tooth development results in significant decrease in the incidence of dental caries.1 Sodium Fluoride Drops were developed to provide systemic Fluoride for use as a supplement
in pediatric patients from 6 months to age 3 and older, living in areas where the drinking water Fluoride level does not exceed 0.6 ppm F.
- Contraindications
-
Warnings
Prolonged daily ingestion of quantities greater than the recommended amount may result in various degrees of dental fluorosis in pediatric patients under age 6 years, especially if the water fluoridation exceeds 0.6 ppm. Read directions carefully before using. Keep out of the reach of infants and children.
-
Precautions
See " Overdosage" section. Incompatibility of Fluoride with dairy foods has been reported due to formation of Calcium Fluoride which is poorly absorbed. Not for use in the eyes.
- Adverse Reactions
-
Overdosage
Prolonged daily ingestion of excessive Fluoride may result in varying degrees of dental fluorosis. The total amount of Sodium Fluoride in a bottle of 50 mL (0.5 mg/mL) Sodium Fluoride Drops (25 mg F) conforms with the recommendations of the American Dental Association for the maximum to be dispensed at one time for safety purposes. If overdose is suspected, call 1-800-222-1222 (American Association of Poison Control Centers), your local poison control center (www.aapcc.org), or emergency room immediately for treatment recommendations.
-
Dosages and Administration
Daily oral dose: (in areas where the drinking water contains less than 0.3 ppm F) age 6 months to 3 years: one half dropperful (1/2 mL); age 3 to 6 years, one dropperful (1 mL); age 6 to 16 years, two dropperfuls (2 mL). When drinking water is partially fluoridated (0.3 to 0.6 ppm F inclusive) dose as follows: age 6 months to 3 years, Fluoride supplementation not indicated; age 3 to 6 years, one half dropperful (1/2 mL); age 6 to 16 years, one dropperful (1 mL).
- How Supplied
- References
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SODIUM FLUORIDE
sodium fluoride solution/ dropsProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:58657-322 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM FLUORIDE (UNII: 8ZYQ1474W7) (FLUORIDE ION - UNII:Q80VPU408O) FLUORIDE ION 0.5 mg in 1 mL Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SODIUM BENZOATE (UNII: OJ245FE5EU) SUCRALOSE (UNII: 96K6UQ3ZD4) Product Characteristics Color Score Shape Size Flavor GRAPE Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58657-322-50 50 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 08/15/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 08/15/2018 Labeler - Method Pharmaceuticals, LLC (060216698)