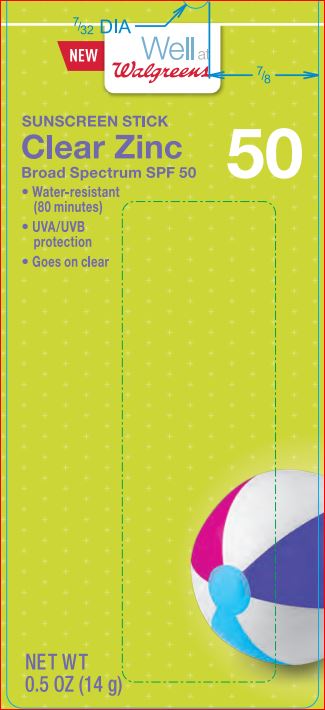
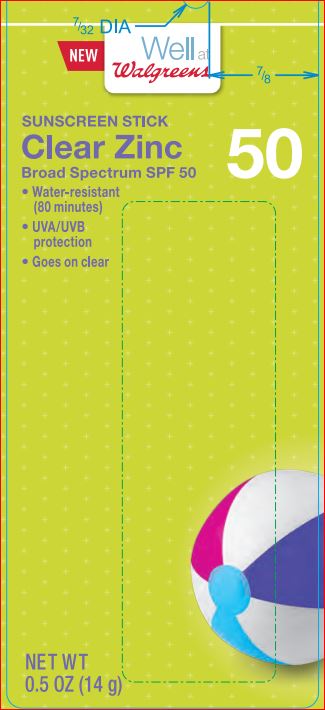
Label: CREAL ZINK SUNSCREEN SPF 50 WELL AT WALGREENS- octinoxate 7.5% octisalate 5.0% zinc oxide 14.5% stick
-
Contains inactivated NDC Code(s)
NDC Code(s): 0363-1117-01 - Packager: Walgreens
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 20, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Warnings
For external use only
Flammable: Do not use near heat, flame, or while smoking.
Do not use on damaged or broken skin
When using this product • Keep out of eyes. Rinse eyes with water to
remove. • Keep away from face to avoid breathing it • Do not puncture or
incinerate. Contents under pressure. Do not store at temperatures above 120ºF.
Stop use and ask a doctor if rash occurs - KEEP OUT OF REACH OF CHILDREN
-
DOSAGE & ADMINISTRATION
Directions
• spray liberally and spread evenly by hand 15 minutes before sun exposure
• reapply: • after 80 minutes of swimming or sweating
• immediately after towel drying • at least every 2 hours
• hold container 4 to 6 inches from the skin to apply
• do not spray directly into face. Spray on hands then apply to face.
• do not apply in windy conditions • use in a well-ventilated area
• Sun Protection Measures. Spending time in the sun increases your risk
of skin cancer and early skin aging. To decrease this risk, regularly use a
sunscreen with a broad spectrum SPF of 15 or higher and other sun
protection measures including:
• limit time in the sun, especially from 10 a.m. – 2 p.m.
• wear long-sleeve shirts, pants, hats, and sunglasses
• children under 6 months: Ask a doctor -
INACTIVE INGREDIENT
Inactive ingredients
Aloe Barbadensis Leaf Extract
Ascorbic Acid
C12-15 Alkyl Benzoate
Camellia Sinensis Leaf Extract
Copernicia Cerifera (Carnauba) Wax
Dimethicone
Helianthus Annuus (Sunflower) Seed Oil
Hydrogenated Microcrystalline Wax
Hydrogenated Polyisobutene
Octyldodecanol
Paraffin
PEG-8 Dimethicone
Polyethylene
Retinyl Palmitate
Silica
Sodium Propoxyhydroxypropyl Thiosulfate Silica
Synthetic Wax
Tocopheryl Acetate
Triethoxycaprylylsilane - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CREAL ZINK SUNSCREEN SPF 50 WELL AT WALGREENS
octinoxate 7.5% octisalate 5.0% zinc oxide 14.5% stickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0363-1117 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Octinoxate (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) Octinoxate 7.5 g in 100 g Octisalate (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) Octisalate 5 g in 100 g Zinc Oxide (UNII: SOI2LOH54Z) (ZINC CATION - UNII:13S1S8SF37) ZINC CATION 14.5 g in 100 g Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) GREEN TEA LEAF (UNII: W2ZU1RY8B0) CARNAUBA WAX (UNII: R12CBM0EIZ) Dimethicone (UNII: 92RU3N3Y1O) SUNFLOWER OIL (UNII: 3W1JG795YI) Octyldodecanol (UNII: 461N1O614Y) PARAFFIN (UNII: I9O0E3H2ZE) PEG-8 Dimethicone (UNII: GIA7T764OD) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) VITAMIN A PALMITATE (UNII: 1D1K0N0VVC) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM PROPOXYHYDROXYPROPYL THIOSULFATE SILICA (UNII: 208G222332) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) Triethoxycaprylylsilane (UNII: LDC331P08E) ASCORBIC ACID (UNII: PQ6CK8PD0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0363-1117-01 14 g in 1 TUBE; Type 0: Not a Combination Product 12/07/2015 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 12/07/2015 Labeler - Walgreens (008965063) Registrant - Product Quest Mfg (927768135) Establishment Name Address ID/FEI Business Operations Product Quest Mfg 927768135 manufacture(0363-1117) , label(0363-1117)