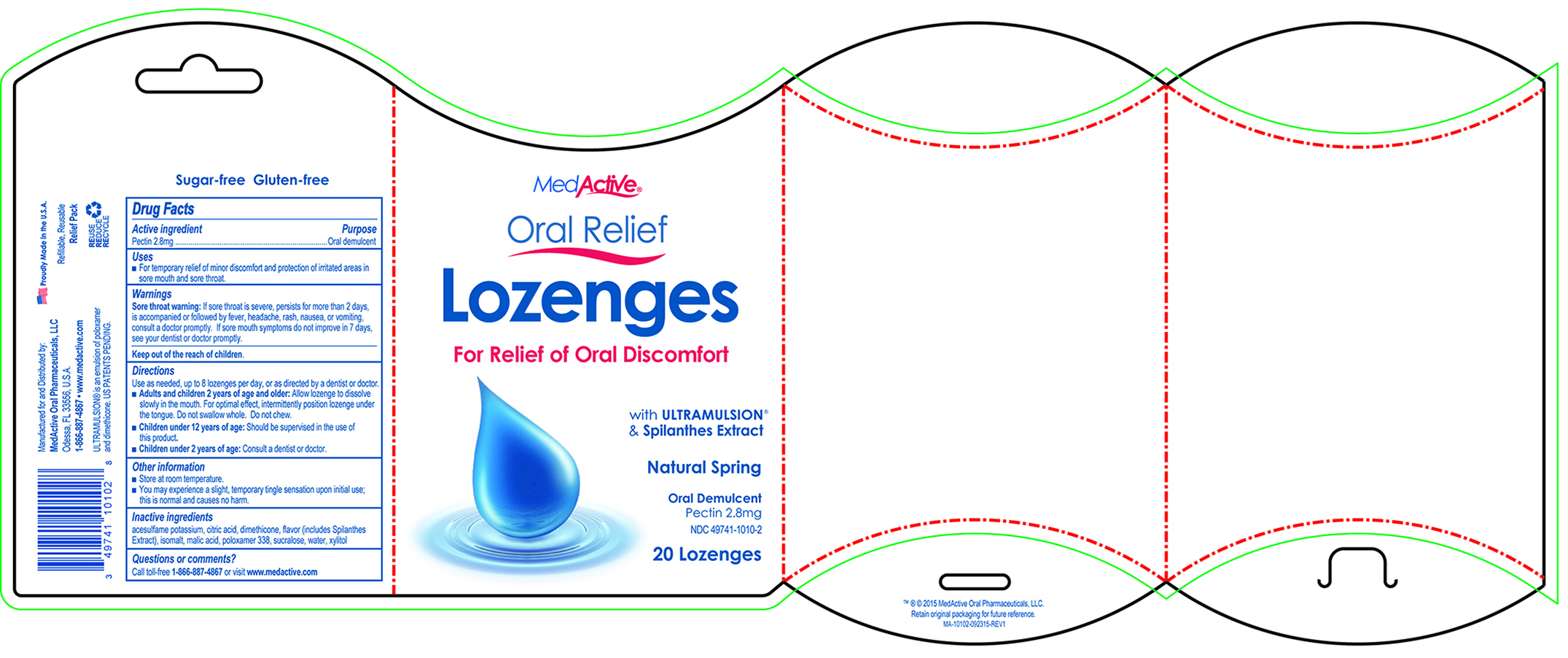
Label: MEDACTIVE ORAL RELIEF- lozenge lozenge
-
Contains inactivated NDC Code(s)
NDC Code(s): 49741-1010-1, 49741-1010-2 - Packager: Integrate Oral Care, LLC.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 30, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- KEEP OUT OF REACH OF CHILDREN
-
DOSAGE & ADMINISTRATION
Directions
Use as needed, up to 8 lozenges per day, or as directed by a dentist or doctor.
- Adults and children 2 years of age and older: Allow lozenge to dissolve slowly in the mouth. For optimal effect, intermittently position lozenge under the tongue. Do not swallow whole. Do not chew.
- Children under 12 years of age: Should be supervised in the use of this product.
- Children under 2 years of age: Consult a dentist or doctor.
- OTHER SAFETY INFORMATION
- INACTIVE INGREDIENT
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MEDACTIVE ORAL RELIEF
lozenge lozengeProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49741-1010 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PECTIN (UNII: 89NA02M4RX) (PECTIN - UNII:89NA02M4RX) PECTIN 2.8 mg Inactive Ingredients Ingredient Name Strength DIMETHICONE (UNII: 92RU3N3Y1O) XYLITOL (UNII: VCQ006KQ1E) POLOXAMER 338 (UNII: F75JV2T505) SUCRALOSE (UNII: 96K6UQ3ZD4) WATER (UNII: 059QF0KO0R) ACESULFAME POTASSIUM (UNII: 23OV73Q5G9) ISOMALT (UNII: S870P55O2W) CITRIC ACID ACETATE (UNII: DSO12WL7AU) MALIC ACID (UNII: 817L1N4CKP) Product Characteristics Color white (Opaque White) Score no score Shape OVAL Size 22mm Flavor FRUIT (NATURAL SPRING) Imprint Code None Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49741-1010-2 20 in 1 BOX 08/31/2015 1 NDC:49741-1010-1 1 in 1 CELLO PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part356 08/31/2015 Labeler - Integrate Oral Care, LLC. (063540457) Registrant - Integrate Oral Care, LLC. (063540457)