Label: GAVILAX- polyethylene glycol 3350 powder, for solution
- NDC Code(s): 43386-312-07, 43386-312-08, 43386-312-14, 43386-312-85
- Packager: Lupin Pharmaceuticals,Inc.
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated January 30, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- WARNINGS
- Ask a doctor before use if you have
- Stop use and ask a doctor if
- Keep out of the reach of children
-
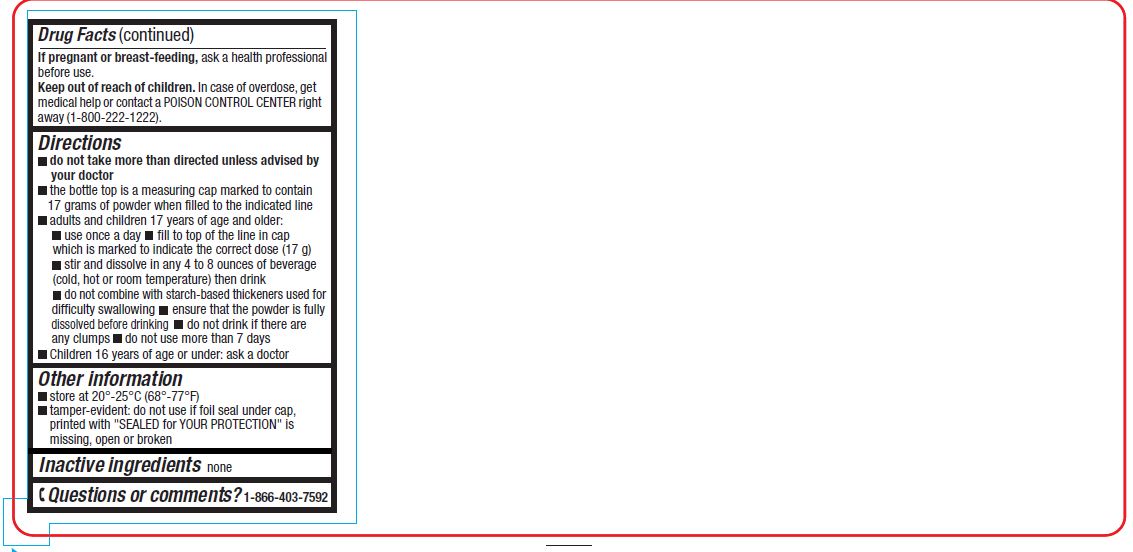
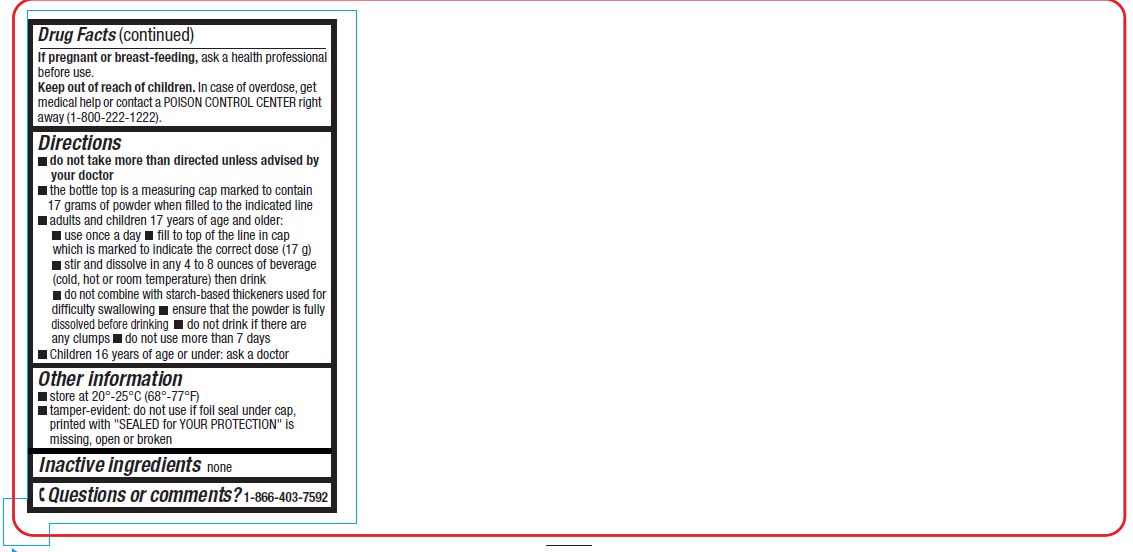
Directions
- do not take more than directed unless advised by your doctor
- the bottle top is a measuring cap marked to contain 17 grams of powder when filled to the indicated line.
- adults and children 17 years of age and older:
- use once a day
- fill to top of line in cap which is marked to indicate the correct dose (17 g)
- stir and dissolve in any 4 to 8 ounces of beverage (cold, hot or room temperature) then drink
- do not combine with starch-based thickeners used for difficulty swallowing
- ensure that the powder is fully dissolved before drinking
- do not drink if there are any clumps
- do not use more than 7 days
- children 16 years of age or under: ask a doctor
- store at 20°- 25°C (68°– 77°F)
- tamper-evident: do not use if printed foil seal under cap, printed with "SEALED for YOUR PROTECTION" is missing, open or broken
- Inactive Ingredient
- Questions or comments?
-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Original Prescription Strength
GaviLax
Polyethylene Glycol 3350
Powder for Oral Solution, Osmotic Laxative
14 ONCE-DAILY DOSES
NET WT 8.3 OZ (238 g)

Original Prescription Strength
GaviLax
Polyethylene Glycol 3350
Powder for Oral Solution, Osmotic Laxative
50 ONCE-DAILY DOSES
NET WT 29.9 OZ (850 g)

Original Prescription Strength
GaviLax
Polyethylene Glycol 3350
Powder for Oral Solution, Osmotic Laxative
7 ONCE-DAILY DOSES
NET WT 4.1 OZ (119 g)

Original Prescription Strength
GaviLax
Polyethylene Glycol 3350
Powder for Oral Solution, Osmotic Laxative
30 ONCE-DAILY DOSES
NET WT 17.9 OZ (510 g)

-
INGREDIENTS AND APPEARANCE
GAVILAX
polyethylene glycol 3350 powder, for solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:43386-312 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POLYETHYLENE GLYCOL 3350 (UNII: G2M7P15E5P) (POLYETHYLENE GLYCOL 3350 - UNII:G2M7P15E5P) POLYETHYLENE GLYCOL 3350 17 g in 17 g Inactive Ingredients Ingredient Name Strength SODIUM HYDROXIDE (UNII: 55X04QC32I) Product Characteristics Color WHITE Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:43386-312-14 510 g in 1 BOTTLE; Type 0: Not a Combination Product 10/09/2009 2 NDC:43386-312-08 238 g in 1 BOTTLE; Type 0: Not a Combination Product 10/09/2009 3 NDC:43386-312-07 119 g in 1 BOTTLE; Type 0: Not a Combination Product 01/31/2040 4 NDC:43386-312-85 850 g in 1 BOTTLE; Type 0: Not a Combination Product 01/31/2040 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA091077 10/09/2009 Labeler - Lupin Pharmaceuticals,Inc. (089153071) Registrant - Novel Laboratories, Inc. (793518643) Establishment Name Address ID/FEI Business Operations Novel Laboratories, Inc. 793518643 ANALYSIS(43386-312) , MANUFACTURE(43386-312) , PACK(43386-312)