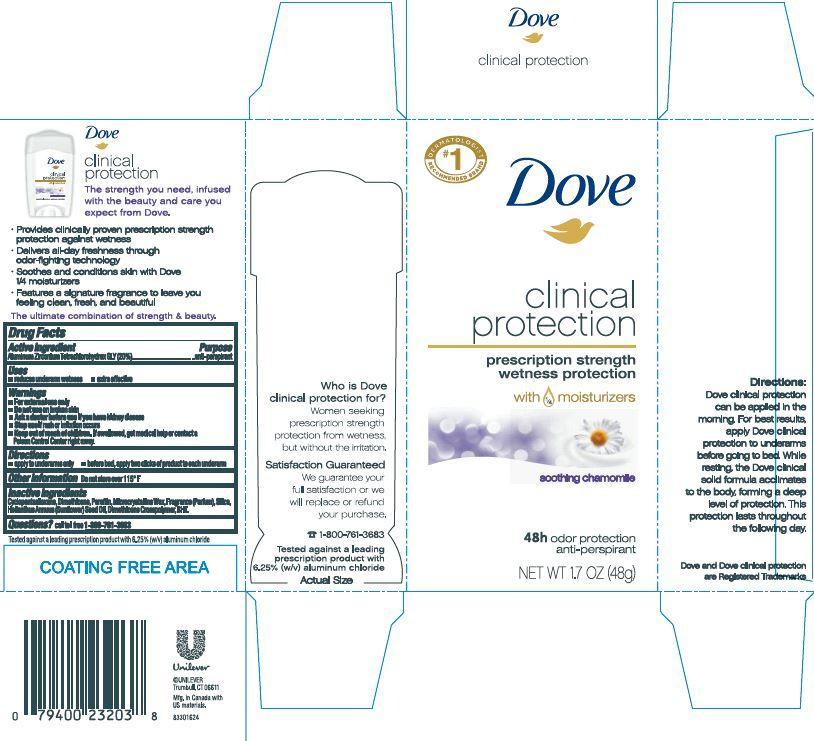
Label: DOVE CLINICAL PROTECTION SOOTHING CHAMOMILE ANTIPERSPIRANT AND DEODORANT- aluminum zirconium tetrachlorohydrex gly stick
-
NDC Code(s):
64942-1222-1,
64942-1222-2,
64942-1222-3,
64942-1222-4, view more64942-1222-5, 64942-1222-6
- Packager: Conopco Inc. d/b/a Unilever
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 5, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- DOSAGE & ADMINISTRATION
- INACTIVE INGREDIENT
- QUESTIONS
- 1.7 oz PDP
- 1.7 oz carton
-
INGREDIENTS AND APPEARANCE
DOVE CLINICAL PROTECTION SOOTHING CHAMOMILE ANTIPERSPIRANT AND DEODORANT
aluminum zirconium tetrachlorohydrex gly stickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:64942-1222 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALUMINUM ZIRCONIUM TETRACHLOROHYDREX GLY (UNII: 8O386558JE) (ALUMINUM ZIRCONIUM TETRACHLOROHYDREX GLY - UNII:8O386558JE) ALUMINUM ZIRCONIUM TETRACHLOROHYDREX GLY 20 g in 100 g Inactive Ingredients Ingredient Name Strength CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) DIMETHICONE (UNII: 92RU3N3Y1O) MICROCRYSTALLINE WAX (UNII: XOF597Q3KY) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SUNFLOWER OIL (UNII: 3W1JG795YI) PARAFFIN (UNII: I9O0E3H2ZE) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:64942-1222-2 1 in 1 CARTON 10/30/2020 1 NDC:64942-1222-1 48 g in 1 CONTAINER; Type 0: Not a Combination Product 2 NDC:64942-1222-4 1 in 1 CARTON 10/30/2020 2 NDC:64942-1222-3 14 g in 1 CONTAINER; Type 0: Not a Combination Product 3 NDC:64942-1222-6 1 in 1 CARTON 10/30/2020 3 NDC:64942-1222-5 62 g in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part350 10/30/2020 Labeler - Conopco Inc. d/b/a Unilever (001375088)