BUNAVAIL- buprenorphine and naloxone film
BioDelivery Sciences International, Inc.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use BUNAVAIL safely and effectively. See full prescribing information for BUNAVAIL.
BUNAVAIL® (buprenorphine and naloxone buccal film), CIII Initial U.S. Approval: 2002 RECENT MAJOR CHANGESINDICATIONS AND USAGEBUNAVAIL CONTAINS BUPRENORPHINE, A PARTIAL OPIOID AGONIST, AND NALOXONE, AN OPIOID ANTAGONIST, AND IS INDICATED FOR THE TREATMENT OF OPIOID DEPENDENCE. BUNAVAIL should be used as part of a complete treatment plan that includes counseling and psychosocial support. (1) DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHSBuccal film:
CONTRAINDICATIONSHypersensitivity to buprenorphine or naloxone. (4) WARNINGS AND PRECAUTIONS
ADVERSE REACTIONSAdverse events commonly observed with buccal administration of BUNAVAIL are headache, nausea, vomiting, hyperhidrosis, constipation, signs and symptoms of withdrawal, insomnia, pain, and peripheral edema. (6) To report SUSPECTED ADVERSE REACTIONS, contact BioDelivery Sciences International at 1-800-469-0261 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. DRUG INTERACTIONS
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide. Revised: 6/2022 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
BUNAVAIL is indicated for the treatment of opioid dependence. BUNAVAIL should be used as part of a complete treatment plan that includes counseling and psychosocial support.
2 DOSAGE AND ADMINISTRATION
2.1 Drug Addiction Treatment Act
Under the Drug Addiction Treatment Act (DATA) codified at 21 U.S.C. 823(g), prescription use of this product in the treatment of opioid dependence is limited to healthcare providers who meet certain qualifying requirements, and who have notified the Secretary of Health and Human Services (HHS) of their intent to prescribe this product for the treatment of opioid dependence and have been assigned a unique identification number that must be included on every prescription.
2.2 Important Dosage and Administration Information
BUNAVAIL is administered buccally as a single daily dose. The difference in bioavailability of BUNAVAIL compared to SUBOXONE sublingual tablet requires a different dosage strength to be administered to the patient. A BUNAVAIL 4.2 mg/0.7 mg buccal film provides equivalent buprenorphine exposure to a SUBOXONE 8 mg/2 mg sublingual tablet.
Medication should be prescribed in consideration of the frequency of visits. Provision of multiple refills is not advised early in treatment or without appropriate patient follow-up visits.
2.3 Patient Access to Naloxone for the Emergency Treatment of Opioid Overdose
Discuss the availability of naloxone for the emergency treatment of opioid overdose with the patient and caregiver. Because patients being treated for opioid use disorder have the potential for relapse, putting them at risk for opioid overdose, strongly consider prescribing naloxone for the emergency treatment of opioid overdose, both when initiating and renewing treatment with BUNAVAIL. Also consider prescribing naloxone if the patient has household members (including children) or other close contacts at risk for accidental ingestion or opioid overdose [see Warnings and Precautions (5.2)].
Advise patients and caregivers that naloxone may also be administered for a known or suspected overdose with BUNAVAIL itself. Higher than normal doses and repeated administration of naloxone may be necessary due to the long duration of action of BUNAVAIL and its affinity for the mu receptor [see Overdosage (10)].
Inform patients and caregivers of their options for obtaining naloxone as permitted by individual state naloxone dispensing and prescribing requirements or guidelines (e.g., by prescription, directly from a pharmacist, or as part of a community-based program) [see Patient Counseling Information (17)].
2.4 Induction
Prior to induction, consideration should be given to the type of opioid dependence (i.e., long- or short-acting opioid products), the time since last opioid use, and the degree of level of opioid dependence.
Patients dependent on heroin or other short-acting opioid products
Patients dependent on heroin or other short-acting opioid products may be inducted with either BUNAVAIL or with sublingual buprenorphine monotherapy. At treatment initiation, the first dose of BUNAVAIL should be administered when objective signs of moderate opioid withdrawal appear, not less than six hours after the patient last used opioids.
It is recommended that an adequate treatment dose, titrated to clinical effectiveness, be achieved as rapidly as possible. In some studies, a too-gradual induction over several days led to a high rate of dropout of buprenorphine patients during the induction period.
On Day 1, an induction dosage of up to 4.2 mg/0.7 mg BUNAVAIL is recommended. Clinicians should start with an initial dose of 2.1 mg/0.3 mg and repeat at approximately 2 hours, under supervision, to a total dose of 4.2 mg/0.7 mg buprenorphine/naloxone based on the control of acute withdrawal symptoms.
On Day 2, a single daily dose of up to 8.4 mg/1.4 mg BUNAVAIL is recommended.
Patients dependent on methadone or long-acting opioid products
Patients dependent upon methadone or long-acting opioid products may be more susceptible to precipitated and prolonged withdrawal during induction than those on short-acting opioid products.
Buprenorphine/naloxone combination products have not been evaluated in adequate and well-controlled studies for induction in patients who are physically dependent on long-acting opioid products, and the naloxone in these combination products is absorbed in small amounts by the sublingual route and could cause worse precipitated and prolonged withdrawal. For this reason, buprenorphine monotherapy is recommended in patients taking long-acting opioids when used according to approved administration instructions. Following induction, the patient may then be transitioned to once-daily BUNAVAIL.
2.5 Maintenance
The dosage of BUNAVAIL from Day 3 onwards should be progressively adjusted in increments/decrements of 2.1 mg/0.3 mg buprenorphine/naloxone to a level that holds the patient in treatment and suppresses opioid withdrawal signs and symptoms.
After treatment induction and stabilization, the maintenance dose of BUNAVAIL is generally in the range of 2.1 mg/0.3 mg buprenorphine/naloxone to 12.6 mg/2.1 mg buprenorphine/naloxone per day depending on the individual patient and clinical response. The recommended target dosage of BUNAVAIL during maintenance is 8.4 mg/1.4 mg as a single daily dose. Dosages higher than 12.6 mg/2.1 mg have not been demonstrated to provide any clinical advantage.
When determining the prescription quantity for unsupervised administration, consider the patient's level of stability, the security of his or her home situation, and other factors likely to affect the ability to manage supplies of take-home medication.
There is no maximum recommended duration of maintenance treatment. Patients may require treatment indefinitely and should continue for as long as patients are benefitting and the use of BUNAVAIL contributes to the intended treatment goals.
2.6 Method of Administration
The patient should:
- use the tongue to wet the inside of the cheek or rinse the mouth with water to moisten the area immediately before placement of BUNAVAIL;
- open the BUNAVAIL package immediately prior to use as indicated by the instructions;
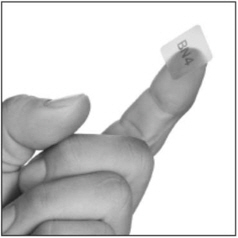
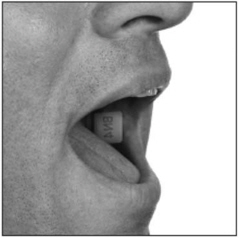
- hold the BUNAVAIL film with clean, dry fingers with the text (BN2, BN4, or BN6) facing up;
- place the side of the BUNAVAIL film with the text (BN2, BN4, or BN6) against the inside of the cheek;
- press and hold the film in place for 5 seconds.
- BUNAVAIL film(s) adheres to the moist buccal mucosa and should stay in place after this period.
If multiple films need to be administered, the patient should immediately apply the next film according to the steps above. Note that when two films are required for one dose, the patient should place one film on the inside of one cheek and the other film on the inside of the other cheek. For doses requiring multiple films, no more than two films should be applied to the inside of one cheek at a time.
BUNAVAIL film(s) completely dissolves after application. Instruct the patient to avoid manipulating the film(s) with the tongue or finger(s) and to avoid drinking or eating food until the film(s) dissolves. BUNAVAIL film should not be chewed or swallowed as this may result in lower peak concentrations and lower bioavailability [see Clinical Pharmacology (12.3)].
Instruct the patient to use the entire film. BUNAVAIL should not be cut or torn.
Proper administration technique should be demonstrated to the patient.
Advise patients to do the following after the product has completely dissolved in the oral mucosa: take a sip of water, swish gently around the teeth and gums, and swallow. Advise patients to wait for at least one hour after taking BUNAVAIL before brushing teeth [see Warnings and Precautions (5.13), Postmarketing Experience (6.2), Information for Patients (17), and the Medication Guide].
2.7 Clinical Supervision
Treatment should be initiated with supervised administration, progressing to unsupervised administration as the patient's clinical stability permits. BUNAVAIL is subject to diversion and abuse. When determining the prescription quantity for unsupervised administration, consider the patient's level of stability, the security of his or her home situation, and other factors likely to affect the ability to manage supplies of take-home medication.
Ideally patients should be seen at reasonable intervals (e.g., at least weekly during the first month of treatment) based upon the individual circumstances of the patient. Medication should be prescribed in consideration of the frequency of visits. Provision of multiple refills is not advised early in treatment or without appropriate patient follow-up visits. Periodic assessment is necessary to determine compliance with the dosing regimen, effectiveness of the treatment plan, and overall patient progress.
Once a stable dosage has been achieved and patient assessment (e.g., urine drug screening) does not indicate illicit drug use, less frequent follow-up visits may be appropriate. A once-monthly visit schedule may be reasonable for patients on a stable dosage of medication who are making progress toward their treatment objectives. Continuation or modification of pharmacotherapy should be based on the healthcare provider's evaluation of treatment outcomes and objectives such as:
- Absence of medication toxicity
- Absence of medical or behavioral adverse effects
- Responsible handling of medications by the patient
- Patient's compliance with all elements of the treatment plan (including recovery-oriented activities, psychotherapy, and/or other psychosocial modalities)
- Abstinence from illicit drug use (including problematic alcohol and/or benzodiazepine use)
If treatment goals are not being achieved, the healthcare provider should re-evaluate the appropriateness of continuing the current treatment.
2.8 Unstable Patients
Healthcare providers will need to decide when they cannot appropriately provide further management for particular patients. For example, some patients may be abusing or dependent on various drugs, or unresponsive to psychosocial intervention such that the healthcare provider does not feel that he/she has the expertise to manage the patient. In such cases, the healthcare provider may want to assess whether to refer the patient to a specialist or more intensive behavioral treatment environment. Decisions should be based on a treatment plan established and agreed upon with the patient at the beginning of treatment. Patients who continue to misuse, abuse, or divert buprenorphine products or other opioids should be provided with, or referred to, more intensive and structured treatment.
2.9 Discontinuing Treatment
The decision to discontinue therapy with BUNAVAIL after a period of maintenance should be made as part of a comprehensive treatment plan. Advise patients of the potential to relapse to illicit drug use following discontinuation of opioid agonist/partial agonist medication-assisted treatment. Taper patients to avoid the occurrence of withdrawal signs and symptoms [see Warnings and Precautions (5.7)].
2.10 Switching between BUNAVAIL Buccal Film and Other Buprenorphine/Naloxone Combination Products
For patients being switched between BUNAVAIL and other buprenorphine/naloxone products dosage adjustments may be necessary. Patients should be monitored for over-medication as well as for withdrawal or other signs of under-dosing.
The difference in bioavailability of BUNAVAIL compared to SUBOXONE sublingual tablet requires a different dosage strength to be administered to the patient. One BUNAVAIL 4.2 mg/0.7 mg buccal film provides equivalent buprenorphine exposure to one SUBOXONE 8 mg/2 mg sublingual tablet.
Patients being switched between SUBOXONE dosage strengths and BUNAVAIL dosage strengths should be started on the corresponding dosage strengths as shown below:
| Suboxone Sublingual Tablet Dosage Strength | Corresponding BUNAVAIL Buccal Film Strength |
|---|---|
| 4 mg buprenorphine/1 mg naloxone | 2.1 mg buprenorphine/0.3 mg naloxone |
| 8 mg buprenorphine/2 mg naloxone | 4.2 mg buprenorphine/0.7 mg naloxone |
| 12 mg buprenorphine/3 mg naloxone | 6.3 mg buprenorphine/1 mg naloxone |
3 DOSAGE FORMS AND STRENGTHS
BUNAVAIL buccal film is supplied as a yellow rectangular buccal film in three dosage strengths:
- buprenorphine 2.1 mg/naloxone 0.3 mg
- buprenorphine 4.2 mg/naloxone 0.7 mg
- buprenorphine 6.3 mg/naloxone 1 mg
4 CONTRAINDICATIONS
BUNAVAIL is contraindicated in patients with a history of hypersensitivity to buprenorphine or naloxone as serious adverse reactions, including anaphylactic shock, have been reported [see Warnings and Precautions (5.9)].
5 WARNINGS AND PRECAUTIONS
5.1 Addiction, Abuse, and Misuse
BUNAVAIL contains buprenorphine, a Schedule III controlled substance that can be abused in a manner similar to other opioids, legal or illicit. Prescribe and dispense buprenorphine with appropriate precautions to minimize risk of misuse, abuse, or diversion, and ensure appropriate protection from theft, including in the home. Clinical monitoring appropriate to the patient's level of stability is essential. Multiple refills should not be prescribed early in treatment or without appropriate patient follow-up visits [see Drug Abuse and Dependence (9.2)].
5.2 Risk of Life-Threatening Respiratory Depression and Central Nervous System (CNS) Depression
Buprenorphine has been associated with life-threatening respiratory depression and death. Many, but not all, post-marketing reports regarding coma and death involved misuse by self-injection or were associated with the concomitant use of buprenorphine and benzodiazepines or other CNS depressants including alcohol. Warn patients of the potential danger of self-administration of benzodiazepines or other CNS depressants while under treatment with BUNAVAIL [see Warnings and Precautions (5.3), Drug Interactions (7)].
Use BUNAVAIL with caution in patients with compromised respiratory function (e.g., chronic obstructive pulmonary disease, cor pulmonale, decreased respiratory reserve, hypoxia, hypercapnia, or pre-existing respiratory depression).
Educate patients and caregivers on how to recognize respiratory depression and emphasize the importance of calling 911 or getting emergency medical help right away in the event of a known or suspected overdose [see Patient Counseling Information (17)].
Opioids can cause sleep-related breathing disorders including central sleep apnea (CSA) and sleep-related hypoxemia. Opioid use increases the risk of CSA in a dose-dependent fashion. In patients who present with CSA, consider decreasing the opioid dosage using best practices for opioid taper [see Dosage and Administration (2.9)].
Patient Access to Naloxone for the Emergency Treatment of Opioid Overdose
Discuss the availability of naloxone for the emergency treatment of opioid overdose with the patient and caregiver.
Because patients being treated for opioid use disorder have the potential for relapse, putting them at risk for opioid overdose, strongly consider prescribing naloxone for the emergency treatment of opioid overdose, both when initiating and renewing treatment with BUNAVAIL. Also consider prescribing naloxone if the patient has household members (including children) or other close contacts at risk for accidental ingestion or opioid overdose [see Dosage and Administration (2.3)].
Advise patients and caregivers that naloxone may also be administered for a known or suspected overdose with BUNAVAIL itself. Higher than normal doses and repeated administration of naloxone may be necessary due to the long duration of action of BUNAVAIL and its affinity for the mu receptor [see Overdosage (10)].
Inform patients and caregivers of their options for obtaining naloxone as permitted by individual state naloxone dispensing and prescribing requirements or guidelines (e.g., by prescription, directly from a pharmacist, or as part of a community-based program).
Educate patients and caregivers on how to recognize respiratory depression and, if naloxone is prescribed, how to treat with naloxone. Emphasize the importance of calling 911 or getting emergency medical help, even if naloxone is administered [see Patient Counseling Information (17)].
5.3 Managing Risks from Concomitant Use of Benzodiazepines or Other CNS Depressants
Concomitant use of buprenorphine and benzodiazepines or other CNS depressants increases the risk of adverse reactions including overdose and death. Medication-assisted treatment of opioid use disorder, however, should not be categorically denied to patients taking these drugs.
Prohibiting or creating barriers to treatment can pose an even greater risk of morbidity and mortality due to the opioid use disorder alone.
As a routine part of orientation to buprenorphine treatment, educate patients about the risks of concomitant use of benzodiazepines, sedatives, opioid analgesics, and alcohol.
Develop strategies to manage use of prescribed or illicit benzodiazepines or other CNS depressants at initiation of buprenorphine treatment, or if it emerges as a concern during treatment. Adjustments to induction procedures and additional monitoring may be required. There is no evidence to support dose limitations or arbitrary caps of buprenorphine as a strategy to address benzodiazepine use in buprenorphine-treated patients. However, if a patient is sedated at the time of buprenorphine dosing, delay or omit the buprenorphine dose if appropriate.
Cessation of benzodiazepines or other CNS depressants is preferred in most cases of concomitant use. In some cases, monitoring in a higher level of care for taper may be appropriate. In others, gradually tapering a patient off of a prescribed benzodiazepine or other CNS depressant or decreasing to the lowest effective dose may be appropriate.
For patients in buprenorphine treatment, benzodiazepines are not the treatment of choice for anxiety or insomnia. Before co-prescribing benzodiazepines, ensure that patients are appropriately diagnosed and consider alternative medications and non-pharmacologic treatments to address anxiety or insomnia. Ensure that other healthcare providers prescribing benzodiazepines or other CNS depressants are aware of the patient's buprenorphine treatment and coordinate care to minimize the risks associated with concomitant use.
If concomitant use is warranted, strongly consider prescribing naloxone for the emergency treatment of opioid overdose, as is recommended for all patients in buprenorphine treatment for opioid use disorder [see Warnings and Precautions (5.2)].
In addition, take measures to confirm that patients are taking their medications as prescribed and are not diverting or supplementing with illicit drugs. Toxicology screening should test for prescribed and illicit benzodiazepines [see Drug Interactions (7)].
5.4 Unintentional Pediatric Exposure
Buprenorphine can cause severe, possibly fatal, respiratory depression in children who are accidentally exposed to it. Store buprenorphine-containing medications safely out of the sight and reach of children and destroy any unused medication appropriately [see Patient Counseling Information (17)].
5.5 Neonatal Opioid Withdrawal Syndrome
Neonatal opioid withdrawal syndrome (NOWS) is an expected and treatable outcome of prolonged use of opioids during pregnancy, whether that use is medically-authorized or illicit. Unlike opioid withdrawal syndrome in adults, NOWS may be life-threatening if not recognized and treated in the neonate. Healthcare professionals should observe newborns for signs of NOWS and manage accordingly [see Use in Specific Populations (8.1)].
Advise pregnant women receiving opioid addiction treatment with BUNAVAIL of the risk of neonatal opioid withdrawal syndrome and ensure that appropriate treatment will be available [see Use in Specific Populations (8.1)]. This risk must be balanced against the risk of untreated opioid addiction which often results in continued or relapsing illicit opioid use and is associated with poor pregnancy outcomes. Therefore, prescribers should discuss the importance and benefits of management of opioid addiction throughout pregnancy.
5.6 Adrenal Insufficiency
Cases of adrenal insufficiency have been reported with opioid use, more often following greater than one month of use. Presentation of adrenal insufficiency may include non-specific symptoms and signs including nausea, vomiting, anorexia, fatigue, weakness, dizziness, and low blood pressure. If adrenal insufficiency is diagnosed, treat with physiologic replacement doses of corticosteroids. Wean the patient off of the opioid to allow adrenal function to recover and continue corticosteroid treatment until adrenal function recovers. Other opioids may be tried as some cases reported use of a different opioid without recurrence of adrenal insufficiency. The information available does not identify any particular opioids as being more likely to be associated with adrenal insufficiency.
5.7 Risk of Opioid Withdrawal with Abrupt Discontinuation
Buprenorphine is a partial agonist at the mu-opioid receptor and chronic administration produces physical dependence of the opioid type, characterized by withdrawal signs and symptoms upon abrupt discontinuation or rapid taper. The withdrawal syndrome is typically milder than seen with full agonists and may be delayed in onset. When discontinuing BUNAVAIL, gradually taper the dosage [see Dosage and Administration (2.9)].
5.8 Risk of Hepatitis, Hepatic Events
Cases of cytolytic hepatitis and hepatitis with jaundice have been observed in individuals receiving buprenorphine in clinical trials and through post-marketing adverse event reports. The spectrum of abnormalities ranges from transient asymptomatic elevations in hepatic transaminases to case reports of death, hepatic failure, hepatic necrosis, hepatorenal syndrome, and hepatic encephalopathy. In many cases, the presence of pre-existing liver enzyme abnormalities, infection with hepatitis B or hepatitis C virus, concomitant usage of other potentially hepatotoxic drugs, and ongoing injecting drug use may have played a causative or contributory role. In other cases, insufficient data were available to determine the etiology of the abnormality. Withdrawal of buprenorphine has resulted in amelioration of acute hepatitis in some cases; however, in other cases no dose reduction was necessary. The possibility exists that buprenorphine had a causative or contributory role in the development of the hepatic abnormality in some cases. Liver function tests prior to initiation of treatment are recommended to establish a baseline. Periodic monitoring of liver function during treatment is also recommended. A biological and etiological evaluation is recommended when a hepatic event is suspected. Depending on the case, BUNAVAIL may need to be carefully discontinued to prevent withdrawal signs and symptoms and a return by the patient to illicit drug use, and strict monitoring of the patient should be initiated.
5.9 Hypersensitivity Reactions
Cases of hypersensitivity to buprenorphine and naloxone containing products have been reported both in clinical trials and in post-marketing experience. Cases of bronchospasm, angioneurotic edema, and anaphylactic shock have been reported. The most common signs and symptoms include rashes, hives and pruritus. A history of hypersensitivity to buprenorphine or naloxone is a contraindication to the use of BUNAVAIL.
5.10 Precipitation of Opioid Withdrawal Signs and Symptoms
Because it contains naloxone, BUNAVAIL is likely to produce withdrawal signs and symptoms if misused parenterally by individuals dependent on full opioid agonists such as heroin, morphine, or methadone. Because of the partial agonist properties of buprenorphine, BUNAVAIL may precipitate opioid withdrawal signs and symptoms in such persons if administered buccally before the agonist effects of the opioid have subsided.
5.11 Risk of Overdose in Opioid Naïve Patients
There have been reported deaths of opioid naïve individuals who received a 2 mg dose of buprenorphine, smaller than the lowest strength of BUNAVAIL, for analgesia. BUNAVAIL is not appropriate as an analgesic.
5.12 Use in Patients with Impaired Hepatic Function
Buprenorphine/naloxone products are not recommended in patients with severe hepatic impairment and may not be appropriate for patients with moderate hepatic impairment. The doses of buprenorphine and naloxone in this fixed-dose combination product cannot be individually titrated, and hepatic impairment results in a reduced clearance of naloxone to a much greater extent than buprenorphine. Therefore, patients with severe hepatic impairment will be exposed to substantially higher levels of naloxone than patients with normal hepatic function. This may result in an increased risk of precipitated withdrawal at the beginning of treatment (induction) and may interfere with buprenorphine's efficacy throughout treatment. In patients with moderate hepatic impairment, the differential reduction of naloxone clearance compared to buprenorphine clearance is not as great as in subjects with severe hepatic impairment. However, buprenorphine/naloxone products are not recommended for initiation of treatment (induction) in patients with moderate hepatic impairment due to the increased risk of precipitated withdrawal. Buprenorphine/naloxone products may be used with caution for maintenance treatment in patients with moderate hepatic impairment who have initiated treatment on a buprenorphine product without naloxone. However, patients should be carefully monitored and consideration given to the possibility of naloxone interfering with buprenorphine's efficacy [see Use in Specific Populations (8.6)].
5.13 Dental Adverse Events
Cases of dental caries, some severe (i.e., tooth fracture, tooth loss), have been reported following the use of transmucosal buprenorphine-containing products. Reported events include cavities, tooth decay, dental abscesses/infection, rampant caries, tooth erosion, fillings falling out, and, in some cases, total tooth loss. Treatment for these events included tooth extraction, root canal, dental surgery, as well as other restorative procedures (i.e., fillings, crowns, implants, dentures). Multiple cases were reported in individuals without any prior history of dental problems.
Refer patients to dental care services and encourage them to have regular dental checkups while taking BUNAVAIL. Educate patients to seek dental care and strategies to maintain or improve oral health while being treated with transmucosal buprenorphine-containing products. Strategies include, but are not limited to, gently rinsing the teeth and gums with water and then swallowing after BUNAVAIL has been completely dissolved in the oral mucosa. Advise patients to wait for at least one hour after taking BUNAVAIL before brushing teeth [see Dosing and Administration (2.6), Information for Patients (17), Medication Guide].
5.14 QTc Prolongation
Thorough QT studies with buprenorphine products have demonstrated QT prolongation ≤15 msec. This QTc prolongation effect does not appear to be mediated by hERG channels. Based on these two findings, buprenorphine is unlikely to be pro-arrhythmic when used alone in patients without risk factors. The risk of combining buprenorphine with other QT-prolonging agents is not known.
Consider these observations in clinical decisions when prescribing Bunavail to patients with risk factors such as hypokalemia, bradycardia, recent conversion from atrial fibrillation, congestive heart failure, digitalis therapy, baseline QT prolongation, subclinical long-QT syndrome, or severe hypomagnesemia.
5.15 Impairment of Ability to Drive or Operate Machinery
BUNAVAIL may impair the mental or physical abilities required for the performance of potentially dangerous tasks such as driving a car or operating machinery, especially during treatment induction and dose adjustment. Caution patients about driving or operating hazardous machinery until they are reasonably certain that BUNAVAIL therapy does not adversely affect their ability to engage in such activities.
5.16 Orthostatic Hypotension
Like other opioids, BUNAVAIL may produce orthostatic hypotension in ambulatory patients.
5.17 Elevation of Cerebrospinal Fluid Pressure
Buprenorphine, like other opioids, may elevate cerebrospinal fluid pressure and should be used with caution in patients with head injury, intracranial lesions, and other circumstances when cerebrospinal pressure may be increased. Buprenorphine can produce miosis and changes in the level of consciousness that may interfere with patient evaluation.
6 ADVERSE REACTIONS
The following serious adverse reactions are described elsewhere in the labeling:
- Addiction, Abuse, and Misuse [see Warnings and Precautions (5.1)]
- Respiratory and CNS Depression [see Warnings and Precautions (5.2, 5.3)]
- Neonatal Opioid Withdrawal Syndrome [see Warnings and Precautions (5.5)]
- Adrenal Insufficiency [see Warnings and Precautions (5.6)]
- Opioid Withdrawal [see Warnings and Precautions (5.7, 5.10)]
- Hepatitis, Hepatic Events [see Warnings and Precautions (5.8)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.9)]
- Orthostatic Hypotension [see Warnings and Precautions (5.16)]
- Elevation of Cerebrospinal Fluid Pressure [see Warnings and Precautions (5.17)]
- Elevation of Intracholedochal Pressure [see Warnings and Precautions (5.18)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of BUNAVAIL is supported by clinical trials using buprenorphine and naloxone sublingual tablets, and other trials using buprenorphine tablets and buprenorphine sublingual solutions, as well as an open-label study in 249 patients treated with BUNAVAIL. In total, safety data from clinical studies are available from over 3000 opioid-dependent subjects exposed to buprenorphine at doses in the range used in the treatment of opioid addiction. Few differences in the adverse event profile were noted among buprenorphine and naloxone sublingual tablets, buprenorphine sublingual tablets and a buprenorphine ethanolic sublingual solution.
The safety and tolerability of BUNAVAIL was evaluated in a 12-week clinical study of BUNAVAIL in 249 opioid-dependent subjects stabilized on buprenorphine and naloxone sublingual tablet or film dosages of buprenorphine 8 to 32 mg/day.
The following adverse reactions were reported to occur by at least 5% of patients in a 12-week study with BUNAVAIL: drug withdrawal syndrome, lethargy and headache.
The adverse reactions listed below represent those that were reported by >1% but less than 5% of patients from the 12-week clinical trial while receiving BUNAVAIL. Events are classified by system organ class.
- General disorders and administration site conditions: fatigue and chills
- Nervous system disorders: somnolence
- Psychiatric disorders: drug dependence and insomnia
- Gastrointestinal disorders: constipation and oral mucosal erythema
- Respiratory, thoracic and mediastinal disorders: rhinorrhea
- Skin and subcutaneous tissue disorders: hyperhidrosis
The following adverse events were reported to occur by at least 5% of patients in a 4-week study with buprenorphine and naloxone sublingual tablets (Table 1).
| Body System / Adverse Event (COSTART terminology) | Buprenorphine/naloxone sublingual tablets 16 mg/4 mg/day | Placebo |
|---|---|---|
| N=107 n (%) | N=107 n (%) |
|
| Body as a Whole | ||
| Asthenia | 7 (6.5%) | 7 (6.5%) |
| Chills | 8 (7.5%) | 8 (7.5%) |
| Headache | 39 (36.4%) | 24 (22.4%) |
| Infection | 6 (5.6%) | 7 (6.5%) |
| Pain | 24 (22.4%) | 20 (18.7%) |
| Pain abdomen | 12 (11.2%) | 7 (6.5%) |
| Pain back | 4 (3.7%) | 12 (11.2%) |
| Withdrawal syndrome | 27 (25.2%) | 40 (37.4%) |
| Cardiovascular System | ||
| Vasodilation | 10 (9.3%) | 7 (6.5%) |
| Digestive System | ||
| Constipation | 13 (12.1%) | 3 (2.8%) |
| Diarrhea | 4 (3.7%) | 16 (15.0%) |
| Nausea | 16 (15.0%) | 12 (11.2%) |
| Vomiting | 8 (7.5%) | 5 (4.7%) |
| Nervous System | ||
| Insomnia | 15 (14.0%) | 17 (15.9%) |
| Respiratory System | ||
| Rhinitis | 5 (4.7%) | 14 (13.1%) |
| Skin and Appendages | ||
| Sweating | 15 (14.0%) | 11 (10.3%) |
The adverse event profile of buprenorphine was also characterized in a dose-controlled study of buprenorphine solutions over a range of doses in four months of treatment. Table 2 shows adverse events reported by at least 5% of subjects in any dose group in the dose-controlled study.
| Body System / Adverse Event (COSTART terminology) | Buprenorphine Dose* | ||||
|---|---|---|---|---|---|
| Very Low*
(N=184) n (%) | Low*
(N=180) n (%) | Moderate*
(N=186) n (%) | High*
(N=181) n (%) | Total*
(N=731) n (%) |
|
|
|||||
| Body as a Whole | |||||
| Abscess | 9 (5%) | 2 (1%) | 3 (2%) | 2 (1%) | 16 (2%) |
| Asthenia | 26 (14%) | 28 (16%) | 26 (14%) | 24 (13%) | 104 (14%) |
| Chills | 11 (6%) | 12 (7%) | 9 (5%) | 10 (6%) | 42 (6%) |
| Fever | 7 (4%) | 2 (1%) | 2 (1%) | 10 (6%) | 21 (3%) |
| Flu Syndrome | 4 (2%) | 13 (7%) | 19 (10%) | 8 (4%) | 44 (6%) |
| Headache | 51 (28%) | 62 (34%) | 54 (29%) | 53 (29%) | 220 (30%) |
| Infection | 32 (17%) | 39 (22%) | 38 (20%) | 40 (22%) | 149 (20%) |
| Injury Accidental | 5 (3%) | 10 (6%) | 5 (3%) | 5 (3%) | 25 (3%) |
| Pain | 47 (26%) | 37 (21%) | 49 (26%) | 44 (24%) | 177 (24%) |
| Pain Back | 18 (10%) | 29 (16%) | 28 (15%) | 27 (15%) | 102 (14%) |
| Withdrawal Syndrome | 45 (24%) | 40 (22%) | 41 (22%) | 36 (20%) | 162 (22%) |
| Digestive System | |||||
| Constipation | 10 (5%) | 23 (13%) | 23 (12%) | 26 (14%) | 82 (11%) |
| Diarrhea | 19 (10%) | 8 (4%) | 9 (5%) | 4 (2%) | 40 (5%) |
| Dyspepsia | 6 (3%) | 10 (6%) | 4 (2%) | 4 (2%) | 24 (3%) |
| Nausea | 12 (7%) | 22 (12%) | 23 (12%) | 18 (10%) | 75 (10%) |
| Vomiting | 8 (4%) | 6 (3%) | 10 (5%) | 14 (8%) | 38 (5%) |
| Nervous System | |||||
| Anxiety | 22 (12%) | 24 (13%) | 20 (11%) | 25 (14%) | 91 (12%) |
| Depression | 24 (13%) | 16 (9%) | 25 (13%) | 18 (10%) | 83 (11%) |
| Dizziness | 4 (2%) | 9 (5%) | 7 (4%) | 11 (6%) | 31 (4%) |
| Insomnia | 42 (23%) | 50 (28%) | 43 (23%) | 51 (28%) | 186 (25%) |
| Nervousness | 12 (7%) | 11 (6%) | 10 (5%) | 13 (7%) | 46 (6%) |
| Somnolence | 5 (3%) | 13 (7%) | 9 (5%) | 11 (6%) | 38 (5%) |
| Respiratory System | |||||
| Cough Increase | 5 (3%) | 11 (6%) | 6 (3%) | 4 (2%) | 26 (4%) |
| Pharyngitis | 6 (3%) | 7 (4%) | 6 (3%) | 9 (5%) | 28 (4%) |
| Rhinitis | 27 (15%) | 16 (9%) | 15 (8%) | 21 (12%) | 79 (11%) |
| Skin and Appendages | |||||
| Sweat | 23 (13%) | 21 (12%) | 20 (11%) | 23 (13%) | 87 (12%) |
| Special Senses | |||||
| Runny Eyes | 13 (7%) | 9 (5%) | 6 (3%) | 6 (3%) | 34 (5%) |
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of buprenorphine. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate a causal relationship to drug exposure.
Serotonin syndrome: Cases of serotonin syndrome, a potentially life-threatening condition, have been reported during concomitant use of opioids with serotonergic drugs.
Adrenal insufficiency: Cases of adrenal insufficiency have been reported with opioid use, more often following greater than one month of use.
Anaphylaxis: Anaphylaxis has been reported with ingredients contained in BUNAVAIL.
Androgen deficiency: Cases of androgen deficiency have occurred with chronic use of opioids [see Clinical Pharmacology (12.2)].
Local reactions: dental decay (including caries, tooth fracture, and tooth loss), glossodynia, glossitis, oral mucosal erythema, oral hypoesthesia, and stomatitis.
7 DRUG INTERACTIONS
Table 3 includes clinically significant drug interactions with BUNAVAIL.
| Benzodiazepine and other Central Nervous System (CNS) Depressants | |
|---|---|
| Clinical Impact: | Due to additive pharmacologic effects, the concomitant use of benzodiazepines and other CNS depressants, including alcohol, increases the risk of respiratory depression, profound sedation, coma, and death. |
| Intervention: | Cessation of benzodiazepines or other CNS depressants is preferred in most cases of concomitant use. In some cases, monitoring in a higher level of care for taper may be appropriate. In others, gradually tapering a patient off of a prescribed benzodiazepine or other CNS depressant or decreasing to the lowest effective dose may be appropriate. Before co-prescribing benzodiazepines for anxiety or insomnia, ensure that patients are appropriately diagnosed and consider alternative medications and non-pharmacologic treatments [see Warnings and Precautions (5.2, 5.3)]. If concomitant use is warranted, strongly consider prescribing naloxone for the emergency treatment of opioid overdose, as is recommended for all patients in treatment for opioid use disorder [see Warnings and Precautions (5.2)]. |
| Examples: | Alcohol, benzodiazepines and other sedatives/hypnotics, anxiolytics, tranquilizers, muscle relaxants, general anesthetics, antipsychotics, and other opioids. |
| Inhibitors of CYP3A4 | |
| Clinical Impact: | The concomitant use of buprenorphine and CYP3A4 inhibitors can increase the plasma concentration of buprenorphine, resulting in increased or prolonged opioid effects, particularly when an inhibitor is added after a stable dose of BUNAVAIL is achieved. After stopping a CYP3A4 inhibitor, as the effects of the inhibitor decline, the buprenorphine plasma concentration will decrease [see Clinical Pharmacology (12.3)], potentially resulting in decreased opioid efficacy or a withdrawal syndrome in patients who had developed physical dependence to buprenorphine. |
| Intervention: | If concomitant use is necessary, consider dosage reduction of BUNAVAIL until stable drug effects are achieved. Monitor patients for respiratory depression and sedation at frequent intervals. If a CYP3A4 inhibitor is discontinued, consider increasing the BUNAVAIL dosage until stable drug effects are achieved. Monitor for signs of opioid withdrawal. |
| Examples: | Macrolide antibiotics (e.g., erythromycin), azole-antifungal agents (e.g., ketoconazole), protease inhibitors (e.g., ritonavir) |
| CYP3A4 Inducers | |
| Clinical Impact: | The concomitant use of buprenorphine and CYP3A4 inducers can decrease the plasma concentration of buprenorphine [see Clinical Pharmacology (12.3)], potentially resulting in decreased efficacy or onset of a withdrawal syndrome in patients who have developed physical dependence to buprenorphine. After stopping a CYP3A4 inducer, as the effects of the inducer decline, the buprenorphine plasma concentration will increase [see Clinical Pharmacology (12.3)], which could increase or prolong both therapeutic effects and adverse reactions and may cause serious respiratory depression. |
| Intervention: | If concomitant use is necessary, consider increasing the BUNAVAIL dosage until stable drug effects are achieved. Monitor for signs of opioid withdrawal. If a CYP3A4 inducer is discontinued, consider BUNAVAIL dosage reduction and monitor for signs of respiratory depression. |
| Examples: | Rifampin, carbamazepine, phenytoin |
| Antiretrovirals: Non-nucleoside reverse transcriptase inhibitors (NNRTIs) | |
| Clinical Impact: | Non-nucleoside reverse transcriptase inhibitors (NNRTIs) are metabolized principally by CYP3A4. Efavirenz, nevirapine, and etravirine are known CYP3A inducers, whereas delavirdine is a CYP3A inhibitor. Significant pharmacokinetic interactions between NNRTIs (e.g., efavirenz and delavirdine) and buprenorphine have been shown in clinical studies, but these pharmacokinetic interactions did not result in any significant pharmacodynamic effects. |
| Intervention: | Patients who are on chronic BUNAVAIL treatment should have their dose monitored if NNRTIs are added to their treatment regimen. |
| Examples: | efavirenz, nevirapine, etravirine, delavirdine |
| Antiretrovirals: Protease inhibitors (PIs) | |
| Clinical Impact: | Studies have shown some antiretroviral protease inhibitors (PIs) with CYP3A4 inhibitory activity (nelfinavir, lopinavir/ritonavir, ritonavir) have little effect on buprenorphine pharmacokinetics and no significant pharmacodynamic effects. Other PIs with CYP3A4 inhibitory activity (atazanavir and atazanavir/ritonavir) resulted in elevated levels of buprenorphine and norbuprenorphine, and patients in one study reported increased sedation. Symptoms of opioid excess have been found in post-marketing reports of patients receiving buprenorphine and atazanavir with and without ritonavir concomitantly. |
| Intervention: | Monitor patients taking BUNAVAIL and atazanavir with and without ritonavir, and reduce dose of BUNAVAIL if warranted. |
| Examples: | atazanavir, ritonavir |
| Antiretrovirals: Nucleoside reverse transcriptase inhibitors (NRTIs) | |
| Clinical Impact: | Nucleoside reverse transcriptase inhibitors (NRTIs) do not appear to induce or inhibit the P450 enzyme pathway, thus no interactions with buprenorphine are expected. |
| Intervention: | None |
| Serotonergic Drugs | |
| Clinical Impact: | The concomitant use of opioids with other drugs that affect the serotonergic neurotransmitter system has resulted in serotonin syndrome. |
| Intervention: | If concomitant use is warranted, carefully observe the patient, particularly during treatment initiation and dose adjustment. Discontinue BUNAVAIL if serotonin syndrome is suspected. |
| Examples: | Selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), triptans, 5-HT3 receptor antagonists, drugs that affect the serotonin neurotransmitter system (e.g., mirtazapine, trazodone, tramadol), certain muscle relaxants (i.e., cyclobenzaprine, metaxalone), monoamine oxidase (MAO) inhibitors (those intended to treat psychiatric disorders and also others, such as linezolid and intravenous methylene blue). |
| Monoamine Oxidase Inhibitors (MAOIs) | |
| Clinical Impact: | MAOI interactions with opioids may manifest as serotonin syndrome or opioid toxicity (e.g., respiratory depression, coma) [see Warnings and Precautions (5.2)] |
| Intervention: | The use of BUNAVAIL is not recommended for patients taking MAOIs or within 14 days of stopping such treatment. |
| Examples: | phenelzine, tranylcypromine, linezolid |
| Muscle Relaxants | |
| Clinical Impact: | Buprenorphine may enhance the neuromuscular blocking action of skeletal muscle relaxants and produce an increased degree of respiratory depression. |
| Intervention: | Monitor patients receiving muscle relaxants and BUNAVAIL for signs of respiratory depression that may be greater than otherwise expected and decrease the dosage of BUNAVAIL and/or the muscle relaxant as necessary. Due to the risk of respiratory depression with concomitant use of skeletal muscle relaxants and opioids, strongly consider prescribing naloxone for the emergency treatment of opioid overdose [see Dosage and Administration (2.3), Warnings and Precautions (5.2, 5.3)]. |
| Diuretics | |
| Clinical Impact: | Opioids can reduce the efficacy of diuretics by inducing the release of antidiuretic hormone. |
| Intervention: | Monitor patients for signs of diminished diuresis and/or effects on blood pressure and increase the dosage of the diuretic as needed. |
| Anticholinergic Drugs | |
| Clinical Impact: | The concomitant use of anticholinergic drugs may increase the risk of urinary retention and/or severe constipation, which may lead to paralytic ileus. |
| Intervention: | Monitor patients for signs of urinary retention or reduced gastric motility when BUNAVAIL is used concomitantly with anticholinergic drugs. |
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
The data on use of buprenorphine, one of the active ingredients in BUNAVAIL, in pregnancy, are limited; however, these data do not indicate an increased risk of major malformations specifically due to buprenorphine exposure. There are limited data from randomized clinical trials in women maintained on buprenorphine that were not designed appropriately to assess the risk of major malformations [see Data]. Observational studies have reported on congenital malformations among buprenorphine-exposed pregnancies, but were also not designed appropriately to assess the risk of congenital malformations specifically due to buprenorphine exposure [see Data]. The extremely limited data on sublingual naloxone exposure in pregnancy are not sufficient to evaluate a drug‐associated risk.
Reproductive and developmental studies in rats and rabbits identified adverse events at clinically relevant and higher doses. Embryofetal death was observed in both rats and rabbits administered buprenorphine during the period of organogenesis at doses approximately 6 and 0.3 times, respectively, the human sublingual dose of 16 mg/day of buprenorphine (equivalent to 8.4 mg/1.4 mg BUNAVAIL). Pre-and postnatal development studies in rats demonstrated increased neonatal deaths at 0.3 times and above, and dystocia at approximately 3 times, the human sublingual dose of 16 mg/day of buprenorphine (equivalent to 8.4 mg/1.4 mg BUNAVAIL). No clear teratogenic effects were seen when buprenorphine was administered during organogenesis with a range of doses equivalent to or greater than the human sublingual dose of 16 mg/day of buprenorphine (equivalent to 8.4 mg/1.4 mg BUNAVAIL). However, increases in skeletal abnormalities were noted in rats and rabbits administered buprenorphine daily during organogenesis at doses approximately 0.6 times and approximately equal to the human sublingual dose of 16 mg/day of buprenorphine (equivalent to 8.4 mg/1.4 mg BUNAVAIL), respectively. In a few studies, some events such as acephalus and omphalocele were also observed but these findings were not clearly treatment-related [see Data]. Based on animal data, advise pregnant women of the potential risk to a fetus.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defects, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Clinical Considerations
Disease-associated maternal and embryo-fetal risk
Untreated opioid addiction in pregnancy is associated with adverse obstetrical outcomes such as low birth weight, preterm birth, and fetal death. In addition, untreated opioid addiction often results in continued or relapsing illicit opioid use.
Dose Adjustment during Pregnancy and the Postpartum Period
Dosage adjustments of buprenorphine may be required during pregnancy, even if the patient was maintained on a stable dose prior to pregnancy. Withdrawal signs and symptoms should be monitored closely and the dose adjusted as necessary.
Fetal/neonatal adverse reactions
Neonatal opioid withdrawal syndrome may occur in newborn infants of mothers who are receiving treatment with BUNAVAIL.
Neonatal opioid withdrawal syndrome presents as irritability, hyperactivity and abnormal sleep pattern, high pitched cry, tremor, vomiting, diarrhea, and/or failure to gain weight. Signs of neonatal withdrawal usually occur in the first days of birth. The duration and severity of neonatal opioid withdrawal syndrome may vary. Observe newborns for signs of neonatal opioid withdrawal syndrome and manage accordingly [see Warnings and Precautions (5.5)].
Data
Human Data
Studies have been conducted to evaluate neonatal outcomes in women exposed to buprenorphine during pregnancy. Limited data from trials, observational studies, case series, and case reports on buprenorphine use in pregnancy do not indicate an increased risk of major malformations specifically due to buprenorphine. Several factors may complicate the interpretation of investigations of the children of women who take buprenorphine during pregnancy, including maternal use of illicit drugs, late presentation for prenatal care, infection, poor compliance, poor nutrition, and psychosocial circumstances. Interpretation of data is complicated further by the lack of information on untreated opioid-dependent pregnant women, who would be the most appropriate group for comparison. Rather, women on another form of opioid medication-assisted treatment or women in the general population are generally used as the comparison group. However, women in these comparison groups may be different from women prescribed buprenorphine-containing products with respect to maternal factors that may lead to poor pregnancy outcomes.
In a multicenter, double-blind, randomized, controlled trial [Maternal Opioid Treatment: Human Experimental Research (MOTHER)] designed primarily to assess neonatal opioid withdrawal effects, opioid-dependent pregnant women were randomized to buprenorphine (n=86) or methadone (n=89) treatment, with enrollment at an average gestational age of 18.7 weeks in both groups. A total of 28 of the 86 women in the buprenorphine group (33%) and 16 of the 89 women in the methadone group (18%) discontinued treatment before the end of pregnancy.
Among women who remained in treatment until delivery, there was no difference between buprenorphine-treated and methadone-treated groups in the number of neonates requiring NOWS treatment or in the peak severity of NOWS. Buprenorphine-exposed neonates required less morphine (mean total dose, 1.1 mg vs. 10.4 mg), had shorter hospital stays (10.0 days vs. 17.5 days), and shorter duration of treatment for NOWS (4.1 days vs. 9.9 days) compared to the methadone-exposed group. There were no differences between groups in other primary outcomes (neonatal head circumference) or secondary outcomes (weight and length at birth, preterm birth, gestational age at delivery, and 1-minute and 5-minute Apgar scores), or in the rates of maternal or neonatal adverse events. The outcomes among mothers who discontinued treatment before delivery and may have relapsed to illicit opioid use are not known. Because of the imbalance in discontinuation rates between the buprenorphine and methadone groups, the study findings are difficult to interpret.
Animal Data
BUNAVAIL has been shown to have differences in bioavailability compared to buprenorphine/naloxone-containing sublingual products. The exposure margins listed below are based on body surface area comparisons (mg/m2) to the recommended human sublingual dose of 16 mg buprenorphine from Suboxone sublingual tablets (equivalent to 8.4 mg/1.4 mg BUNAVAIL).
Effects on embryo-fetal development were studied in Sprague-Dawley rats and Russian white rabbits following oral (1:1) and intramuscular (IM) (3:2) administration of mixtures of buprenorphine and naloxone during the period of organogenesis. Following oral administration to rats, no teratogenic effects were observed at buprenorphine doses up to 250 mg/kg/day (estimated exposure approximately 150 times the human sublingual dose of 16 mg) in the presence of maternal toxicity (mortality).
Following oral administration to rabbits, no teratogenic effects were observed at buprenorphine doses up to 40 mg/kg/day (estimated exposure approximately 50 times the human sublingual dose of 16 mg) in the absence of clear maternal toxicity. No definitive drug-related teratogenic effects were observed in rats and rabbits at IM doses up to 30 mg/kg/day (estimated exposure approximately 20 times and 35 times, respectively, the human sublingual dose of 16 mg). Maternal toxicity resulting in mortality was noted in these studies in both rats and rabbits. Acephalus was observed in one rabbit fetus from the low-dose group and omphalocele was observed in two rabbit fetuses from the same litter in the mid-dose group; no findings were observed in fetuses from the high-dose group. Maternal toxicity was seen in the high-dose group but not at the lower doses where the findings were observed. Following oral administration of buprenorphine to rats, dose-related post-implantation losses, evidenced by increases in the numbers of early resorptions with consequent reductions in the numbers of fetuses, were observed at doses of 10 mg/kg/day or greater (estimated exposure approximately 6 times the human sublingual dose of 16 mg).
In the rabbit, increased post-implantation losses occurred at an oral dose of 40 mg/kg/day. Following IM administration in the rat and the rabbit, post-implantation losses, as evidenced by decreases in live fetuses and increases in resorptions, occurred at 30 mg/kg/day.
Buprenorphine was not teratogenic in rats or rabbits after IM or subcutaneous (SC) doses up to 5 mg/kg/day (estimated exposure was approximately 3 and 6 times, respectively, the human sublingual dose of 16 mg), after IV doses up to 0.8 mg/kg/day (estimated exposure was approximately 0.5 times and equal to, respectively, the human sublingual dose of 16 mg), or after oral doses up to 160 mg/kg/day in rats (estimated exposure was approximately 95 times the human sublingual dose of 16 mg) and 25 mg/kg/day in rabbits (estimated exposure was approximately 30 times the human sublingual dose of 16 mg). Significant increases in skeletal abnormalities (e.g., extra thoracic vertebra or thoraco-lumbar ribs) were noted in rats after SC administration of 1 mg/kg/day and up (estimated exposure was approximately 0.6 times the human sublingual dose of 16 mg) but were not observed at oral doses up to 160 mg/kg/day.
Increases in skeletal abnormalities in rabbits after IM administration of 5 mg/kg/day (estimated exposure was approximately 6 times the human sublingual dose of 16 mg) or oral administration of 1 mg/kg/day or greater (estimated exposure was approximately equal to the human sublingual dose of 16 mg) were not statistically significant.
In rabbits, buprenorphine produced statistically significant pre-implantation losses at oral doses of 1 mg/kg/day or greater and post-implantation losses that were statistically significant at IV doses of 0.2 mg/kg/day or greater (estimated exposure approximately 0.3 times the human sublingual dose of 16 mg). No maternal toxicity was noted at doses causing post-implantation loss in this study.
Dystocia was noted in pregnant rats treated intramuscularly with buprenorphine during gestation and lactation at 5 mg/kg/day (approximately 3 times the human sublingual dose of 16 mg). Fertility, pre-, and post-natal development studies with buprenorphine in rats indicated increases in neonatal mortality after oral doses of 0.8 mg/kg/day and up (approximately 0.5 times the human sublingual dose of 16 mg), after IM doses of 0.5 mg/kg/day and up (approximately 0.3 times the human sublingual dose of 16 mg), and after SC doses of 0.1 mg/kg/day and up (approximately 0.06 times the human sublingual dose of 16 mg). An apparent lack of milk production during these studies likely contributed to the decreased pup viability and lactation indices. Delays in the occurrence of righting reflex and startle response were noted in rat pups at an oral dose of 80 mg/kg/day (approximately 50 times the human sublingual dose of 16 mg).
8.2 Lactation
Risk Summary
Based on two studies in 13 lactating women maintained on buprenorphine treatment, buprenorphine and its metabolite norbuprenorphine were present in low levels in human milk and available data have not shown adverse reactions in breastfed infants. There are no data on the combination product buprenorphine/naloxone in breastfeeding, however oral absorption of naloxone is limited. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for BUNAVAIL and any potential adverse effects on the breastfed child from the drug or from the underlying maternal condition.
Clinical Considerations
Advise the nursing mother taking BUNAVAIL to monitor the infant for increased drowsiness and breathing difficulties.
Data
Data were consistent from two studies (N=13) of breastfeeding infants whose mothers were maintained on sublingual doses of buprenorphine ranging from 2.4 to 24 mg/day, showing that the infants were exposed to less than 1% of the maternal daily dose.
In a study of six lactating women who were taking a median sublingual buprenorphine dose of 0.29 mg/kg/day 5 to 8 days after delivery, breast milk provided a median infant dose of 0.42 mcg/kg/day of buprenorphine and 0.33 mcg/kg/day of norbuprenorphine, equal to 0.2% and 0.12%, respectively, of the maternal weight-adjusted dose (relative dose/kg (%) of norbuprenorphine was calculated from the assumption that buprenorphine and norbuprenorphine are equipotent).
Data from a study of seven lactating women who were taking a median sublingual buprenorphine dose of 7 mg/day an average of 1.12 months after delivery indicated that the mean milk concentrations (Cavg) of buprenorphine and norbuprenorphine were 3.65 mcg/L and 1.94 mcg/L respectively. Based on the study data, and assuming milk consumption of 150 mL/kg/day, an exclusively breastfed infant would receive an estimated mean absolute infant dose (AID) of 0.55 mcg/kg/day of buprenorphine and 0.29 mcg/kg/day of norbuprenorphine, or a mean relative infant dose (RID) of 0.38% and 0.18%, respectively, of the maternal weight-adjusted dose.
8.3 Females and Males of Reproductive Potential
Infertility
Chronic use of opioids may cause reduced fertility in females and males of reproductive potential. It is not known whether these effects on fertility are reversible [see Nonclinical Toxicology (13.1)].
8.4 Pediatric Use
The safety and effectiveness of BUNAVAIL have not been established in pediatric patients.
This product is not appropriate for the treatment of neonatal abstinence syndrome in neonates, because it contains naloxone, an opioid antagonist.
8.5 Geriatric Use
Clinical studies of BUNAVAIL did not include sufficient numbers of subjects aged 65 and over to determine whether they responded differently than younger subjects. Other reported clinical experience has not identified differences in responses between elderly and younger patients. Due to possible decreased hepatic, renal, or cardiac function and of concomitant disease or other drug therapy in geriatric patients, the decision to prescribe BUNAVAIL should be made cautiously in individuals 65 years of age or older and these patients should be monitored for signs and symptoms of toxicity or overdose.
8.6 Hepatic Impairment
The effect of hepatic impairment on the pharmacokinetics of buprenorphine and naloxone has been evaluated in a pharmacokinetic study. Both drugs are extensively metabolized in the liver. While no clinically significant changes have been observed in subjects with mild hepatic impairment, the plasma levels have been shown to be higher and half-life values have been shown to be longer for both buprenorphine and naloxone in subjects with moderate and severe hepatic impairment. The magnitude of the effects on naloxone is greater than that on buprenorphine in both moderately and severely impaired subjects. The difference in magnitude of the effects on naloxone and buprenorphine are greater in subjects with severe hepatic impairment than in subjects with moderate hepatic impairment, and therefore the clinical impact of these effects is likely to be greater in patients with severe hepatic impairment than in patients with moderate hepatic impairment. Buprenorphine/naloxone products should be avoided in patients with severe hepatic impairment and may not be appropriate for patients with moderate hepatic impairment [see Warnings and Precautions (5.12), Clinical Pharmacology (12.3)].
9 DRUG ABUSE AND DEPENDENCE
9.1 Controlled Substance
BUNAVAIL contains buprenorphine, a Schedule III substance under the Controlled Substances Act.
Under the Drug Addiction Treatment Act (DATA) codified at 21 U.S.C. 823(g), prescription use of this product in the treatment of opioid dependence is limited to healthcare providers who meet certain qualifying requirements, and who have notified the Secretary of Health and Human Services (HHS) of their intent to prescribe this product for the treatment of opioid dependence and have been assigned a unique identification number that must be included on every prescription.
9.2 Abuse
Buprenorphine, like morphine and other opioids, has the potential for being abused and is subject to criminal diversion. This should be considered when prescribing or dispensing buprenorphine in situations when the clinician is concerned about an increased risk of misuse, abuse, or diversion. Healthcare professionals should contact their state professional licensing board or state controlled substances authority for information on how to prevent and detect abuse or diversion of this product.
Patients who continue to misuse, abuse, or divert buprenorphine products or other opioids should be provided with, or referred to, more intensive and structured treatment.
Abuse of buprenorphine poses a risk of overdose and death. This risk is increased with the abuse of buprenorphine and alcohol and other substances, especially benzodiazepines.
The healthcare provider may be able to more easily detect misuse or diversion by maintaining records of medication prescribed including date, dose, quantity, frequency of refills, and renewal requests of medication prescribed.
Proper assessment of the patient, proper prescribing practices, periodic re-evaluation of therapy, and proper handling and storage of the medication are appropriate measures that help to limit abuse of opioid drugs.
9.3 Dependence
Buprenorphine is a partial agonist at the mu-opioid receptor and chronic administration produces physical dependence of the opioid type, characterized by moderate withdrawal signs and symptoms upon abrupt discontinuation or rapid taper. The withdrawal syndrome is typically milder than seen with full agonists and may be delayed in onset [see Warnings and Precautions (5.7)].
Neonatal opioid withdrawal syndrome (NOWS) is an expected and treatable outcome of prolonged use of opioids during pregnancy [see Warnings and Precautions (5.5)].
10 OVERDOSAGE
Clinical Presentation
The manifestations of acute overdose include pinpoint pupils, sedation, hypotension, respiratory depression, and death.
Treatment of Overdose
In the event of overdose, the respiratory and cardiac status of the patient should be monitored carefully. When respiratory or cardiac functions are depressed, primary attention should be given to the re-establishment of adequate respiratory exchange through provision of a patent airway and institution of assisted or controlled ventilation. Oxygen, IV fluids, vasopressors, and other supportive measures should be employed as indicated.
In the case of overdose, the primary management should be the re-establishment of adequate ventilation with mechanical assistance of respiration, if required. Naloxone may be of value for the management of buprenorphine overdose. Higher than normal doses and repeated administration may be necessary. The long duration of action of BUNAVAIL should be taken into consideration when determining the length of treatment and medical surveillance needed to reverse the effects of an overdose. Insufficient duration of monitoring may put patients at risk.
11 DESCRIPTION
BUNAVAIL (buprenorphine and naloxone buccal film) is a citrus flavored oral transmucosal form of buprenorphine, an opioid partial agonist, and naloxone, an opioid antagonist, intended for application to the buccal mucosa. Each dose unit is a yellow rectangular film, with ink marking on the mucoadhesive side. The film adheres upon contact with the moist buccal mucosa. BUNAVAIL contains buprenorphine HCl, a mu-opioid receptor partial agonist and a kappa-opioid receptor antagonist, and naloxone HCl dihydrate, an opioid receptor antagonist, at a ratio of ~6:1 (ratio of free bases). It is available in three strengths: 2.1 mg buprenorphine with 0.3 mg naloxone in a 2.2 cm2 film; 4.2 mg buprenorphine with 0.7 mg naloxone in a 4.4 cm2 film; and 6.3 mg buprenorphine with 1 mg naloxone in a 6.5 cm2 film. Each film also contains carboxymethylcellulose sodium, citric acid, citrus blend flavor, dibasic sodium phosphate, blue ink, hydroxyethyl cellulose, hydroxypropyl cellulose, methylparaben, monobasic sodium phosphate, polycarbophil, propylene glycol, propylparaben, yellow iron oxide, sodium benzoate, sodium hydroxide, sodium saccharin, vitamin E acetate, and purified water. The blue ink contains FD&C Blue No.1, ethanol, purified shellac, acetone, ammonium hydroxide and water.
Chemically, buprenorphine HCl, USP is 6,14-Ethenomorphinan-7-methanol, 17-(cyclopropyl-methyl)-α-(1,1-dimethylethyl)-4,5-epoxy-18,19-dihydro-3-hydroxy-6-methoxy-α-methyl-, hydrochloride, [5α, 7α (S)]. It has the following chemical structure:
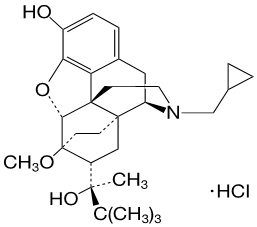
Buprenorphine HCl has the molecular formula C29H41NO4 ∙ HCl and the molecular weight is 504.10. It is a white or off-white crystalline powder, sparingly soluble in water, freely soluble in methanol, soluble in alcohol, and practically insoluble in cyclohexane.
Chemically, naloxone HCl dihydrate, USP is morphinan-6-one, 4,5-epoxy-3,14-dihydroxy-17-(2-propenyl)-, hydrochloride, (5α)-, dihydrate. It has the following chemical structure:
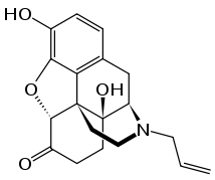 ∙HCl ∙2H2O
∙HCl ∙2H2O
Naloxone hydrochloride dihydrate has the molecular formula C19H21NO4 ∙ HCl ∙ 2H2O and the molecular weight is 399.87. It is a white to slightly off-white powder, and is freely soluble in water, soluble in alcohol, and practically insoluble in toluene and ether.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
BUNAVAIL contains buprenorphine and naloxone. Buprenorphine is a partial agonist at the mu-opioid receptor and an antagonist at the kappa-opioid receptor. Naloxone is a potent antagonist at mu-opioid receptors and produces opioid withdrawal signs and symptoms, if administered parenterally, in individuals physically dependent on full opioid agonists.
12.2 Pharmacodynamics
Subjective Effects
Comparisons of buprenorphine to full opioid agonists such as methadone and hydromorphone suggest that sublingual buprenorphine produces typical opioid agonist effects which are limited by a ceiling effect.
In opioid-experienced subjects who were not physically dependent, acute sublingual doses of buprenorphine/naloxone tablets produced opioid agonist effects which reached a maximum between doses of 8/2 mg and 16/4 mg buprenorphine/naloxone, which corresponds to BUNAVAIL buccal doses of 4.2 mg/0.7 mg and 8.4 mg/1.4 mg buprenorphine/naloxone, respectively.
Opioid agonist ceiling effects were also observed in a double-blind, parallel group, dose-ranging comparison of single doses of buprenorphine sublingual solution (1, 2, 4, 8, 16, or 32 mg), placebo and a full agonist control at various doses. The treatments were given in ascending dose order at intervals of at least one week to 16 opioid-experienced subjects who were not physically dependent. Both active drugs produced typical opioid agonist effects. For all measures for which the drugs produced an effect, buprenorphine produced a dose-related response. However, in each case, there was a dose that produced no further effect. In contrast, the highest dose of the full agonist control always produced the greatest effects. Agonist objective rating scores remained elevated for the higher doses of buprenorphine (8 to 32 mg) longer than for the lower doses and did not return to baseline until 48 hours after drug administration. The onset of effects appeared more rapidly with buprenorphine than with the full agonist control, with most doses nearing peak effect after 100 minutes for buprenorphine compared to 150 minutes for the full agonist control.
Physiological Effects
Buprenorphine in IV (2, 4, 8, 12, and 16 mg) and sublingual (12 mg) doses have been administered in opioid-experienced subjects who were not physically dependent to examine cardiovascular, respiratory, and subjective effects at doses comparable to those used for treatment of opioid dependence. Compared to placebo, there were no statistically significant differences among any of the treatment conditions for blood pressure, heart rate, respiratory rate, O2 saturation, or skin temperature across time. Systolic BP was higher in the 8 mg group than placebo (3-hour AUC values). Minimum and maximum effects were similar across all treatments. Subjects remained responsive to low voice and responded to computer prompts. Some subjects showed irritability, but no other changes were observed.
The respiratory effects of sublingual buprenorphine were compared with the effects of methadone in a double-blind, parallel group, dose ranging comparison of single doses of buprenorphine sublingual solution (1, 2, 4, 8, 16, or 32 mg) and oral methadone (15, 30, 45, or 60 mg) in non-dependent, opioid-experienced volunteers. In this study, hypoventilation not requiring medical intervention was reported more frequently after buprenorphine doses of 4 mg and higher than after methadone. Both drugs decreased O2 saturation to the same degree.
Effect of Naloxone
Physiologic and subjective effects following acute sublingual administration of buprenorphine tablets and buprenorphine/naloxone tablets were similar at equivalent dose levels of buprenorphine. Naloxone had no clinically significant effect when administered by the sublingual route, although blood levels of the drug were measurable. Buprenorphine/naloxone, when administered sublingually to an opioid-dependent cohort, was recognized as an opioid agonist, whereas when administered intramuscularly, combinations of buprenorphine with naloxone produced opioid antagonist actions similar to naloxone. This finding suggests that the naloxone in buprenorphine/naloxone products may deter injection of buprenorphine/naloxone products by persons with active substantial heroin or other full agonist mu-opioid dependence. However, clinicians should be aware that some opioid-dependent persons, particularly those with a low level of full agonist mu-opioid physical dependence or those whose opioid physical dependence is predominantly to buprenorphine, abuse buprenorphine/naloxone combinations by the intravenous or intranasal route. In methadone-maintained patients and heroin-dependent subjects, IV administration of buprenorphine/naloxone combinations precipitated opioid withdrawal signs and symptoms and was perceived as unpleasant and dysphoric. In morphine-stabilized subjects, intravenously administered combinations of buprenorphine with naloxone produced opioid antagonist and withdrawal signs and symptoms that were ratio-dependent; the most intense withdrawal signs and symptoms were produced by 2:1 and 4:1 ratios, less intense by an 8:1 ratio.
Effects on the Endocrine System
Opioids inhibit the secretion of adrenocorticotropic hormone (ACTH), cortisol, and luteinizing hormone (LH) in humans [see Adverse Reactions (6.2)]. They also stimulate prolactin, growth hormone (GH) secretion, and pancreatic secretion of insulin and glucagon.
Chronic use of opioids may influence the hypothalamic-pituitary-gonadal axis, leading to androgen deficiency that may manifest as low libido, impotence, erectile dysfunction, amenorrhea, or infertility. The causal role of opioids in the clinical syndrome of hypogonadism is unknown because the various medical, physical, lifestyle, and psychological stressors that may influence gonadal hormone levels have not been adequately controlled for in studies conducted to date. Patients presenting with symptoms of androgen deficiency should undergo laboratory evaluation.
12.3 Pharmacokinetics
Absorption
Plasma levels of buprenorphine and naloxone increased with the buccal dose of BUNAVAIL. There was wide inter-patient variability in the buccal absorption of buprenorphine and naloxone, but within subjects the variability was low. Both Cmax and AUC of buprenorphine increased with the increase in dose (in the range of 0.875 to 6.3 mg), although the increase was not directly dose-proportional. Naloxone did not affect the pharmacokinetics of buprenorphine.
BUNAVAIL has been shown to have different bioavailability compared to SUBOXONE tablet. The exposure of buprenorphine from one BUNAVAIL 4.2 mg/0.7 mg buccal film was equivalent to one SUBOXONE 8 mg/2 mg sublingual tablet. The naloxone exposure from BUNAVAIL was 33% less than with SUBOXONE sublingual tablets.
The co-administration of liquids reduced the systemic exposure up to 59% for buprenorphine and up to 76% for naloxone from BUNAVAIL, depending on the pH of the liquid, in comparison to the administration of BUNAVAIL when no liquid was co-administered [see Method of Administration (2.6)].
Distribution
Buprenorphine is approximately 96% protein bound, primarily to alpha and beta globulin. Naloxone is approximately 45% protein bound, primarily to albumin.
Elimination
Based on all studies performed with BUNAVAIL, buprenorphine has a mean elimination half-life from plasma ranging from 16.4 to 27.5 hours and naloxone has a mean elimination half-life from plasma ranging from 1.9 to 2.4 hours.
Metabolism
Buprenorphine undergoes both N-dealkylation to norbuprenorphine and glucuronidation. The N-dealkylation pathway is mediated primarily by CYP3A4. Norbuprenorphine, the major metabolite, can further undergo glucuronidation. Norbuprenorphine has been found to bind opioid receptors in vitro; however, it has not been studied clinically for opioid-like activity. Naloxone undergoes direct glucuronidation to naloxone-3-glucuronide as well as N-dealkylation, and reduction of the 6-oxo group.
Excretion
A mass balance study of buprenorphine showed complete recovery of radiolabel in urine (30%) and feces (69%) collected up to 11 days after dosing. Almost all of the dose was accounted for in terms of buprenorphine, norbuprenorphine, and two unidentified buprenorphine metabolites. In urine, most of buprenorphine and norbuprenorphine was conjugated (buprenorphine, 1% free and 9.4% conjugated; norbuprenorphine, 2.7% free and 11% conjugated). In feces, almost all of the buprenorphine and norbuprenorphine was free (buprenorphine, 33% free and 5% conjugated; norbuprenorphine, 21% free and 2% conjugated).
Drug Interaction Studies
CYP3A4 Inhibitors and Inducers
Buprenorphine has been found to be a CYP2D6 and CYP3A4 inhibitor and its major metabolite, norbuprenorphine, has been found to be a moderate CYP2D6 inhibitor in in vitro studies employing human liver microsomes. However, the relatively low plasma concentrations of buprenorphine and norbuprenorphine resulting from therapeutic doses are not expected to raise significant drug-drug interaction concerns [see Drug Interactions (7)].
Specific Populations
Hepatic Impairment
In a pharmacokinetic study, the disposition of buprenorphine and naloxone were determined in subjects with varied degrees of hepatic impairment as indicated by Child-Pugh criteria. The disposition of buprenorphine and naloxone in patients with hepatic impairment were compared to disposition in subjects with normal hepatic function.
In subjects with mild hepatic impairment, the changes in mean Cmax, AUC0-last, and half-life values of both buprenorphine and naloxone were not clinically significant. No dosing adjustment is needed in patients with mild hepatic impairment.
For subjects with moderate and severe hepatic impairment, mean Cmax, AUC0-last, and half-life values of both buprenorphine and naloxone were increased; the effects on naloxone are greater than those on buprenorphine (Table 4).
| Hepatic Impairment | PK Parameters | Increase in buprenorphine compared to healthy subjects | Increase in naloxone compared to healthy subjects |
|---|---|---|---|
| Moderate | Cmax | 8% | 170% |
| AUC0-last | 64% | 218% | |
| Half-life | 35% | 165% | |
| Severe | Cmax | 72% | 1030% |
| AUC0-last | 181% | 1302% | |
| Half-life | 57% | 122% |
The difference in magnitude of the effects on naloxone and buprenorphine are greater in subjects with severe hepatic impairment than subjects with moderate hepatic impairment [see Warnings and Precautions (5.12), Use in Specific Populations (8.6)].
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
BUNAVAIL has been shown to have differences in bioavailability compared to buprenorphine/naloxone-containing sublingual products. The exposure margins listed below are based on body surface area comparisons (mg/m2) to the recommended human sublingual dose of 16 mg buprenorphine from Suboxone, which is equivalent to a recommended human buccal dose (RHD) of 8.4 mg buprenorphine from BUNAVAIL.
Carcinogenicity
A carcinogenicity study of buprenorphine/naloxone (4:1 ratio of the free bases) was performed in Alderley Park rats. Buprenorphine/naloxone was administered in the diet at doses of approximately 7, 31, and 123 mg/kg/day for 104 weeks (estimated buprenorphine exposure was approximately 4, 18, and 44 times the Recommended Human Dose - RHD based on buprenorphine AUC comparisons). A statistically significant increase in Leydig cell adenomas was observed in all dose groups. No other drug-related tumors were noted.
Carcinogenicity studies of buprenorphine were conducted in Sprague-Dawley rats and CD-1 mice. Buprenorphine was administered in the diet to rats at doses of 0.6, 5.5, and 56 mg/kg/day (estimated exposure was approximately 0.4, 3, and 35 times the RHD) for 27 months. As in the buprenorphine/naloxone carcinogenicity study in rat, statistically significant dose-related increases in Leydig cell tumors occurred. In an 86-week study in CD-1 mice, buprenorphine was not carcinogenic at dietary doses up to 100 mg/kg/day (estimated buprenorphine exposure was approximately 30 times the RHD).
Mutagenicity
The 4:1 combination of buprenorphine and naloxone was not mutagenic in a bacterial mutation assay (Ames test) using four strains of S. typhimurium and two strains of E. coli. The combination was not clastogenic in an in vitro cytogenetic assay in human lymphocytes or in an IV micronucleus test in the rat.
Buprenorphine was studied in a series of tests utilizing gene, chromosome, and DNA interactions in both prokaryotic and eukaryotic systems. Results were negative in yeast (S. cerevisiae) for recombinant, gene convertant, or forward mutations; negative in Bacillus subtilis "rec" assay, negative for clastogenicity in CHO cells, Chinese hamster bone marrow and spermatogonia cells, and negative in the mouse lymphoma L5178Y assay.
Results were equivocal in the Ames test: negative in studies in two laboratories, but positive for frame shift mutation at a high dose (5 mg/plate) in a third study. Results were positive in the Green-Tweets (E.coli) survival test, positive in a DNA synthesis inhibition (DSI) test with testicular tissue from mice, for both in vivo and in vitro incorporation of [3H]thymidine, and positive in unscheduled DNA synthesis (UDS) test using testicular cells from mice.
Impairment of Fertility
Dietary administration of buprenorphine in the rat at dose levels of 500 ppm or greater (equivalent to approximately 47 mg/kg/day or greater; estimated exposure approximately 28 times the RHD) produced a reduction in fertility demonstrated by reduced female conception rates. A dietary dose of 100 ppm (equivalent to approximately 10 mg/kg/day; estimated exposure approximately 6 times the RHD) had no adverse effect on fertility.
16 HOW SUPPLIED/STORAGE AND HANDLING
BUNAVAIL is supplied in individually-sealed foil packages. BUNAVAIL is supplied in three dosage strengths. Each unit is individually wrapped in a child resistant foil package. These foil packages are packed 30 per box.
Each dosage unit is a yellow, rectangular film, with a dosage marking printed on the mucoadhesive side. The dosage strength of each unit is indicated by the dosage marking on the mucoadhesive side of the dose unit, and the dosage strength is marked on the foil package and the 30-unit box. See package and carton for product information.
| BUNAVAIL | Dosage Marking | Package Color * | NDC Number Carton | NDC Number Foil Package |
|---|---|---|---|---|
|
||||
| buprenorphine 2.1 mg/naloxone 0.3 mg | BN2 | Purple | NDC 59385-012-30 | NDC 59385-012-01 |
| buprenorphine 4.2 mg/naloxone 0.7 mg | BN4 | Blue | NDC 59385-014-30 | NDC 59385-014-01 |
| buprenorphine 6.3 mg/naloxone 1 mg | BN6 | Orange | NDC 59385-016-30 | NDC 59385-016-01 |
Store at 20°C to 25°C (68°F to 77°F), with excursions permitted between 15°C and 30°C (59°F and 86°F). Protect BUNAVAIL from freezing and moisture. Do not use if the foil package has been damaged.
Store BUNAVAIL securely and dispose of properly [see Patient Counseling Information (17)].
17 PATIENT COUNSELING INFORMATION
Advise patients to read the FDA-approved patient labeling (Medication Guide).
Storage and Disposal
Because of the risks associated with accidental ingestion, misuse, and abuse, advise patients to store BUNAVAIL securely, out of sight and reach of children, and in a location not accessible by others, including visitors to the home [see Warnings and Precautions (5.1, 5.4), Drug Abuse and Dependence (9.2)]. Inform patients that leaving BUNAVAIL unsecured can pose a deadly risk to others in the home.
Advise patients and caregivers that when medicines are no longer needed, they should be disposed of promptly. Expired, unwanted, or unused BUNAVAIL should be disposed of by removing the BUNAVAIL film from the foil packaging and flushing the unused medication down the toilet (if a drug take-back option is not readily available). Inform patients that they can visit www.fda.gov/drugdisposal for a complete list of medicines recommended for disposal by flushing, as well as additional information on disposal of unused medicines.
Safe Use
Before initiating treatment with BUNAVAIL, explain the points listed below to caregivers and patients. Instruct patients to read the Medication Guide each time BUNAVAIL is dispensed because new information may be available.
- BUNAVAIL must be administered whole. Advise patients not to chew or swallow BUNAVAIL.
- Inform patients and caregivers that potentially fatal additive effects may occur if BUNAVAIL is used with benzodiazepines or other CNS depressants, including alcohol. Counsel patients that such medications should not be used concomitantly unless supervised by a healthcare provider [see Warnings and Precautions (5.2, 5.3), Drug Interactions (7)].
- Educate patients and caregivers on how to recognize respiratory depression and emphasize the importance of calling 911 or getting emergency medical help right away in the event of a known or suspected overdose [see Warnings and Precautions (5.2)].
-
Patient Access to Naloxone for the Emergency Treatment of Opioid Overdose
Because patients being treated for opioid use disorder are at risk for relapse, discuss the importance of having access to naloxone with the patient and caregiver. Also discuss the importance of having access to naloxone if there are household members (including children) or other close contacts at risk for accidental ingestion or opioid overdose.
Inform patients and caregivers of the options for obtaining naloxone as permitted by individual state naloxone dispensing and prescribing requirements or guidelines (e.g., by prescription, directly from a pharmacist, or as part of a community-based program).
Educate patients and caregivers on how to recognize the signs and symptoms of an opioid overdose.
Explain to patients and caregivers that naloxone's effects are temporary, and that they must call 911 or get emergency medical help right away in all cases of known or suspected opioid overdose, even if naloxone is administered. Repeat administration may be necessary, particularly for overdose involving BUNAVAIL, because naloxone is often not effective at the doses available for patient access [see Dosage and Administration (2.3), Warnings and Precautions (5.2), Overdosage (10)].
If naloxone is prescribed, also advise patients and caregivers:- How to treat with naloxone in the event of an opioid overdose
- To tell family and friends about their naloxone and to keep it in a place where family and friends can easily access it in an emergency
- To read the Patient Information (or other educational material) that will come with their naloxone. Emphasize the importance of doing this before an opioid emergency happens, so the patient and caregiver will know what to do.
- Advise patients that BUNAVAIL contains an opioid that can be a target for people who abuse prescription medications or street drugs, to keep their films in a safe place, and to protect them from theft.
- Instruct patients to keep BUNAVAIL in a secure place, out of the sight and reach of children. Accidental or deliberate ingestion by a child may cause respiratory depression that can result in death. Advise patients to seek medical attention if a child is exposed to BUNAVAIL.
- Inform patients that opioids could cause a rare but potentially life-threatening condition resulting from concomitant administration of serotonergic drugs. Warn patients of the symptoms of serotonin syndrome and to seek medical attention right away if symptoms develop. Instruct patients to inform their healthcare providers if they are taking, or plan to take, serotonergic medications [see Drug Interactions (7)].
- Inform patients that opioids could cause adrenal insufficiency, a potentially life-threatening condition. Adrenal insufficiency may present with non-specific symptoms and signs such as nausea, vomiting, anorexia, fatigue, weakness, dizziness, and low blood pressure. Advise patients to seek medical attention if they experience a constellation of these symptoms [see Warnings and Precautions (5.6)].
- Advise patients to never give BUNAVAIL to anyone else, even if he or she has the same signs and symptoms. It may cause harm or death.
- Advise patients that selling or giving away this medication is against the law.
- Advise patients that, after BUNAVAIL has completely dissolved in the oral mucosa, to take a sip of water, swish it gently around their teeth and gums, and swallow. Advise patients to wait for at least one hour after taking BUNAVAIL before brushing teeth [see Warnings and Precautions (5.13)].
- Refer patients to dental care services and encourage them to have regular dental checkups while taking BUNAVAIL. Instruct patients to inform their dentist that they have started therapy on BUNAVAIL [see Warnings and Precautions (5.13)].
- Caution patients that BUNAVAIL may impair the mental or physical abilities required for the performance of potentially dangerous tasks such as driving or operating machinery. Caution should be taken especially during drug induction and dose adjustment and until individuals are reasonably certain that buprenorphine therapy does not adversely affect their ability to engage in such activities [see Warnings and Precautions (5.15)].
- Advise patients that they should not change the dosage of BUNAVAIL without consulting their healthcare provider.
- Advise patients to take BUNAVAIL once a day.
- Advise patients that if they miss a dose of BUNAVAIL they should take it as soon as they remember. If it is almost time for the next dose, they should skip the missed dose and take the next dose at the regular time.
- Inform patients that BUNAVAIL can cause drug dependence and that withdrawal signs and symptoms may occur when the medication is discontinued.
- Advise patients seeking to discontinue treatment with buprenorphine for opioid dependence to work closely with their healthcare provider on a tapering schedule and inform them of the potential to relapse to illicit drug use associated with discontinuation of opioid agonist/partial agonist medication-assisted treatment.
- Advise patients that, like other opioids, BUNAVAIL may produce orthostatic hypotension in ambulatory individuals [see Warnings and Precautions (5.16)].
- Advise patients to inform their healthcare provider if any other prescription medications, over-the-counter medications, or herbal preparations are prescribed or currently being used [see Drug Interactions (7)].
- Advise women that if they are pregnant while being treated with BUNAVAIL, the baby may have signs of withdrawal at birth and that appropriate treatment is available [see Warnings and Precautions (5.5), Use in Specific Populations (8.1)].
- Inform patients that chronic use of opioids may cause reduced fertility. It is not known whether these effects on fertility are reversible [see Nonclinical Toxicology (13.1)].
- Advise women who are breastfeeding to monitor the infant for drowsiness and difficulty breathing [see Use in Specific Populations (8.2)].
- Advise patients to inform their family members that, in the event of emergency, the treating healthcare provider or emergency room staff should be informed that the patient is physically dependent on an opioid and that the patient is being treated with BUNAVAIL buccal films.
Manufactured for:
BioDelivery Sciences International, Inc., Raleigh, North Carolina 27612 USA
BUN-001-PI-Jun2022
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: June 2022 | ||||
| Medication Guide BUNAVAIL® (bue-nah-vale) (buprenorphine and naloxone buccal film), CIII |
|||||
| IMPORTANT: Keep BUNAVAIL in a secure place away from children. Accidental use by a child is a medical emergency and can result in death. If a child accidentally uses BUNAVAIL, get emergency help or call 911 right away. Tell your healthcare provider if you are living in a household where there are small children. | |||||
What is the most important information I should know about BUNAVAIL?
|
|||||
|
|
||||
|
|||||
What is BUNAVAIL?
|
|||||
Who should not take BUNAVAIL?
|
|||||
| Before taking BUNAVAIL, tell your healthcare provider about all of your medical conditions, including if you have: | |||||
|
|
|
|||
Tell your healthcare provider if you are:
|
|||||
How should I take BUNAVAIL?
|
|||||
Read the Instructions for Use at the end of this Medication Guide for detailed instructions on how to take BUNAVAIL.
|
|||||
What should I avoid while taking BUNAVAIL?
|
|||||
| What are possible side effects of BUNAVAIL? BUNAVAIL can cause serious side effects including:
|
|||||
|
|
||||
| Your healthcare provider should do blood tests to check the liver before you start taking and while you take BUNAVAIL. | |||||
|
|||||
|
|
||||
|
|||||
|
|
||||
| BUNAVAIL sublingual film may affect fertility in males and females. Talk to your healthcare provider if this is a concern for you. These are not all the possible side effects of BUNAVAIL. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||||
| General information about the safe and effective use of BUNAVAIL.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use BUNAVAIL for a condition for which it was not prescribed. Do not give BUNAVAIL to other people, even if they have the same symptoms you have. It may harm them, and it is against the law. You can ask your pharmacist or healthcare provider or for information that is written for health professionals. |
|||||
INSTRUCTIONS FOR USE
BUNAVAIL® (bue-nah-vale) (buprenorphine and naloxone buccal film), CIII
This "Instructions for Use" contains information on how to correctly take BUNAVAIL sublingual film.
Important Information You Need to Know Before Taking BUNAVAIL sublingual film:
- Always take BUNAVAIL exactly as your doctor tells you. Your doctor may change your dose after seeing how it affects you. Do not change your dose unless your doctor tells you to change it.
- Do not take BUNAVAIL more often than prescribed by your doctor.
- After induction (your first few days of dosing), take BUNAVAIL 1 time a day.
- You may be prescribed a dose of 2 or more BUNAVAIL buccal films to be taken at the same time.
- Use the entire BUNAVAIL buccal film. Do not cut, tear, chew, or swallow the buccal film. Your doctor should show you how to use BUNAVAIL the right way.
- Follow the same instructions every time you take a dose of BUNAVAIL buccal film.
- BUNAVAIL comes in a child-resistant foil package. Do not open the foil package until you are ready to use the BUNAVAIL buccal film. After opening the foil package, use BUNAVAIL buccal film right away.
Take the dose prescribed by your doctor as follows:- To open the BUNAVAIL buccal film foil package, fold along the dotted lines and tear down at slits or cut with scissors in the direction of the arrows (see Figure A).

FIGURE A
- To open the BUNAVAIL buccal film foil package, fold along the dotted lines and tear down at slits or cut with scissors in the direction of the arrows (see Figure A).
- Use your tongue to wet the inside of your cheek or rinse your mouth with water to moisten the area in your mouth before you place BUNAVAIL.
- Hold the BUNAVAIL buccal film with clean, dry fingers with the text (BN2, BN4, or BN6) facing up (see Figure B).
- Place the BUNAVAIL buccal film inside your mouth with the text (BN2, BN4, or BN6) against the inside of your moistened cheek (see Figure C).
- With your finger, press the BUNAVAIL buccal film against your cheek. Hold it there for 5 seconds.
- Take your finger away from the BUNAVAIL buccal film. It will stick to the inside of your cheek (see Figure D).
- Leave the film in place until it dissolves.
- If your doctor tells you to use more than one BUNAVAIL buccal film at the same time, place the next film on the inside of your other cheek following the instructions above. Do not place more than two buccal films to the inside of one cheek at a time.
Figure B Figure C Figure D
- Avoid touching or moving the buccal film with your tongue or fingers.
- Avoid drinking or eating food until after the buccal film has dissolved.
- After BUNAVAIL is completely dissolved, rinse your mouth with water and swallow. Wait for at least one hour before brushing teeth.
- If you miss a dose of BUNAVAIL, take your medicine when you remember. If it is almost time for your next dose, skip the missed dose and take the next dose at your regular time. Do not take 2 doses at the same time unless your doctor tells you to. If you are not sure about your dosing, call your doctor.
- Do not stop taking BUNAVAIL suddenly. You could become sick and have withdrawal symptoms because your body has become used to the medicine. Physical dependence is not the same as drug addiction. Your doctor can tell you more about the differences between physical dependence and drug addiction. To have fewer withdrawal symptoms, ask your doctor how to stop using BUNAVAIL the right way.
- If you take too much BUNAVAIL or overdose, call Poison Control or get emergency medical help right away.
Storing BUNAVAIL
- Store BUNAVAIL at 20°C to 25°C (68°F to 77°F), with excursions permitted between 15°C and 30°C (59°F and 86°F).
- Keep BUNAVAIL dry.
- Do not freeze BUNAVAIL.
- Do not use BUNAVAIL buccal film if the foil package has been damaged.
- Keep BUNAVAIL in a safe place, out of sight and reach of children.
Disposing of BUNAVAIL
- Dispose of expired, unwanted, or unused BUNAVAIL by removing the BUNAVAIL film from the foil packaging, and promptly flushing down the toilet (if a drug takeback option is not readily available). Visit http://www.fda.gov/drugdisposal for additional information on disposal of unused medicines.
- If you need help with disposal of BUNAVAIL, call 1-800-469-0261.
This "Instructions for Use" has been approved by the U.S. Food and Drug Administration. BUN-001-MG-Jun2022
PRINCIPAL DISPLAY PANEL - 2.1 mg/0.3 mg Film Pouch Carton
NDC 59385-012-30
30 films/carton
CIII
Bunavail®
(buprenorphine and naloxone buccal film)
2.1 mg / 0.3 mg
Each buccal film contains
2.1 mg buprenorphine and 0.3 mg naloxone
Rx Only
Dispense the enclosed Medication Guide to each patient.
Use entire film. Do not cut, tear, chew, or swallow film.
Keep Bunavail out of the sight and reach of children.
Children who accidentally take Bunavail
will need emergency medical care.
www.Bunavail.com
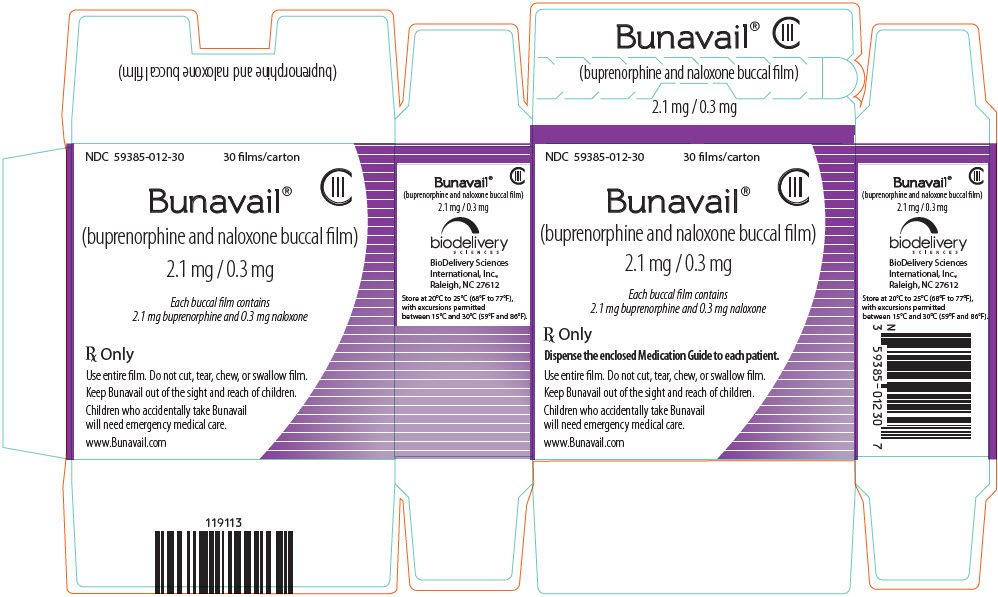
PRINCIPAL DISPLAY PANEL - 4.2 mg/0.7 mg Film Pouch Carton
NDC 59385-014-30
30 films/carton
CIII
Bunavail®
(buprenorphine and naloxone buccal film)
4.2 mg / 0.7 mg
Each buccal film contains
4.2 mg buprenorphine and 0.7 mg naloxone
Rx Only
Dispense the enclosed Medication Guide to each patient.
Use entire film. Do not cut, tear, chew, or swallow film.
Keep Bunavail out of the sight and reach of children.
Children who accidentally take Bunavail
will need emergency medical care.
www.Bunavail.com
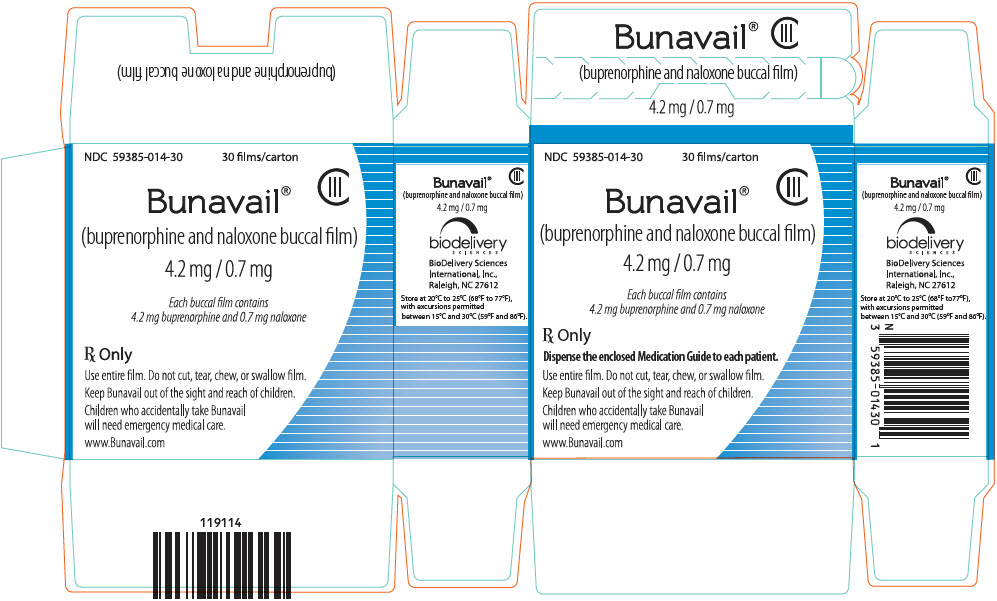
PRINCIPAL DISPLAY PANEL - 6.3 mg/1 mg Film Pouch Carton
NDC 59385-016-30
30 films/carton
CIII
Bunavail®
(buprenorphine and naloxone buccal film)
6.3 mg / 1 mg
Each buccal film contains
6.3 mg buprenorphine and 1 mg naloxone
Rx Only
Dispense the enclosed Medication Guide to each patient.
Use entire film. Do not cut, tear, chew, or swallow film.
Keep Bunavail out of the sight and reach of children.
Children who accidentally take Bunavail
will need emergency medical care.
www.Bunavail.com
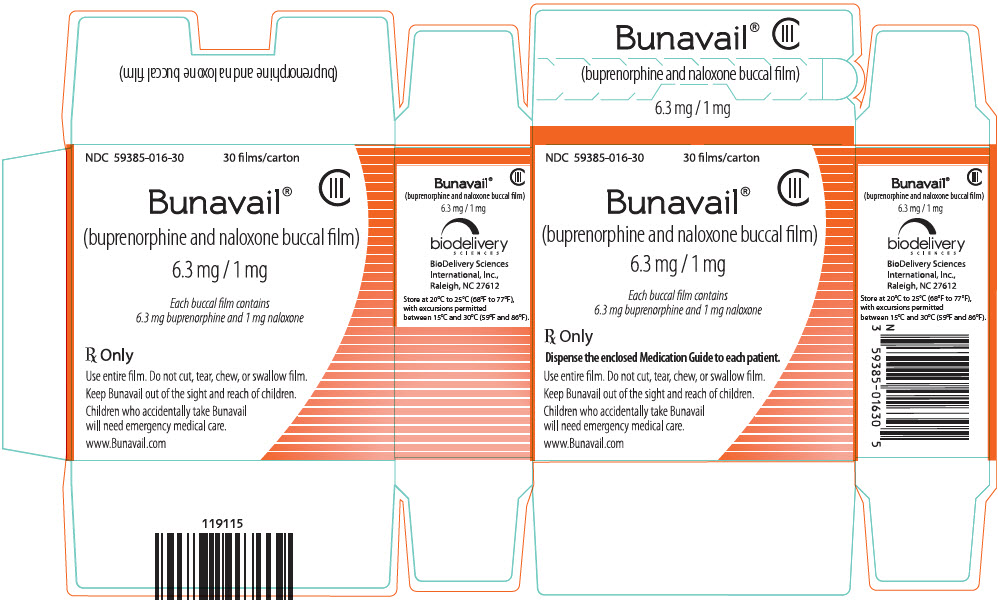
| BUNAVAIL
buprenorphine and naloxone film |
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
| BUNAVAIL
buprenorphine and naloxone film |
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
| BUNAVAIL
buprenorphine and naloxone film |
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
| Labeler - BioDelivery Sciences International, Inc. (016058955) |