Label: CHLORPROMAZINE HYDROCHLORIDE injection
- NDC Code(s): 55150-318-01, 55150-318-25, 55150-319-01, 55150-319-25
- Packager: Eugia US LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
Drug Label Information
Updated August 16, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
BOXED WARNING
(What is this?)
WARNING
Increased Mortality in Elderly Patients with Dementia-Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of seventeen placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug- treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. Observational studies suggest that, similar to atypical antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality. The extent to which the findings of increased mortality in observational studies may be attributed to the antipsychotic drug as opposed to some characteristic(s) of the patients is not clear.
Chlorpromazine hydrochloride injection is not approved for the treatment of patients with dementia-related psychosis (see WARNINGS). -
DESCRIPTION
Chlorpromazine hydrochloride, USP is white or slightly creamy white, crystalline powder chemically designated as 2-Chloro-10-[3-(dimethylamino)propyl]-phenothiazine monohydrochloride and has the following structural formula:
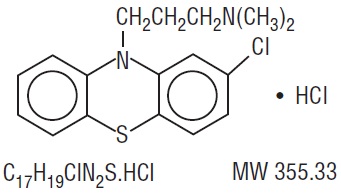
Chlorpromazine hydrochloride injection, USP is a clear, slightly yellow colored solution intended for deep intramuscular use. Each mL contains chlorpromazine hydrochloride USP 25 mg, ascorbic acid 2 mg, sodium metabisulfite 1 mg, sodium sulfite 1 mg and sodium chloride 6 mg in Water for Injection. pH is between 3.4 to 5.4. -
CLINICAL PHARMACOLOGY
The precise mechanism whereby the therapeutic effects of chlorpromazine are produced is not known. The principal pharmacological actions are psychotropic. It also exerts sedative and antiemetic activity. Chlorpromazine has actions at all levels of the central nervous system-primarily at subcortical levels-as well as on multiple organ systems.
Chlorpromazine has strong antiadrenergic and weaker peripheral anticholinergic activity; ganglionic blocking action is relatively slight. It also possesses slight antihistaminic and antiserotonin activity. -
INDICATIONS AND USAGE
For the treatment of schizophrenia; to control nausea and vomiting; for relief of restlessness and apprehension before surgery; for acute intermittent porphyria; as an adjunct in the treatment of tetanus; to control the manifestations of the manic type of manic-depressive illness; for relief of intractable hiccups; for the treatment of severe behavioral problems in children (1 to 12 years of age) marked by combativeness and/or explosive hyperexcitable behavior (out of proportion to immediate provocations), and in the short-term treatment of hyperactive children who show excessive motor activity with accompanying conduct disorders consisting of some or all of the following symptoms: impulsivity, difficulty sustaining attention, aggressivity, mood lability, and poor frustration tolerance.
- CONTRAINDICATIONS
-
WARNINGS
Increased Mortality in Elderly Patients with Dementia-Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Chlorpromazine hydrochloride injection is not approved for the treatment of patients with dementia-related psychosis (see BOXED WARNING).
The extrapyramidal symptoms which can occur secondary to chlorpromazine may be confused with the central nervous system signs of an undiagnosed primary disease responsible for the vomiting, e.g., Reye’s syndrome or other encephalopathy. The use of chlorpromazine and other potential hepatotoxins should be avoided in children and adolescents whose signs and symptoms suggest Reye’s syndrome.
Chlorpromazine hydrochloride injection contains sodium metabisulfite and sodium sulfite, sulfites that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in nonasthmatic people.
Tardive Dyskinesia
Tardive dyskinesia, a syndrome consisting of potentially irreversible, involuntary, dyskinetic movements, may develop in patients treated with antipsychotic drugs. Although the prevalence of the syndrome appears to be highest among the elderly, especially elderly women, it is impossible to rely upon prevalence estimates to predict, at the inception of antipsychotic treatment, which patients are likely to develop the syndrome. Whether antipsychotic drug products differ in their potential to cause tardive dyskinesia is unknown.
Both the risk of developing the syndrome and the likelihood that it will become irreversible are believed to increase as the duration of treatment and the total cumulative dose of antipsychotic drugs administered to the patient increase. However, the syndrome can develop, although much less commonly, after relatively brief treatment periods at low doses.
There is no known treatment for established cases of tardive dyskinesia, although the syndrome may remit, partially or completely, if antipsychotic treatment is withdrawn. Antipsychotic treatment itself, however, may suppress (or partially suppress) the signs and symptoms of the syndrome and thereby may possibly mask the underlying disease process. The effect that symptomatic suppression has upon the long-term course of the syndrome is unknown.
Given these considerations, antipsychotics should be prescribed in a manner that is most likely to minimize the occurrence of tardive dyskinesia. Chronic antipsychotic treatment should generally be reserved for patients who suffer from a chronic illness that, 1) is known to respond to antipsychotic drugs, and, 2) for whom alternative, equally effective, but potentially less harmful treatments are not available or appropriate. In patients who do require chronic treatment, the smallest dose and the shortest duration of treatment producing a satisfactory clinical response should be sought. The need for continued treatment should be reassessed periodically.
If signs and symptoms of tardive dyskinesia appear in a patient on antipsychotics, drug discontinuation should be considered. However, some patients may require treatment despite the presence of the syndrome.
For further information about the description of tardive dyskinesia and its clinical detection, please refer to the sections on PRECAUTIONS and ADVERSE REACTIONS.
Neuroleptic Malignant Syndrome (NMS)
A potentially fatal symptom complex sometimes referred to as Neuroleptic Malignant Syndrome (NMS) has been reported in association with antipsychotic drugs. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status and evidence of autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis and cardiac dysrhythmias).
The diagnostic evaluation of patients with this syndrome is complicated. In arriving at a diagnosis, it is important to identify cases where the clinical presentation includes both serious medical illness (e.g., pneumonia, systemic infection, etc.) and untreated or inadequately treated extrapyramidal signs and symptoms (EPS). Other important considerations in the differential diagnosis include central anticholinergic toxicity, heat stroke, drug fever and primary central nervous system (CNS) pathology.
The management of NMS should include 1) immediate discontinuation of antipsychotic drugs and other drugs not essential to concurrent therapy, 2) intensive symptomatic treatment and medical monitoring and 3) treatment of any concomitant serious medical problems for which specific treatments are available. There is no general agreement about specific pharmacological treatment regimens for uncomplicated NMS.
If a patient requires antipsychotic drug treatment after recovery from NMS, the potential reintroduction of drug therapy should be carefully considered. The patient should be carefully monitored, since recurrences of NMS have been reported.
An encephalopathic syndrome (characterized by weakness, lethargy, fever, tremulousness and confusion, extrapyramidal symptoms, leukocytosis, elevated serum enzymes, BUN and FBS) has occurred in a few patients treated with lithium plus an antipsychotic. In some instances, the syndrome was followed by irreversible brain damage. Because of a possible causal relationship between these events and the concomitant administration of lithium and antipsychotics, patients receiving such combined therapy should be monitored closely for early evidence of neurologic toxicity and treatment discontinued promptly if such signs appear. This encephalopathic syndrome may be similar to or the same as neuroleptic malignant syndrome (NMS).
Patients with bone marrow depression or who have previously demonstrated a hypersensitivity reaction (e.g., blood dyscrasias, jaundice) with a phenothiazine should not receive any phenothiazine, including chlorpromazine, unless in the judgment of the physician the potential benefits of treatment outweigh the possible hazard.
Chlorpromazine may impair mental and/or physical abilities, especially during the first few days of therapy. Therefore, caution patients about activities requiring alertness (e.g., operating vehicles or machinery).
The use of alcohol with this drug should be avoided due to possible additive effects and hypotension.
Chlorpromazine may counteract the antihypertensive effect of guanethidine and related compounds.
Falls
Chlorpromazine hydrochloride injection may cause somnolence, postural hypotension, motor and sensory instability, which may lead to falls and, consequently, fractures or other injuries. For patients with diseases, conditions, or medications that could exacerbate these effects, complete fall risk assessments when initiating antipsychotic treatment and recurrently for patients on long-term antipsychotic therapy.
Pregnancy
NON-TERATOGENIC EFFECTS
Neonates exposed to antipsychotic drugs, during the third trimester of pregnancy are at risk for extrapyramidal and/or withdrawal symptoms following delivery. There have been reports of agitation, hypertonia, hypotonia, tremor, somnolence, respiratory distress and feeding disorder in these neonates. These complications have varied in severity; while in some cases symptoms have been self-limited, in other cases neonates have required intensive care unit support and prolonged hospitalization.
Chlorpromazine hydrochloride should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Usage in Pregnancy
Safety for the use of chlorpromazine during pregnancy has not been established. Therefore, it is not recommended that the drug be given to pregnant patients except when, in the judgment of the physician, it is essential. The potential benefits should clearly outweigh possible hazards. There are reported instances of prolonged jaundice, extrapyramidal signs, hyperreflexia or hyporeflexia in newborn infants whose mothers received phenothiazines.
Reproductive studies in rodents have demonstrated potential for embryotoxicity, increased neonatal mortality and nursing transfer of the drug. Tests in the offspring of the drug-treated rodents demonstrate decreased performance. The possibility of permanent neurological damage cannot be excluded.
Nursing Mothers
There is evidence that chlorpromazine is excreted in the breast milk of nursing mothers. Because of the potential for serious adverse reactions in nursing infants from chlorpromazine, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother. -
PRECAUTIONS
Leukopenia, Neutropenia and Agranulocytosis
In clinical trial and postmarketing experience, events of leukopenia/neutropenia and agranulocytosis have been reported temporally related to antipsychotic agents.
Possible risk factors for leukopenia/neutropenia include preexisting low white blood cell count (WBC) and history of drug induced leukopenia/neutropenia. Patients with a preexisting low WBC or a history of drug induced leukopenia/neutropenia should have their complete blood count (CBC) monitored frequently during the first few months of therapy and should discontinue chlorpromazine hydrochloride injection at the first sign of a decline in WBC in the absence of other causative factors.
Patients with neutropenia should be carefully monitored for fever or other symptoms or signs of infection and treated promptly if such symptoms or signs occur. Patients with severe neutropenia (absolute neutrophil count <1000/mm3) should discontinue chlorpromazine hydrochloride injection and have their WBC followed until recovery.General
Given the likelihood that some patients exposed chronically to antipsychotics will develop tardive dyskinesia, it is advised that all patients in whom chronic use is contemplated be given, if possible, full information about this risk. The decision to inform patients and/or their guardians must obviously take into account the clinical circumstances and the competency of the patient to understand the information provided.
Chlorpromazine should be administered cautiously to persons with cardiovascular, liver or renal disease. There is evidence that patients with a history of hepatic encephalopathy due to cirrhosis have increased sensitivity to the CNS effects of chlorpromazine (i.e., impaired cerebration and abnormal slowing of the EEG).
Because of its CNS depressant effect, chlorpromazine should be used with caution in patients with chronic respiratory disorders such as severe asthma, emphysema and acute respiratory infections, particularly in children (1 to 12 years of age).
Because chlorpromazine can suppress the cough reflex, aspiration of vomitus is possible.
Chlorpromazine prolongs and intensifies the action of CNS depressants such as anesthetics, barbiturates and narcotics. When chlorpromazine is administered concomitantly, about 1/4 to 1/2 the usual dosage of such agents is required. When chlorpromazine is not being administered to reduce requirements of CNS depressants, it is best to stop such depressants before starting chlorpromazine treatment. These agents may subsequently be reinstated at low doses and increased as needed.
Note: Chlorpromazine does not intensify the anticonvulsant action of barbiturates. Therefore, dosage of anticonvulsants, including barbiturates, should not be reduced if chlorpromazine is started. Instead, start chlorpromazine at low doses and increase as needed.
Use with caution in persons who will be exposed to extreme heat, organophosphorus insecticides and in persons receiving atropine or related drugs.
Antipsychotic drugs elevate prolactin levels; the elevation persists during chronic administration. Tissue culture experiments indicate that approximately 1/3 of human breast cancers are prolactin- dependent in vitro, a factor of potential importance if the prescribing of these drugs is contemplated in a patient with a previously detected breast cancer. Although disturbances such as galactorrhea, amenorrhea, gynecomastia and impotence have been reported, the clinical significance of elevated serum prolactin levels is unknown for most patients. An increase in mammary neoplasms has been found in rodents after chronic administration of antipsychotic drugs. Neither clinical nor epidemiologic studies conducted to date, however, have shown an association between chronic administration of these drugs and mammary tumorigenesis; the available evidence is considered too limited to be conclusive at this time.
Chromosomal aberrations in spermatocytes and abnormal sperm have been demonstrated in rodents treated with certain antipsychotics.
As with all drugs which exert an anticholinergic effect, and/or cause mydriasis, chlorpromazine should be used with caution in patients with glaucoma.
Chlorpromazine diminishes the effect of oral anticoagulants.
Phenothiazines can produce alpha-adrenergic blockade.
Chlorpromazine may lower the convulsive threshold; dosage adjustments of anticonvulsants may be necessary. Potentiation of anticonvulsant effects does not occur. However, it has been reported that chlorpromazine may interfere with the metabolism of phenytoin and thus precipitate phenytoin toxicity.
Concomitant administration with propranolol results in increased plasma levels of both drugs.
Thiazide diuretics may accentuate the orthostatic hypotension that may occur with phenothiazines.
The presence of phenothiazines may produce false-positive phenylketonuria (PKU) test results.
Drugs which lower the seizure threshold, including phenothiazine derivatives, should not be used with metrizamide. As with other phenothiazine derivatives, chlorpromazine should be discontinued at least 48 hours before myelography, should not be resumed for at least 24 hours postprocedure, and should not be used for the control of nausea and vomiting occurring either prior to myelography or postprocedure with metrizamide.
Long-Term Therapy
To lessen the likelihood of adverse reactions related to cumulative drug effect, patients with a history of long-term therapy with chlorpromazine and/or other antipsychotics should be evaluated periodically to decide whether the maintenance dosage could be lowered or drug therapy discontinued.
Antiemetic Effect
The antiemetic action of chlorpromazine may mask the signs and symptoms of overdosage of other drugs and may obscure the diagnosis and treatment of other conditions such as intestinal obstruction, brain tumor and Reye’s syndrome. (See WARNINGS.)
When chlorpromazine is used with cancer chemotherapeutic drugs, vomiting as a sign of the toxicity of these agents may be obscured by the antiemetic effects of chlorpromazine.
Abrupt Withdrawal
Like other phenothiazines, chlorpromazine is not known to cause psychic dependence and does not produce tolerance or addiction. There may be, however, following abrupt withdrawal of high-dose therapy, some symptoms resembling those of physical dependence such as gastritis, nausea and vomiting, dizziness and tremulousness. These symptoms can usually be avoided or reduced by gradual reduction of the dosage or by continuing concomitant anti-parkinsonism agents for several weeks after chlorpromazine is withdrawn. -
ADVERSE REACTIONS
Note: Some adverse effects of chlorpromazine may be more likely to occur, or occur with greater intensity, in patients with special medical problems, e.g., patients with mitral insufficiency or pheochromocytoma have experienced severe hypotension following recommended doses.
Drowsiness, usually mild to moderate, may occur, particularly during the first or second week, after which it generally disappears. If troublesome, dosage may be lowered.
Jaundice
Overall incidence has been low, regardless of indication or dosage. Most investigators conclude it is a sensitivity reaction. Most cases occur between the second and fourth weeks of therapy. The clinical picture resembles infectious hepatitis, with laboratory features of obstructive jaundice, rather than those of parenchymal damage. It is usually promptly reversible on withdrawal of the medication; however, chronic jaundice has been reported.
There is no conclusive evidence that preexisting liver disease makes patients more susceptible to jaundice. Alcoholics with cirrhosis have been successfully treated with chlorpromazine without complications. Nevertheless, the medication should be used cautiously in patients with liver disease. Patients who have experienced jaundice with a phenothiazine should not, if possible, be reexposed to chlorpromazine or other phenothiazines.
If fever with grippe-like symptoms occurs, appropriate liver studies should be conducted. If tests indicate an abnormality, stop treatment.
Liver function tests in jaundice induced by the drug may mimic extrahepatic obstruction; withhold exploratory laparotomy until extrahepatic obstruction is confirmed.
Hematological Disorders, including agranulocytosis, eosinophilia, leukopenia, hemolytic anemia, aplastic anemia, thrombocytopenic purpura and pancytopenia have been reported.
AGRANULOCYTOSIS
Warn patients to report the sudden appearance of sore throat or other signs of infection. If white blood cell and differential counts indicate cellular depression, stop treatment and start antibiotic and other suitable therapy.
Most cases have occurred between the 4th and 10th weeks of therapy; patients should be watched closely during that period.
Moderate suppression of white blood cells is not an indication for stopping treatment unless accompanied by the symptoms described above.
Cardiovascular
HYPOTENSIVE EFFECTS
Postural hypotension, simple tachycardia, momentary fainting and dizziness may occur after the first injection; occasionally after subsequent injections; rarely, after the first oral dose. Usually recovery is spontaneous and symptoms disappear within 1/2 to 2 hours. Occasionally, these effects may be more severe and prolonged, producing a shock-like condition.
To minimize hypotension after injection, keep patient lying down and observe for at least 1/2 hour. To control hypotension, place patient in head-low position with legs raised. If a vasoconstrictor is required, norepinephrine and phenylephrine are the most suitable. Other pressor agents, including epinephrine, should not be used as they may cause a paradoxical further lowering of blood pressure.
EKG CHANGES
Particularly nonspecific, usually reversible Q and T wave distortions–have been observed in some patients receiving phenothiazine tranquilizers, including chlorpromazine.
Note: Sudden death, apparently due to cardiac arrest, has been reported.
CNS Reactions
NEUROMUSCULAR (EXTRAPYRAMIDAL) REACTIONS
Neuromuscular reactions include dystonia, motor restlessness, pseudo-parkinsonism and tardive dyskinesia, and appear to be dose-related. They are discussed in the following paragraphs:
Dystonia
Class effect: Symptoms of dystonia, prolonged abnormal contractions of muscle groups, may occur in susceptible individuals during the first few days of treatment. Dystonic symptoms include: spasm of the neck muscles, sometimes progressing to tightness of the throat, swallowing difficulty, difficulty breathing, and/or protrusion of the tongue. While these symptoms can occur at low doses, they occur more frequently and with greater severity with high potency and at higher doses of first generation antipsychotic drugs. An elevated risk of acute dystonia is observed in males and younger age groups.
Motor Restlessness
Symptoms may include agitation or jitteriness and sometimes insomnia. These symptoms often disappear spontaneously. At times these symptoms may be similar to the original neurotic or psychotic symptoms. Dosage should not be increased until these side effects have subsided.
If these symptoms become too troublesome, they can usually be controlled by a reduction of dosage or change of drug. Treatment with anti-parkinsonian agents, benzodiazepines or propranolol may be helpful.
Pseudo-parkinsonism
Symptoms may include: mask-like facies, drooling, tremors, pillrolling motion, cogwheel rigidity and shuffling gait. In most cases, these symptoms are readily controlled when an anti-parkinsonism agent is administered concomitantly. Anti-parkinsonism agents should be used only when required. Generally, therapy of a few weeks to 2 or 3 months will suffice. After this time, patients should be evaluated to determine their need for continued treatment. (Note: Levodopa has not been found effective in antipsychotic-induced pseudo-parkinsonism.) Occasionally it is necessary to lower the dosage of chlorpromazine or to discontinue the drug.
Tardive Dyskinesia
As with all antipsychotic agents, tardive dyskinesia may appear in some patients on long-term therapy or may appear after drug therapy has been discontinued. The syndrome can also develop, although much less frequently, after relatively brief treatment periods at low doses. This syndrome appears in all age groups. Although its prevalence appears to be highest among elderly patients, especially elderly women, it is impossible to rely upon prevalence estimates to predict at the inception of antipsychotic treatment which patients are likely to develop the syndrome. The symptoms are persistent and in some patients appear to be irreversible. The syndrome is characterized by rhythmical involuntary movements of the tongue, face, mouth or jaw (e.g., protrusion of tongue, puffing of cheeks, puckering of mouth, chewing movements). Sometimes these may be accompanied by involuntary movements of extremities. In rare instances, these involuntary movements of the extremities are the only manifestations of tardive dyskinesia. A variant of tardive dyskinesia, tardive dystonia, has also been described.
There is no known effective treatment for tardive dyskinesia; anti-parkinsonism agents do not alleviate the symptoms of this syndrome. If clinically feasible, it is suggested that all antipsychotic agents be discontinued if these symptoms appear. Should it be necessary to reinstitute treatment, or increase the dosage of the agent, or switch to a different antipsychotic agent, the syndrome may be masked. It has been reported that fine vermicular movements of the tongue may be an early sign of the syndrome and if the medication is stopped at that time the syndrome may not develop.
ADVERSE BEHAVIORAL EFFECTS
Psychotic symptoms and catatonic-like states have been reported rarely.
OTHER CNS EFFECTS
Neuroleptic Malignant Syndrome (NMS) has been reported in association with antipsychotic drugs (see WARNINGS).
Cerebral edema has been reported.
Convulsive seizures (petit mal and grand mal) have been reported, particularly in patients with EEG abnormalities or history of such disorders.
Abnormality of the cerebrospinal fluid proteins has also been reported.
Allergic Reactions of a mild urticarial type or photosensitivity are seen. Avoid undue exposure to sun. More severe reactions, including exfoliative dermatitis and toxic epidermal necrolysis (TEN), have been reported occasionally.
Contact dermatitis has been reported in nursing personnel; accordingly, the use of rubber gloves when administering chlorpromazine injectable is recommended.
In addition, asthma, laryngeal edema, angioneurotic edema and anaphylactoid reactions have been reported.
Endocrine Disorders
Lactation and moderate breast engorgement may occur in females on large doses. If persistent, lower dosage or withdraw drug. False-positive pregnancy tests have been reported, but are less likely to occur when a serum test is used. Amenorrhea and gynecomastia have also been reported.
Hyperglycemia, hypoglycemia and glycosuria have been reported.
Autonomic Reactions
Occasional dry mouth; nasal congestion; nausea; obstipation; constipation; adynamic ileus; urinary retention; priapism; miosis and mydriasis; atonic colon; ejaculatory disorders/impotence.
Special Considerations in Long-Term Therapy
Skin pigmentation and ocular changes have occurred in some patients taking substantial doses of chlorpromazine for prolonged periods.
SKIN PIGMENTATION
Rare instances of skin pigmentation have been observed in hospitalized mental patients, primarily females who have received the drug usually for 3 years or more in dosages ranging from 500 mg to 1,500 mg daily. The pigmentary changes, restricted to exposed areas of the body, range from an almost imperceptible darkening of the skin to a slate gray color, sometimes with a violet hue. Histological examination reveals a pigment, chiefly in the dermis, which is probably a melanin-like complex. The pigmentation may fade following discontinuance of the drug.
OCULAR CHANGES
Ocular changes have occurred more frequently than skin pigmentation and have been observed both in pigmented and nonpigmented patients receiving chlorpromazine usually for 2 years or more in dosages of 300 mg daily and higher. Eye changes are characterized by deposition of fine particulate matter in the lens and cornea. In more advanced cases, star-shaped opacities have also been observed in the anterior portion of the lens. The nature of the eye deposits has not yet been determined. A small number of patients with more severe ocular changes have had some visual impairment. In addition to these corneal and lenticular changes, epithelial keratopathy and pigmentary retinopathy have been reported. Reports suggest that the eye lesions may regress after withdrawal of the drug. Since the occurrence of eye changes seems to be related to the dosage levels and/or duration of therapy, it is suggested that long-term patients on moderate to high dosage levels have periodic ocular examinations.
ETIOLOGY
The etiology of both of these reactions is not clear, but exposure to light, along with dosage/duration of therapy, appears to be the most significant factor. If either of these reactions is observed, the physician should weigh the benefits of continued therapy against the possible risks and, on the merits of the individual case, determine whether or not to continue present therapy, lower the dosage, or withdraw the drug.
Other Adverse Reactions
Mild fever may occur after large intramuscular doses. Hyperpyrexia has been reported. Increases in appetite and weight sometimes occur. Peripheral edema and a systemic lupus erythematosus-like syndrome have been reported.
Note: There have been occasional reports of sudden death in patients receiving phenothiazines. In some cases, the cause appeared to be cardiac arrest or asphyxia due to failure of the cough reflex. -
OVERDOSAGE
(See also ADVERSE REACTIONS.)
Symptoms
Primarily symptoms of central nervous system depression to the point of somnolence or coma.
Hypotension and extrapyramidal symptoms.
Other possible manifestations include agitation and restlessness, convulsions, fever, autonomic reactions such as dry mouth and ileus, EKG changes and cardiac arrhythmias.
Treatment
It is important to determine other medications taken by the patient since multiple drug therapy is common in overdosage situations. Treatment is essentially symptomatic and supportive. Early gastric lavage is helpful. Keep patient under observation and maintain an open airway, since involvement of the extrapyramidal mechanism may produce dysphagia and respiratory difficulty in severe overdosage. Do not attempt to induce emesis because a dystonic reaction of the head or neck may develop that could result in aspiration of vomitus. Extrapyramidal symptoms may be treated with anti-parkinsonism drugs, barbiturates or diphenhydramine. See prescribing information for these products. Care should be taken to avoid increasing respiratory depression.
If administration of a stimulant is desirable, amphetamine, dextroamphetamine or caffeine with sodium benzoate is recommended. Stimulants that may cause convulsions (e.g., picrotoxin or pentylenetetrazol) should be avoided.
If hypotension occurs, the standard measures for managing circulatory shock should be initiated. If it is desirable to administer a vasoconstrictor, norepinephrine and phenylephrine are most suitable. Other pressor agents, including epinephrine, are not recommended because phenothiazine derivatives may reverse the usual elevating action of these agents and cause a further lowering of blood pressure. Limited experience indicates that phenothiazines are not dialyzable. -
DOSAGE AND ADMINISTRATION
Adults
Adjust dosage to individual and the severity of his condition, recognizing that the milligram for milligram potency relationship among all dosage forms has not been precisely established clinically. It is important to increase dosage until symptoms are controlled. Dosage should be increased more gradually in debilitated or emaciated patients. In continued therapy, gradually reduce dosage to the lowest effective maintenance level, after symptoms have been controlled for a reasonable period.
Increase parenteral dosage only if hypotension has not occurred. Before using intramuscularly, see Important Notes on Injection.
ELDERLY PATIENTS
In general, dosages in the lower range are sufficient for most elderly patients. Since they appear to be more susceptible to hypotension and neuromuscular reactions, such patients should be observed closely. Dosage should be tailored to the individual, response carefully monitored, and dosage adjusted accordingly. Dosage should be increased more gradually in elderly patients.
PSYCHOTIC DISORDERS
Increase dosage gradually until symptoms are controlled. Maximum improvement may not be seen for weeks or even months. Continue optimum dosage for 2 weeks; then gradually reduce dosage to the lowest effective maintenance level. Daily dosage of 200 mg is not unusual. Some patients require higher dosages (e.g., 800 mg daily is not uncommon in discharged mental patients).
Hospitalized Patients: Acute Schizophrenic or Manic States
Intramuscular: 25 mg (1 mL). If necessary, give additional 25 to 50 mg injection in 1 hour. Increase subsequent intramuscular doses gradually over several days–up to 400 mg q4 to 6h in exceptionally severe cases–until patient is controlled. Usually the patient becomes quiet and cooperative within 24 to 48 hours and oral doses may be substituted.
Prompt Control of Severe Symptoms
Intramuscular: 25 mg (1 mL). If necessary, repeat in 1 hour. Subsequent doses should be oral, 25 to 50 mg tid.
NAUSEA AND VOMITING
Intramuscular: 25 mg (1 mL). If no hypotension occurs, give 25 to 50 mg q3 to 4h prn, until vomiting stops. Then switch to oral dosage.
During Surgery
Intramuscular: 12.5 mg (0.5 mL). Repeat in 1/2 hour if necessary and if no hypotension occurs. Intravenous: 2 mg per fractional injection, at 2-minute intervals. Do not exceed 25 mg. Dilute to 1 mg/mL, i.e., 1 mL (25 mg) mixed with 24 mL of saline.
PRESURGICAL APPREHENSION
Intramuscular: 12.5 to 25 mg (0.5 to 1 mL), 1 to 2 hours before operation.
INTRACTABLE HICCUPS
If symptoms persist for 2 to 3 days after trial with oral therapy, give 25 to 50 mg (1 to 2 mL) intramuscularly. Should symptoms persist, use slow intravenous infusion with patient flat in bed: 25 to 50 mg (1 to 2 mL) in 500 to 1,000 mL of saline. Follow blood pressure closely.
ACUTE INTERMITTENT PORPHYRIA
Intramuscular: 25 mg (1 mL) tid or qid until patient can take oral therapy.
TETANUS
Intramuscular: 25 to 50 mg (1 to 2 mL) given 3 or 4 times daily, usually in conjunction with barbiturates. Total doses and frequency of administration must be determined by the patient’s response, starting with low doses and increasing gradually. Intravenous: 25 to 50 mg (1 to 2 mL). Dilute to at least 1 mg per mL and administer at a rate of 1 mg per minute.
Pediatric Patients (6 months to 12 years of age)
Chlorpromazine should generally not be used in pediatric patients under 6 months of age except where potentially lifesaving. It should not be used in conditions for which specific pediatric dosages have not been established.
SEVERE BEHAVIORAL PROBLEMS
Outpatients
Select route of administration according to severity of patient’s condition and increase dosage gradually as required. Intramuscular: 1/4 mg/lb body weight q6 to 8h, prn.
Hospitalized Patients
As with outpatients, start with low doses and increase dosage gradually. In severe behavior disorders, higher dosages (50 to 100 mg daily, and in older children, 200 mg daily or more) may be necessary. There is little evidence that behavior improvement in severely disturbed mentally retarded patients is further enhanced by doses beyond 500 mg per day. Maximum Intramuscular Dosage: Patients up to 5 years (or 50 lbs.), not over 40 mg/day; 5 to 12 years (or 50 to 100 lbs.), not over 75 mg/day except in unmanageable cases.
NAUSEA AND VOMITING
Dosage and frequency of administration should be adjusted according to the severity of the symptoms and response of the patient. The duration of activity following intramuscular administration may last up to 12 hours. Subsequent doses may be given by the same route if necessary. Intramuscular: 1/4 mg/lb body weight q6 to 8h, prn. Maximum Intramuscular Dosage: Pediatric patients 6 months to 5 years (or 50 lbs.), not over 40 mg/day; 5 to 12 years (or 50 to 100 lbs.), not over 75 mg/day except in severe cases.
During Surgery
Intramuscular: 1/8 mg/lb body weight. Repeat in 1/2 hour if necessary and if no hypotension occurs. Intravenous: 1 mg per fractional injection at 2-minute intervals and not exceeding recommended intramuscular dosage. Always dilute to 1 mg/mL, i.e., 1 mL (25 mg) mixed with 24 mL of saline.
PRESURGICAL APPREHENSION
1/4 mg/lb body weight Intramuscular 1 to 2 hours before operation.
TETANUS
Intramuscular or Intravenous: 1/4 mg/lb body weight q6 to 8h. When given intravenously, dilute to at least 1 mg/mL and administer at a rate of 1 mg per 2 minutes. In patients up to 50 lbs., do not exceed 40 mg daily; 50 to 100 lbs., do not exceed 75 mg except in severe cases.
Important Notes on Injection
Inject slowly, deep into upper outer quadrant of buttock.
Because of possible hypotensive effects, reserve parenteral administration for bedfast patients or for acute ambulatory cases, and keep patient lying down for at least 1/2 hour after injection. If irritation is a problem, dilute injection with saline or 2% procaine; mixing with other agents in the syringe is not recommended. Subcutaneous injection is not advised. AVOID INJECTING UNDILUTED CHLORPROMAZINE HYDROCHLORIDE INJECTION INTO VEIN. INTRAVENOUS ROUTE IS ONLY FOR SEVERE HICCUPS, SURGERY AND TETANUS.
Because of the possibility of contact dermatitis, avoid getting solution on hands or clothing.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. -
HOW SUPPLIED
Chlorpromazine hydrochloride injection USP, 25 mg/mL is a clear, slightly yellow colored solution essentially free from visible particles and is available in the following packages:
25 mg per mL
1 mL ampule NDC 55150-318-25
packaged in carton of 25
50 mg per 2 mL (25 mg / mL)
2 mL ampule NDC 55150-319-25
packaged in carton of 25
Storage
Protect from light, or discoloration may occur. Slight yellowing will not alter potency. Discard if markedly discolored. Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Protect from freezing.
To report SUSPECTED ADVERSE REACTIONS, contact AuroMedics Pharma LLC at 1-866-850-2876, or the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Distributed by:
AuroMedics Pharma LLC
279 Princeton-Hightstown Rd.
E. Windsor, NJ 08520
Manufactured by:
Eugia Pharma Specialities Limited
Hyderabad - 500032
India
Revised: October 2021 - PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 25 mg per mL - 1 mL Ampule Label
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 25 mg per mL - 1 mL Container-Carton [25 Ampules]
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 50 mg per 2 mL (25 mg/mL) - 2 mL Ampule Label
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 50 mg per 2 mL (25 mg/mL) - 2 mL Container-Carton [25 Ampules]
-
INGREDIENTS AND APPEARANCE
CHLORPROMAZINE HYDROCHLORIDE
chlorpromazine hydrochloride injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:55150-318 Route of Administration INTRAMUSCULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CHLORPROMAZINE HYDROCHLORIDE (UNII: 9WP59609J6) (CHLORPROMAZINE - UNII:U42B7VYA4P) CHLORPROMAZINE HYDROCHLORIDE 25 mg in 1 mL Inactive Ingredients Ingredient Name Strength ASCORBIC ACID (UNII: PQ6CK8PD0R) 2 mg in 1 mL SODIUM METABISULFITE (UNII: 4VON5FNS3C) 1 mg in 1 mL SODIUM SULFITE (UNII: VTK01UQK3G) 1 mg in 1 mL SODIUM CHLORIDE (UNII: 451W47IQ8X) 6 mg in 1 mL WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55150-318-25 25 in 1 CARTON 07/07/2020 1 NDC:55150-318-01 1 mL in 1 AMPULE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA211816 07/07/2020 CHLORPROMAZINE HYDROCHLORIDE
chlorpromazine hydrochloride injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:55150-319 Route of Administration INTRAMUSCULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CHLORPROMAZINE HYDROCHLORIDE (UNII: 9WP59609J6) (CHLORPROMAZINE - UNII:U42B7VYA4P) CHLORPROMAZINE HYDROCHLORIDE 25 mg in 1 mL Inactive Ingredients Ingredient Name Strength ASCORBIC ACID (UNII: PQ6CK8PD0R) 2 mg in 1 mL SODIUM METABISULFITE (UNII: 4VON5FNS3C) 1 mg in 1 mL SODIUM SULFITE (UNII: VTK01UQK3G) 1 mg in 1 mL SODIUM CHLORIDE (UNII: 451W47IQ8X) 6 mg in 1 mL WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55150-319-25 25 in 1 CARTON 07/07/2020 1 NDC:55150-319-01 2 mL in 1 AMPULE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA211816 07/07/2020 Labeler - Eugia US LLC (968961354) Establishment Name Address ID/FEI Business Operations Eugia Pharma Specialities Limited 650498244 ANALYSIS(55150-318, 55150-319) , MANUFACTURE(55150-318, 55150-319)






![PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 25 mg per mL - 1 mL Container-Carton [25 Ampules]](/dailymed/image.cfm?name=chlorpromazine-fig2.jpg&setid=07e6144a-b460-4887-b8e2-dd371c14a55c)

![PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 50 mg per 2 mL (25 mg / mL) - 2 mL Container-Carton [25 Ampules]](/dailymed/image.cfm?name=chlorpromazine-fig4.jpg&setid=07e6144a-b460-4887-b8e2-dd371c14a55c)