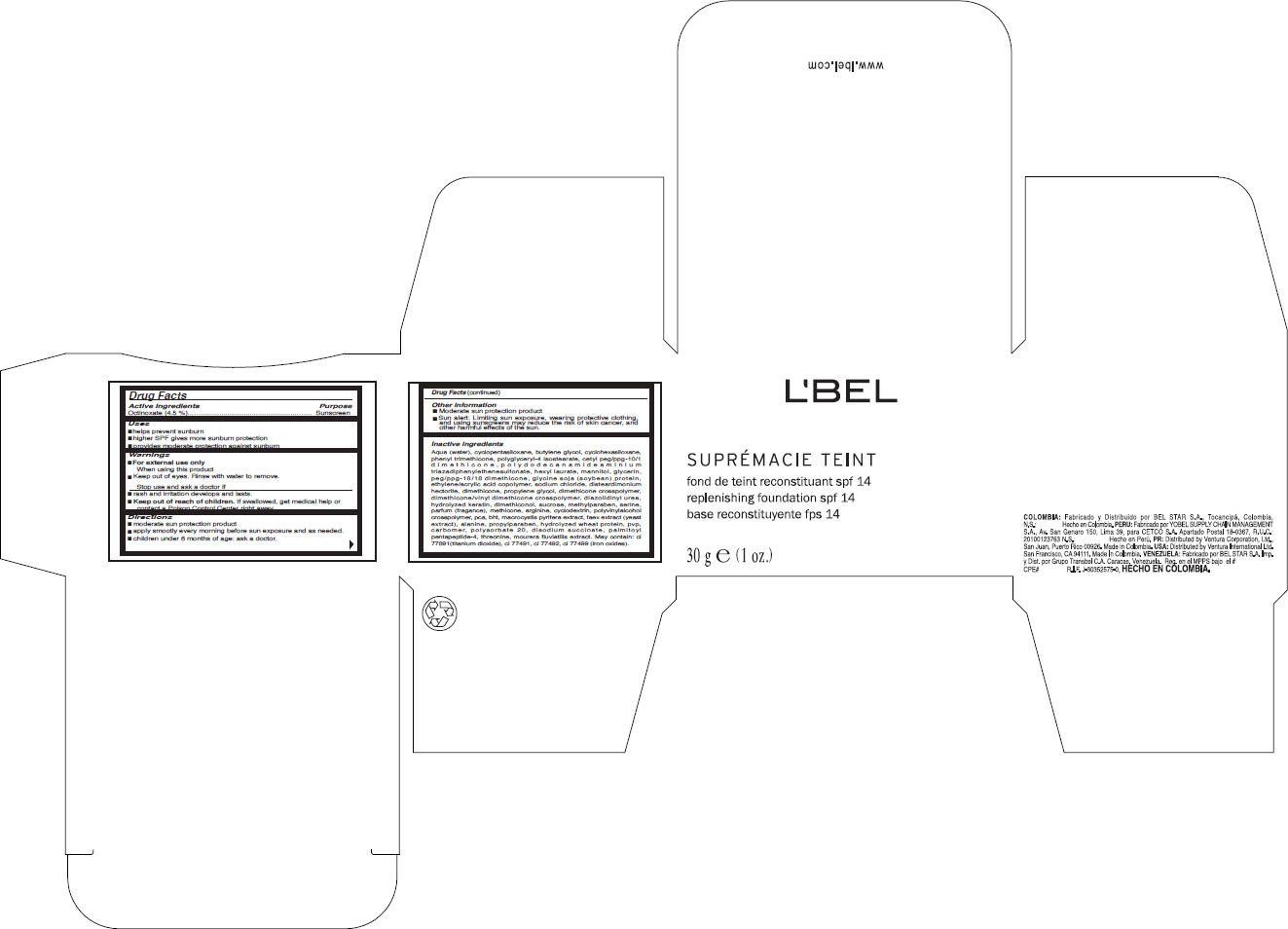
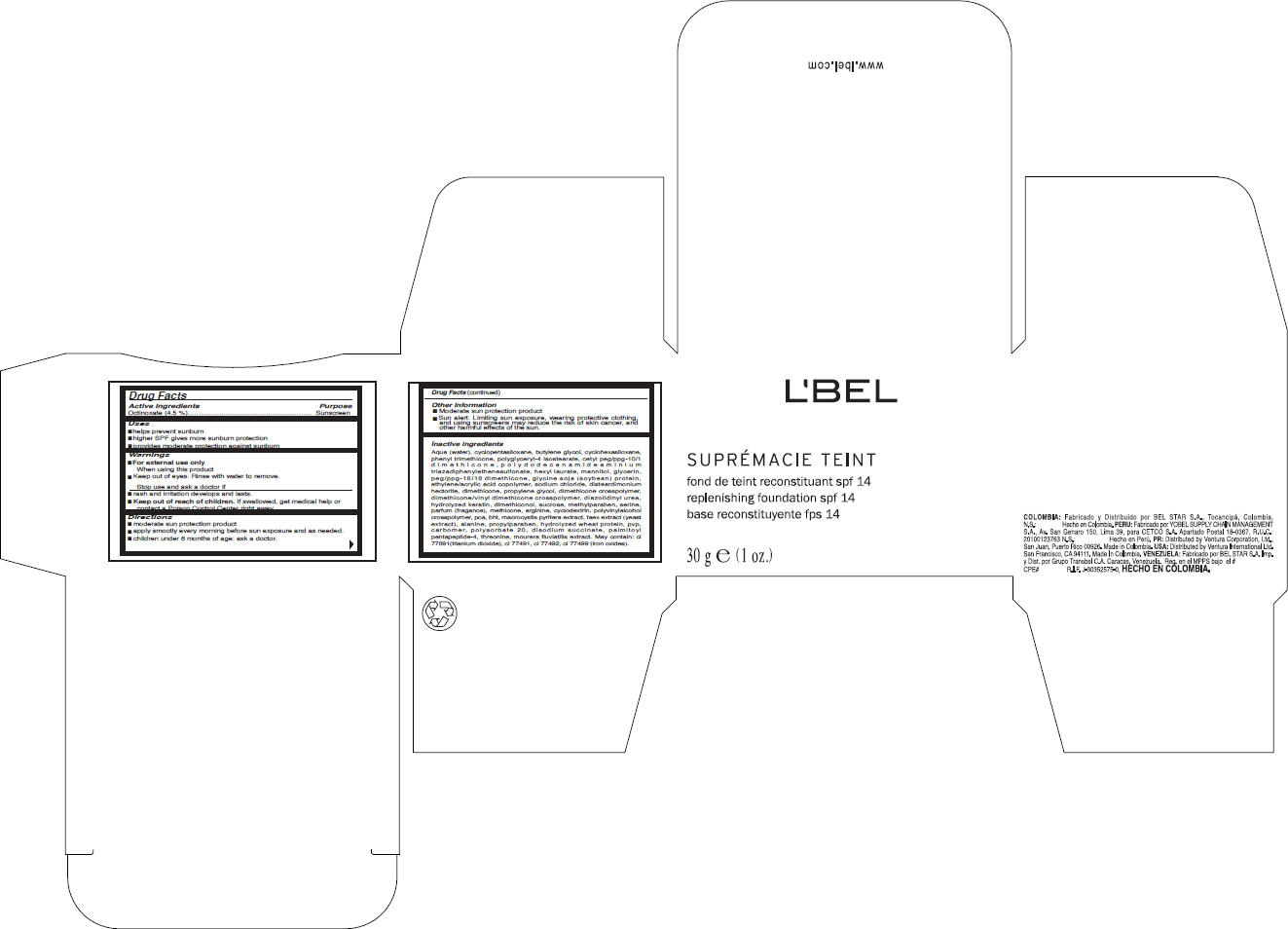
Label: LBEL- octinoxate cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 14783-085-63, 14783-085-64 - Packager: VENTURA INTERNATIONAL LTD.,
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 10, 2010
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredients
- Purpose
- Uses
- Warnings
- Directions
- Other information
-
Inactive ingredients
Aqua (water), cyclopentasiloxane, butylene glycol, cyclohexasiloxane, phenyl trimethicone, polyglyceryl-4 isostearate, cetyl peg/ppg-10/1 dimethicone, polydodecanamideaminium triazadiphenylethenesulfonate, hexyl laurate, mannitol, glycerin, peg/ppg-18/18 dimethicone, glycine soja (soybean) protein, ethylene/acrylic acid copolymer, sodium chloride, disteardimonium hectorite, dimethicone, propylene glycol, dimethicone crosspolymer, dimethicone/vinyl dimethicone crosspolymer, diazolidinyl urea, hydrolyzed keratin, dimethiconol, sucrose, methylparaben, serine, parfum (fragance), methicone, arginine, cyclodextrin, polyvinylalcohol crosspolymer, pca, bht, macrocystis pyrifera extract, faex extract (yeast extract), alanine, propylparaben, hydrolyzed wheat protein, pvp, carbomer, polysorbate 20, disodium succinate, palmitoyl pentapeptide-4, threonine, mourera fluviatilis extract. May contain: ci 77891(titanium dioxide), ci 77491, ci 77492, ci 77499 (iron oxides).
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 30 g Carton
-
INGREDIENTS AND APPEARANCE
LBEL SUPREMACIE TEINT
octinoxate creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:14783-085 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Octinoxate (UNII: 4Y5P7MUD51) (Octinoxate - UNII:4Y5P7MUD51) Octinoxate 1.35 g in 30 g Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) cyclomethicone 5 (UNII: 0THT5PCI0R) butylene glycol (UNII: 3XUS85K0RA) cyclomethicone 6 (UNII: XHK3U310BA) phenyl trimethicone (UNII: DR0K5NOJ4R) polyglyceryl-4 isostearate (UNII: 820DPX33S7) dimethicone (UNII: 92RU3N3Y1O) hexyl laurate (UNII: 4CG9F9W01Q) mannitol (UNII: 3OWL53L36A) glycerin (UNII: PDC6A3C0OX) glycine (UNII: TE7660XO1C) sodium chloride (UNII: 451W47IQ8X) hectorite (UNII: 08X4KI73EZ) propylene glycol (UNII: 6DC9Q167V3) diazolidinyl urea (UNII: H5RIZ3MPW4) sucrose (UNII: C151H8M554) methylparaben (UNII: A2I8C7HI9T) serine (UNII: 452VLY9402) arginine (UNII: 94ZLA3W45F) butylated hydroxytoluene (UNII: 1P9D0Z171K) yeast (UNII: 3NY3SM6B8U) alanine (UNII: OF5P57N2ZX) propylparaben (UNII: Z8IX2SC1OH) wheat (UNII: 4J2I0SN84Y) polysorbate 20 (UNII: 7T1F30V5YH) sodium succinate anhydrous (UNII: V8ZGC8ISR3) threonine (UNII: 2ZD004190S) titanium dioxide (UNII: 15FIX9V2JP) ferric oxide red (UNII: 1K09F3G675) ferric oxide yellow (UNII: EX438O2MRT) ferrosoferric oxide (UNII: XM0M87F357) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:14783-085-63 1 in 1 BOX 1 NDC:14783-085-64 30 g in 1 JAR Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 08/15/2010 Labeler - VENTURA INTERNATIONAL LTD., (603192787)