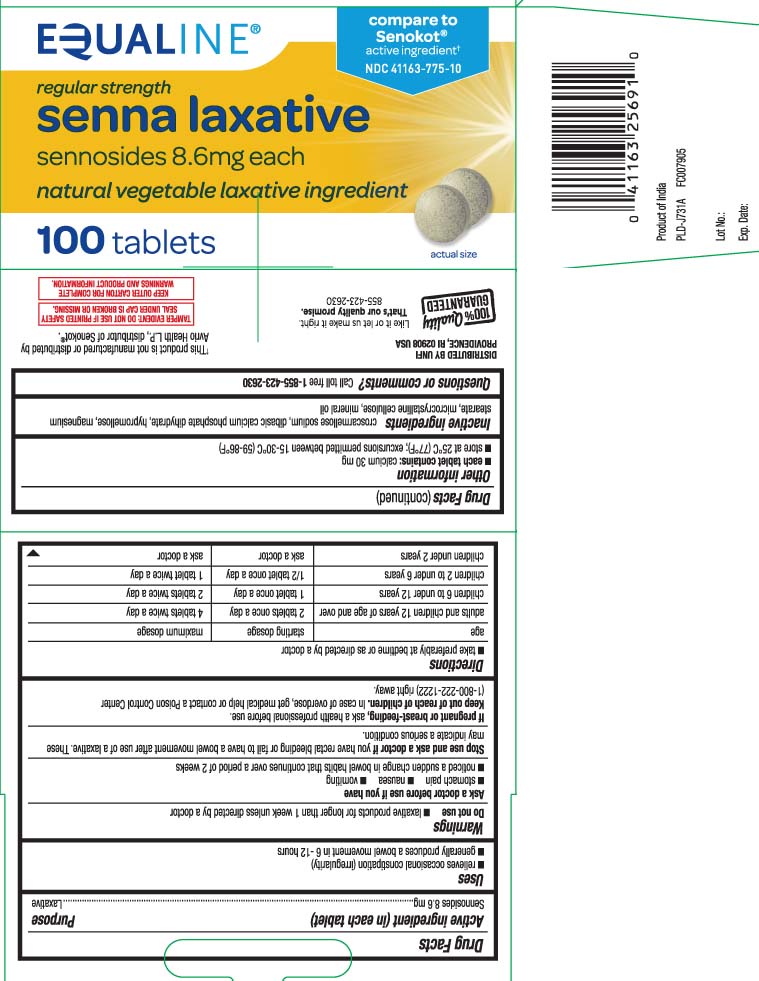
Label: SENNA LAXATIVE- sennosides tablet
- NDC Code(s): 41163-775-10
- Packager: United Natural Foods, Inc. dba UNFI
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated June 12, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each tablet)
- Purpose
- Uses
- Warnings
-
Directions
- take preferably at bedtime or as directed by a doctor
age starting dosage maximum dosage adults and children 12 years of age and over 2 tablets once a day 4 tablets twice a day children 6 to under 12 years 1 tablet once a day 2 tablets twice a day children 2 to under 6 years 1/2 tablet once a day 1 tablet twice a day children under 2 years ask a doctor ask a doctor
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel
compare to Senokot® active ingredient†
regular strength
senna laxative
sennosides 8.6 mg each
natural vegetable laxative ingredient
tablets
†This product is not manufactured or distributed by Avrio Health L.P., owner of the registered trademark Senokot®.
TAMPER EVIDENT: DO NOT USE IF PRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING.
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION
DISTRIBUTED BY UNFI
PROVIDENCE, RI 02908 USA
- Product Labeling
-
INGREDIENTS AND APPEARANCE
SENNA LAXATIVE
sennosides tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:41163-775 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SENNOSIDES (UNII: 3FYP5M0IJX) (SENNOSIDES - UNII:3FYP5M0IJX) SENNOSIDES 8.6 mg Inactive Ingredients Ingredient Name Strength CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) DIBASIC CALCIUM PHOSPHATE DIHYDRATE (UNII: O7TSZ97GEP) HYPROMELLOSES (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) MINERAL OIL (UNII: T5L8T28FGP) Product Characteristics Color brown Score no score Shape ROUND Size 10mm Flavor Imprint Code TCL080 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:41163-775-10 1 in 1 BOX 10/29/2021 1 100 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M007 10/29/2021 Labeler - United Natural Foods, Inc. dba UNFI (943556183)