ALUPENT
-
metaproterenol sulfate aerosol, metered
Physicians Total Care, Inc.
----------
Prescribing Information
Pharmacist:
Tear off “Instruction for Use” and dispense with container in carton.
DESCRIPTION
Alupent® (metaproterenol sulfate USP) Inhalation Aerosol is a bronchodilator administered by oral inhalation. The Alupent Inhalation Aerosol containing 75 mg of metaproterenol sulfate as micronized powder is sufficient medication for 100 inhalations. The Alupent Inhalation Aerosol containing 150 mg of metaproterenol sulfate as micronized powder is sufficient medication for 200 inhalations. Each metered dose delivers through the mouthpiece 0.65 mg of metaproterenol sulfate (each ml contains 15 mg). The inert ingredients are dichlorodifluoromethane, dichlorotetrafluoroethane and trichloromonofluoromethane as propellants, and sorbitan trioleate.
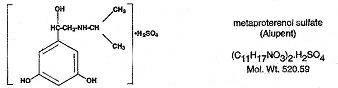
Alupent, 1-(3,5-dihydroxyphenyl)-2-isopropylaminoethanol sulfate, is a white, crystalline, racemic mixture of two optically active isomers. It has the following chemical structure:
CLINICAL PHARMACOLOGY
In vitro studies and in vivo pharmacologic studies have demonstrated that Alupent® (metaproterenol sulfate USP) has a preferential effect on beta-2 adrenergic receptors compared with isoproterenol. While it is recognized that beta-2 adrenergic receptors are the predominant receptors in bronchial smooth muscle, recent data indicate that there is a population of beta-2 receptors in the human heart existing in a concentration between 10-50%. The precise function of these, however, is not yet established (See WARNINGS section).
The pharmacologic effects of beta adrenergic agonist drugs, including Alupent, are at least in part attributable to stimulation through beta adrenergic receptors of intracellular adenyl cyclase, the enzyme which catalyzes the conversion of adenosine triphosphate (ATP) to cyclic-3',5'-adenosine monophosphate (c-AMP). Increased c-AMP levels are associated with relaxation of bronchial smooth muscle and inhibition of release of mediators of immediate hypersensitivity from cells, especially from mast cells.
Pharmacokinetics
Absorption, biotransformation and excretion studies in humans following administration by inhalation have shown that approximately 3 percent of the actuated dose is absorbed intact through the lungs. The major metabolite, metaproterenol-3-0-sulfate, is produced in the gastrointestinal tract. Alupent is not metabolized by catechol-0-methyltransferase nor have glucuronide conjugates been isolated to date.
Pulmonary function tests performed concomitantly usually show improvement following aerosol Alupent administration, e.g. an increase in the one-second forced expiratory volume (FEV1) maximum expiratory flow rate, forced vital capacity, and/or a decrease in airway resistance. The resultant decrease in airway obstruction may relieve the dyspnea associated with bronchospasm.
Controlled single- and multiple-dose studies have been performed with pulmonary function monitoring. The duration of effect of a single dose of two to three inhalations of Alupent (that is, the period of time during which there is a 20 percent or greater increase in FEV1) has varied from 1 to 5 hours.
In repetitive-dosing studies (up to q.i.d.) the duration of effect for a similar dose of Alupent has ranged from about 1 to 2.5 hours. Present studies are inadequate to explain the divergence in duration of the FEV1 effect between single- and repetitive-dosing studies, respectively.
Recent studies in laboratory animals (minipigs, rodents and dogs) recorded the occurrence of cardiac arrhythmias and sudden death (with histologic evidence of myocardial necrosis) when beta agonists and methylxanthines were administered concurrently. The significance of these findings when applied to humans is currently unknown.
INDICATIONS AND USAGE
Alupent® (metaproterenol sulfate USP) is indicated as a bronchodilator for bronchial asthma and for reversible bronchospasm which may occur in association with bronchitis and emphysema.
CONTRAINDICATIONS
Use in patients with cardiac arrhythmias associated with tachycardia is contraindicated.
Although rare, immediate hypersensitivity reactions can occur. Therefore, Alupent® (metaproterenol sulfate USP) Inhalation Aerosol is contraindicated in patients with a history of hypersensitivity to any of its components.
WARNINGS
Fatalities have been reported following excessive use of Alupent® (metaproterenol sulfate USP) as with other sympathomimetic inhalation preparations, and the exact cause is unknown. Cardiac arrest was noted in several cases.
Alupent, like other beta adrenergic agonists, can produce a significant cardiovascular effect in some patients, as measured by pulse rate, blood pressure, symptoms, and/or ECG changes. As with other beta adrenergic aerosols, Alupent can produce paradoxical bronchospasm (which can be life threatening). If it occurs, the preparation should be discontinued immediately and alternative therapy instituted.
Alupent should not be used more often than prescribed. Patients should be advised to contact their physician in the event that they do not respond to their usual dose of a sympathomimetic amine aerosol.
PRECAUTIONS
General
Extreme care must be exercised with respect to the administration of additional sympathomimetic agents.
Since metaproterenol is a sympathomimetic amine, it should be used with caution in patients with cardiovascular disorders, including ischemic heart disease, hypertension or cardiac arrhythmias, in patients with hyperthyroidism or diabetes mellitus, and in patients who are unusually responsive to sympathomimetic amines or who have convulsive disorders. Significant changes in systolic and diastolic blood pressure could be expected to occur in some patients after use of any beta adrenergic bronchodilator.
Information for Patients
Appropriate care should be exercised when considering the administration of additional sympathomimetic agents. A sufficient interval of time should elapse prior to administration of another sympathomimetic agent.
Drug Interactions
Other beta adrenergic aerosol bronchodilators should not be used concomitantly with Alupent® (metaproterenol sulfate USP) because they may have additive effects. Beta adrenergic agonists should be administered with caution to patients being treated with monoamine oxidase inhibitors or tricyclic antidepressants, since the action of beta adrenergic agonists on the vascular system may be potentiated.
Carcinogenesis/Mutagenesis/Impairment of Fertility
In an 18-month study in mice, Alupent produced an increase in benign ovarian tumors in females at doses corresponding to 320 and 640 times the maximum recommended dose (based on a 50 kg individual). In a two-year study in rats, a non-significant incidence of benign leiomyomata of the mesovarium was noted at 640 times the maximum recommended dose. The relevance of the findings to man is not known. Mutagenic studies with Alupent have not been conducted. Reproduction studies in rats revealed no evidence of impaired fertility.
Pregnancy
Teratogenic Effects
PREGNANCY CATEGORY C:
Alupent has been shown to be teratogenic and embryotoxic in rabbits when given in doses corresponding to 640 times the maximum recommended dose. These effects included skeletal abnormalities, hydrocephalus and skull bone separation. Results of other studies in rabbits, rats or mice have not revealed any teratogenic, embryocidal or fetotoxic effects. There are no adequate and well-controlled studies in pregnant women. Alupent should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
ADVERSE REACTIONS
Adverse reactions are similar to those noted with other sympathomimetic agents. The most frequent adverse reaction to Alupent® (metaproterenol sulfate USP) administered by metered-dose inhaler among 251 patients in 90-day controlled clinical trials was nervousness. This was reported in 6.8% of patients. Less frequent adverse experiences, occurring in 1-4% of patients were headache, dizziness, palpitations, gastrointestinal distress, tremor, throat irritation, nausea, vomiting, cough and asthma exacerbation. Tachycardia occurred in less than 1% of patients.
OVERDOSAGE
The expected symptoms with overdosage are those of excessive beta-stimulation and/or any of the symptoms listed under adverse reactions, e.g. angina, hypertension or hypotension, arrhythmias, nervousness, headache, tremor, dry mouth, palpitation, nausea, dizziness, fatigue, malaise and insomnia.
Treatment consists of discontinuation of metaproterenol together with appropriate symptomatic therapy.
DOSAGE AND ADMINISTRATION
The usual single dose is two to three inhalations. With repetitive dosing, inhalation should usually not be repeated more often than about every three to four hours. Total dosage per day should not exceed 12 inhalations.
Alupent® (metaproterenol sulfate USP) Inhalation Aerosol is not recommended for children under 12 years of age.
It is recommended that the physician titrate dosage according to each individual patient’s response to therapy.
HOW SUPPLIED
Each 200 inhalations of Alupent Inhalation Aerosol contains 150 mg of metaproterenol sulfate as a micronized powder in inert propellants. Each metered dose delivers through the mouthpiece 0.65 mg metaproterenol sulfate (each ml contains 15 mg). Alupent Inhalation Aerosol with Mouthpiece (NDC 54868-1043-1), net contents 14g (10ml). The mouthpiece is white with a clear, colorless sleeve and a blue protective cap. Alupent Inhalation Aerosol
Note: The indented statement below is required by the Federal government’s Clean Air Act for all products containing or manufactured with chlorofluorocarbons (CFCs):
A notice similar to the above WARNING has been placed in the information for the patient of this product under the Environmental Protection Agency’s (EPA’s) regulations. The patient’s warning states that the patient should consult his or her physician if there are questions about alternatives.
Store between 59°F (15°C) and 77°F (25°C). Avoid excessive humidity.
Distributed by Boehringer Ingelheim Pharmaceuticals, Inc., Ridgefield, CT 06877
Licensed from: Boehringer Ingelheim International GmbH
Manufactured by 3M Pharmaceuticals, St. Paul, MN 55144-1000
Printed in U.S.A. Revised 2/99
4041090 029
Additional barcode labeling by:
Physicians Total Care, Inc.
Tulsa, Oklahoma 74146
Patient’s Instructions for Use
Alupent®
(metaproterenol sulfate USP)
Inhalation Aerosol
- Insert metal canister into clear end of mouthpiece.
- Remove protective cap, invert canister and shake well before each use.
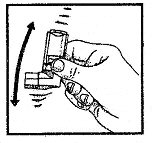
- Avoid spraying in eyes.
- Enclose mouthpiece with the lips. The base of the canister should be held vertically. The Alupent® canister is to be used only with the white Alupent® Inhalation Aerosol mouthpiece. This mouthpiece should not be used with other aerosol medications.
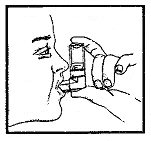
- Exhale deeply, then inhale slowly through the mouth and at the same time firmly press once on the upended canister base; continue to inhale deeply. Hold your breath for a few seconds and then remove the mouthpiece from the mouth and exhale slowly.
- One inhalation is often enough to obtain relief. The inhalation can be repeated once or twice, if necessary, or as your physician directs. Wait at least two minutes before repeating the inhalation. In most cases, the dose should not be repeated more often than every 3 to 4 hours. No more than 12 inhalations should be taken in one day.
- Replace protective cap after use.
WARNING: Do not exceed the dose prescribed by your physician. If difficulty in breathing persists, contact your physician immediately.
Note: When full, the container holds enough medication for at least 200 inhalations: at least 100 inhalations are in the sample unit. Check regularly, by shaking the cylinder or container, to determine whether it contains any medication. When it first seems empty, there are still about ten doses left. Refill containers for the plastic mouthpiece are available when prescribed by your physician.
Keep the mouthpiece clean. Wash with hot water. If soap is used, rinse thoroughly with plain water.
Never open the container holding the medication. Opening it is dangerous and renders the contents useless.
Caution: Contents Under Pressure. Do not puncture or incinerate container. Do not expose to heat or store at temperatures above 120°F. Keep out of reach of small children
Note: The indented statement below is required by the Federal government’s Clean Air Act for all products containing or manufactured with chlorofluorocarbons (CFCs):
- This product contains trichloromonofluoromethane (CFC-11), dichlorodifluoromethane (CFC-12) and dichlorotetrafluoroethane (CFC-114), substances which harm the environment by destroying ozone in the upper atmosphere.
Your physician has determined that this product is likely to help your personal health. USE THIS PRODUCT AS DIRECTED, UNLESS INSTRUCTED TO DO OTHERWISE BY YOUR PHYSICIAN. If you have any questions about alternatives, consult with your physician.
Distributed by
Boehringer Ingelheim Pharmaceuticals, Inc.,
Ridgefield, CT 06877
Licensed from Boehringer
Ingelheim International GmbH
Printed in U.S.A
2/99
| ALUPENT
metaproterenol sulfate aerosol, metered |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| NDA | NDA016402 | 07/31/1973 | 06/30/2007 |
| Labeler - Physicians Total Care, Inc. (194123980) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Physicians Total Care, Inc. | 194123980 | relabel | |
Revised: 10/2012 Physicians Total Care, Inc.