QORE 24 ANTIMICROBIAL HAND PURIFIER
-
benzalkonium chloride spray
Qore Systems LLC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
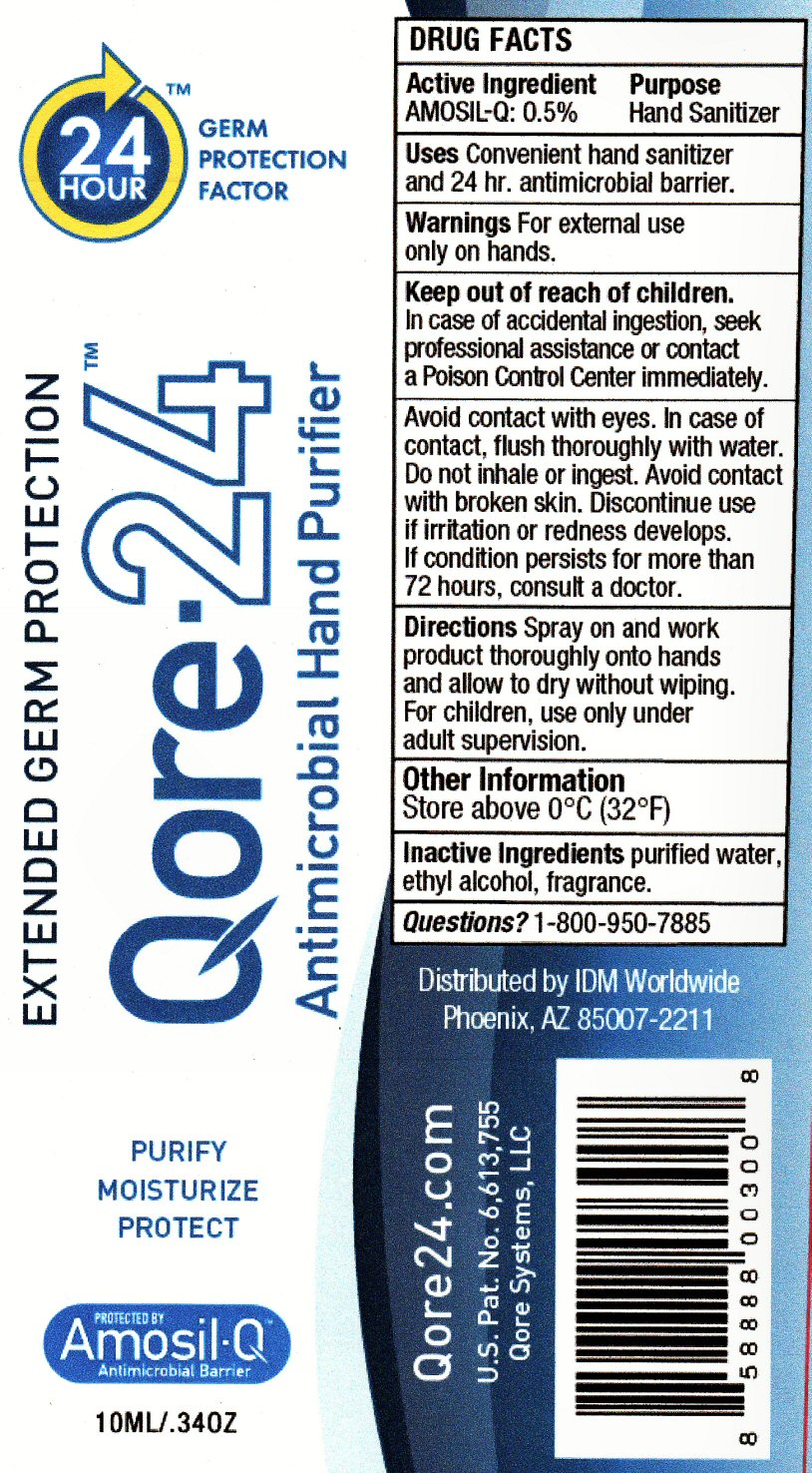
Keep Out of Reach of Children Section
Keep out of reach of children. In case of accidental ingestion, seek professional assistance or contact a Poison Control Center immediately.
Warnings Section
Warnings For external use only on hands. In case of accidental ingestion, seek professional assistance or contact a Poison Control Center immediately.
Avoid contact with eyes. In case of contact, flush thoroughly with water. Do not inhale or ingest. Avoid contact with broken skin. Discontinue use if irritation or redness developes. If condition persists for more than 72 hours, consult a doctor.
Directions Section
Directions Spray on and work product thoroughly onto hands and allow to dry without wiping. For children, use only under adult supervision.
Other Information and Questions Section
Other Information Store above 0 degrees C (32 degrees F)
Questions? 1-800-950-7885
Inactive Ingredients Section
Inactive ingredients
purified water, ethylalcohol, fragrance, (3-(Trimethoxysil)Propyl)OctadecyDimethylAmmonium Chloride 0.4%
Package Label Section Qore-24
24 HOUR tm GERM PROTECTION FACTOR
EXTENDED GERM PROTECTION Qore-24 tm Antimicrobial Hand Purifier PURIFY MOISTURIZE PROTECT
PROTECTED BY Amosil-Q Antimicrobial Barrier 10mL/.34OZ
Qore24.com U.S.Pat. No. 6,613,755
Distributed by IDM Worldwide Phoenix, AX 85007-2211
| QORE 24
ANTIMICROBIAL HAND PURIFIER
benzalkonium chloride spray |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC monograph not final | part333E | 01/01/2012 | |
| Labeler - Qore Systems LLC (063816117) |
| Registrant - Qore Systems LLC (063816117) |
Revised: 02/2012 Qore Systems LLC