VIEKIRA PAK- dasabuvir and ombitasvir and paritaprevir and ritonavir
AbbVie Inc.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use VIEKIRA PAK safely and effectively. See full prescribing information for VIEKIRA PAK.
VIEKIRA PAK (ombitasvir, paritaprevir, and ritonavir tablets; dasabuvir tablets), co-packaged for oral use Initial U.S. Approval: 2014 WARNING: RISK OF HEPATITIS B VIRUS REACTIVATION IN PATIENTS COINFECTED WITH HCV AND HBVSee full prescribing information for complete boxed warning.Hepatitis B virus (HBV) reactivation has been reported, in some cases resulting in fulminant hepatitis, hepatic failure, and death. (5.1)
RECENT MAJOR CHANGES
INDICATIONS AND USAGEVIEKIRA PAK is indicated for the treatment of adult patients with chronic hepatitis C virus (HCV):
VIEKIRA PAK includes ombitasvir, a hepatitis C virus NS5A inhibitor, paritaprevir, a hepatitis C virus NS3/4A protease inhibitor, ritonavir, a CYP3A inhibitor and dasabuvir, a hepatitis C virus non-nucleoside NS5B palm polymerase inhibitor. (1)
DOSAGE AND ADMINISTRATIONTesting Prior to the Initiation of Therapy:
Recommended dosage: Two ombitasvir, paritaprevir, ritonavir 12.5/75/50 mg tablets once daily (in the morning) and one dasabuvir 250 mg tablet twice daily (morning and evening) with a meal without regard to fat or calorie content. (2.1)
Treatment Regimen and Duration by Patient Population
DOSAGE FORMS AND STRENGTHSCONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
ADVERSE REACTIONSIn subjects receiving VIEKIRA PAK with ribavirin, the most commonly reported adverse reactions (greater than 10% of subjects) were fatigue, nausea, pruritus, other skin reactions, insomnia and asthenia. In subjects receiving VIEKIRA PAK without ribavirin, the most commonly reported adverse reactions (greater than or equal to 5% of subjects) were nausea, pruritus and insomnia. (6.1)
DRUG INTERACTIONS
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide. Revised: 12/2019 |
||||||||||||||||||
FULL PRESCRIBING INFORMATION
WARNING: RISK OF HEPATITIS B VIRUS REACTIVATION IN PATIENTS COINFECTED WITH HCV AND HBV
Test all patients for evidence of current or prior hepatitis B virus (HBV) infection before initiating treatment with VIEKIRA PAK. HBV reactivation has been reported in HCV/HBV coinfected patients who were undergoing or had completed treatment with HCV direct acting antivirals and were not receiving HBV antiviral therapy. Some cases have resulted in fulminant hepatitis, hepatic failure, and death. Monitor HCV/HBV coinfected patients for hepatitis flare or HBV reactivation during HCV treatment and post-treatment follow-up. Initiate appropriate patient management for HBV infection as clinically indicated [see Warnings and Precautions (5.1)].
1 INDICATIONS AND USAGE
VIEKIRA PAK is indicated for the treatment of adult patients with chronic hepatitis C virus (HCV) [see Dosage and Administration (2.2) and Clinical Studies (14)]:
- genotype 1b without cirrhosis or with compensated cirrhosis
- genotype 1a without cirrhosis or with compensated cirrhosis for use in combination with ribavirin.
2 DOSAGE AND ADMINISTRATION
2.1 Testing Prior to the Initiation of Therapy
- Test all patients for evidence of current or prior HBV infection by measuring hepatitis B surface antigen (HBsAg) and hepatitis B core antibody (anti-HBc) before initiating HCV treatment with VIEKIRA PAK [see Warnings and Precautions (5.1)].
- Prior to initiation of VIEKIRA PAK, assess for laboratory and clinical evidence of hepatic decompensation [see Warnings and Precautions (5.2 and 5.3)].
2.2 Recommended Dosage in Adults
VIEKIRA PAK is ombitasvir, paritaprevir, ritonavir fixed dose combination tablets copackaged with dasabuvir tablets.
The recommended oral dosage of VIEKIRA PAK is two ombitasvir, paritaprevir, ritonavir tablets once daily (in the morning) and one dasabuvir tablet twice daily (morning and evening). Take VIEKIRA PAK with a meal without regard to fat or calorie content [see Clinical Pharmacology (12.3)].
VIEKIRA PAK is used in combination with ribavirin (RBV) in certain patient populations (see Table 1). When administered with VIEKIRA PAK, the recommended dosage of RBV is based on weight: 1000 mg/day for subjects <75 kg and 1200 mg/day for those ≥75 kg, divided and administered twice-daily with food. For ribavirin dosage modifications, refer to the ribavirin prescribing information.
For patients with HCV/HIV-1 co-infection, follow the dosage recommendations in Table 1. Refer to Drug Interactions (7) for dosage recommendations for concomitant HIV-1 antiviral drugs.
Table 1 shows the recommended VIEKIRA PAK treatment regimen and duration based on patient population.
| Patient Population | Treatment* | Duration |
| Genotype 1a,
without cirrhosis | VIEKIRA PAK + ribavirin | 12 weeks |
| Genotype 1a,
with compensated cirrhosis (Child-Pugh A) | VIEKIRA PAK + ribavirin | 24 weeks** |
| Genotype 1b,
with or without compensated cirrhosis (Child-Pugh A) | VIEKIRA PAK | 12 weeks |
| *Note: Follow the genotype 1a dosing recommendations in patients with an unknown genotype 1 subtype or with mixed genotype 1 infection. **VIEKIRA PAK administered with ribavirin for 12 weeks may be considered for some patients based on prior treatment history [see Clinical Studies (14.3)]. |
||
2.3 Use in Liver Transplant Recipients
In liver transplant recipients with normal hepatic function and mild fibrosis (Metavir fibrosis score 2 or lower), the recommended duration of VIEKIRA PAK with ribavirin is 24 weeks, irrespective of HCV genotype 1 subtype [see Clinical Studies (14.6)]. When VIEKIRA PAK is administered with calcineurin inhibitors in liver transplant recipients, dosage adjustment of calcineurin inhibitors is needed [see Drug Interactions (7)].
3 DOSAGE FORMS AND STRENGTHS
VIEKIRA PAK is ombitasvir, paritaprevir, ritonavir fixed dose combination tablets copackaged with dasabuvir tablets.
- Ombitasvir, paritaprevir, ritonavir 12.5/75/50 mg tablets are pink-colored, film-coated, oblong biconvex shaped, debossed with “AV1” on one side.
- Dasabuvir 250 mg tablets are beige-colored, film-coated, oval-shaped, debossed with “AV2” on one side. Each tablet contains 270.3 mg dasabuvir sodium monohydrate equivalent to 250 mg dasabuvir.
4 CONTRAINDICATIONS
- If VIEKIRA PAK is administered with ribavirin, the contraindications to ribavirin also apply to this combination regimen. Refer to the ribavirin prescribing information for a list of contraindications for ribavirin.
- VIEKIRA PAK is contraindicated:
○ In patients with moderate to severe hepatic impairment (Child-Pugh B and C) due to risk of potential toxicity [see Warnings and Precautions (5.2), Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
○ With drugs that are highly dependent on CYP3A for clearance and for which elevated plasma concentrations are associated with serious and/or life-threatening events [see Drug Interactions (7) and Clinical Pharmacology (12.3)]:
- Alpha1-adrenoreceptor antagonist: alfuzosin HCL
- Anti-anginal: ranolazine
- Antiarrhythmic: dronedarone
- Anti-gout: colchicine in patients with renal and/or hepatic impairment
- Antipsychotic: lurasidone, pimozide
- Ergot derivatives: ergotamine, dihydroergotamine, methylergonovine
- Ethinyl estradiol-containing products such as combined oral contraceptives
- GI Motility Agent: cisapride
- HMG-CoA Reductase Inhibitors: atorvastatin, lovastatin, simvastatin
- Immunosuppressants: everolimus, sirolimus, tacrolimus
- Microsomal triglyceride transfer protein inhibitor: lomitapide
- Non-nucleoside reverse transcriptase inhibitor: efavirenz
- Phosphodiesterase-5(PDE5) inhibitor: sildenafil when dosed as Revatio for the treatment of pulmonary arterial hypertension (PAH)
- Sedatives/hypnotics: triazolam, orally administered midazolam
○ With drugs that are moderate or strong inducers of CYP3A and strong inducers of CYP2C8 and may lead to reduced efficacy of VIEKIRA PAK [see Drug Interactions (7) and Clinical Pharmacology (12.3)]:
- Anticonvulsants: carbamazepine, phenytoin, phenobarbital
- Androgen receptor inhibitor: apalutamide
- Antimycobacterial: rifampin
- Herbal Product: St. John’s Wort (Hypericum perforatum)
○ With drugs that are strong inhibitors of CYP2C8 and may increase dasabuvir plasma concentrations and the risk of QT prolongation [see Drug Interactions (7) and Clinical Pharmacology (12.3)]:
- Antihyperlipidemic agent: gemfibrozil
○ In patients with known hypersensitivity to ritonavir (e.g. toxic epidermal necrolysis (TEN) or Stevens-Johnson syndrome).
5 WARNINGS AND PRECAUTIONS
5.1 Risk of Hepatitis B Virus Reactivation in Patients Coinfected with HCV and HBV
Hepatitis B virus (HBV) reactivation has been reported in HCV/HBV coinfected patients who were undergoing or had completed treatment with HCV direct acting antivirals, and who were not receiving HBV antiviral therapy. Some cases have resulted in fulminant hepatitis, hepatic failure and death. Cases have been reported in patients who are HBsAg positive and also in patients with serologic evidence of resolved HBV infection (i.e., HBsAg negative and anti-HBc positive). HBV reactivation has also been reported in patients receiving certain immunosuppressant or chemotherapeutic agents; the risk of HBV reactivation associated with treatment with HCV direct-acting antivirals may be increased in these patients.
HBV reactivation is characterized as an abrupt increase in HBV replication manifesting as a rapid increase in serum HBV DNA level. In patients with resolved HBV infection reappearance of HBsAg can occur. Reactivation of HBV replication may be accompanied by hepatitis, i.e., increases in aminotransferase levels and, in severe cases, increases in bilirubin levels, liver failure, and death can occur.
Test all patients for evidence of current or prior HBV infection by measuring HBsAg and anti-HBc before initiating HCV treatment with VIEKIRA PAK. In patients with serologic evidence of HBV infection, monitor for clinical and laboratory signs of hepatitis flare or HBV reactivation during HCV treatment with VIEKIRA PAK and during post-treatment follow-up. Initiate appropriate patient management for HBV infection as clinically indicated.
5.2 Risk of Hepatic Decompensation and Hepatic Failure in Patients with Cirrhosis
Hepatic decompensation and hepatic failure, including liver transplantation or fatal outcomes, have been reported postmarketing in patients treated with VIEKIRA PAK. Most patients with these severe outcomes had evidence of advanced cirrhosis prior to initiating therapy with VIEKIRA PAK. Reported cases typically occurred within one to four weeks of initiating therapy and were characterized by the acute onset of rising direct serum bilirubin levels without ALT elevations in association with clinical signs and symptoms of hepatic decompensation. Because these events are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
VIEKIRA PAK is contraindicated in patients with moderate to severe hepatic impairment (Child-Pugh B and C) [see Contraindications (4), Adverse Reactions (6.2), Use in Specific Populations (8.6), and Clinical Pharmacology (12.3)].
For patients with cirrhosis:
- Monitor for clinical signs and symptoms of hepatic decompensation (such as ascites, hepatic encephalopathy, variceal hemorrhage).
- Hepatic laboratory testing including direct bilirubin levels should be performed at baseline and during the first 4 weeks of starting treatment and as clinically indicated.
- Discontinue VIEKIRA PAK in patients who develop evidence of hepatic decompensation.
5.3 Increased Risk of ALT Elevations
During clinical trials with VIEKIRA PAK with or without ribavirin, elevations of ALT to greater than 5 times the upper limit of normal (ULN) occurred in approximately 1% of all subjects [see Adverse Reactions (6.1)]. ALT elevations were typically asymptomatic, occurred during the first 4 weeks of treatment, and declined within two to eight weeks of onset with continued dosing of VIEKIRA PAK with or without ribavirin.
These ALT elevations were significantly more frequent in female subjects who were using ethinyl estradiol-containing medications such as combined oral contraceptives, contraceptive patches or contraceptive vaginal rings. Ethinyl estradiol-containing medications must be discontinued prior to starting therapy with VIEKIRA PAK [see Contraindications (4)]. Alternative methods of contraception (e.g, progestin only contraception or non-hormonal methods) are recommended during VIEKIRA PAK therapy. Ethinyl estradiol-containing medications can be restarted approximately 2 weeks following completion of treatment with VIEKIRA PAK.
Women using estrogens other than ethinyl estradiol, such as estradiol and conjugated estrogens used in hormone replacement therapy had a rate of ALT elevation similar to those not receiving any estrogens; however, due to the limited number of subjects taking these other estrogens, caution is warranted for co-administration with VIEKIRA PAK [see Adverse Reactions (6.1)].
Hepatic laboratory testing should be performed during the first 4 weeks of starting treatment and as clinically indicated thereafter. If ALT is found to be elevated above baseline levels, it should be repeated and monitored closely:
- Patients should be instructed to consult their health care professional without delay if they have onset of fatigue, weakness, lack of appetite, nausea and vomiting, jaundice or discolored feces.
- Consider discontinuing VIEKIRA PAK if ALT levels remain persistently greater than 10 times the ULN.
- Discontinue VIEKIRA PAK if ALT elevation is accompanied by signs or symptoms of liver inflammation or increasing direct bilirubin, alkaline phosphatase, or INR.
5.4 Risks Associated With Ribavirin Combination Treatment
If VIEKIRA PAK is administered with ribavirin, the warnings and precautions for ribavirin, in particular the pregnancy avoidance warning, apply to this combination regimen. Refer to the ribavirin prescribing information for a full list of the warnings and precautions for ribavirin.
5.5 Risk of Adverse Reactions or Reduced Therapeutic Effect Due to Drug Interactions
The concomitant use of VIEKIRA PAK and certain other drugs may result in known or potentially significant drug interactions, some of which may lead to:
- Loss of therapeutic effect of VIEKIRA PAK and possible development of resistance
- Possible clinically significant adverse reactions from greater exposures of concomitant drugs or components of VIEKIRA PAK.
See Table 4 for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations [see Drug Interactions (7)]. Consider the potential for drug interactions prior to and during VIEKIRA PAK therapy; review concomitant medications during VIEKIRA PAK therapy; and monitor for the adverse reactions associated with the concomitant drugs [see Contraindications (4) and Drug Interactions (7)].
5.6 Risk of HIV-1 Protease Inhibitor Drug Resistance in HCV/HIV-1 Co-infected Patients
The ritonavir component of VIEKIRA PAK is also an HIV-1 protease inhibitor and can select for HIV-1 protease inhibitor resistance-associated substitutions. Any HCV/HIV-1 co-infected patients treated with VIEKIRA PAK should also be on a suppressive antiretroviral drug regimen to reduce the risk of HIV-1 protease inhibitor drug resistance.
6 ADVERSE REACTIONS
If VIEKIRA PAK is administered with ribavirin (RBV), refer to the prescribing information for ribavirin for a list of ribavirin-associated adverse reactions.
The following adverse reaction is described below and elsewhere in the labeling:
- Risk of Hepatic Decompensation and Hepatic Failure in Patients with Cirrhosis [see Warnings and Precautions (5.2)]
- Increased Risk of ALT Elevations [see Warnings and Precautions (5.3)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of VIEKIRA PAK cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety assessment was based on data from seven clinical trials in more than 2,000 subjects who received VIEKIRA PAK with or without ribavirin for 12 or 24 weeks.
VIEKIRA PAK with Ribavirin in Placebo-Controlled Trials
The safety of VIEKIRA PAK in combination with ribavirin was assessed in 770 subjects with chronic HCV genotype 1 (GT1) infection in two placebo-controlled trials (SAPPHIRE-I and -II) [see Clinical Studies (14.1, 14.2)]. Adverse reactions that occurred more often in subjects treated with VIEKIRA PAK in combination with ribavirin compared to placebo were fatigue, nausea, pruritus, other skin reactions, insomnia, and asthenia (see Table 2). The majority of the adverse reactions were mild in severity. Two percent of subjects experienced a serious adverse event (SAE). The proportion of subjects who permanently discontinued treatment due to adverse reactions was less than 1%.
| SAPPHIRE-I and -II | ||
| VIEKIRA PAK + RBV
12 Weeks N = 770 % | Placebo
12 Weeks N = 255 % |
|
| Fatigue | 34 | 26 |
| Nausea | 22 | 15 |
| Pruritus* | 18 | 7 |
| Skin reactions$ | 16 | 9 |
| Insomnia | 14 | 8 |
| Asthenia | 14 | 7 |
| *Grouped term ‘pruritus’ included the preferred terms pruritus and pruritus generalized. $Grouped terms: rash, erythema, eczema, rash maculo-papular, rash macular, dermatitis, rash papular, skin exfoliation, rash pruritic, rash erythematous, rash generalized, dermatitis allergic, dermatitis contact, exfoliative rash, photosensitivity reaction, psoriasis, skin reaction, ulcer, urticaria. |
||
VIEKIRA PAK with and without Ribavirin in Regimen-Controlled Trials
VIEKIRA PAK with and without ribavirin was assessed in 401 and 509 subjects with chronic HCV infection, respectively, in three clinical trials (PEARL-II, PEARL-III and PEARL-IV) [see Clinical Studies (14.1, 14.2)]. Pruritus, nausea, insomnia, and asthenia were identified as adverse events occurring more often in subjects treated with VIEKIRA PAK in combination with ribavirin (see Table 3). The majority of adverse events were mild to moderate in severity. The proportion of subjects who permanently discontinued treatment due to adverse events was less than 1% for both VIEKIRA PAK in combination with ribavirin and VIEKIRA PAK alone.
| PEARL-II, -III and -IV | ||
| VIEKIRA PAK + RBV
12 Weeks N = 401 % | VIEKIRA PAK
12 Weeks N = 509 % |
|
| Nausea | 16 | 8 |
| Pruritus* | 13 | 7 |
| Insomnia | 12 | 5 |
| Asthenia | 9 | 4 |
| *Grouped term ‘pruritus’ included the preferred terms pruritus and pruritus generalized. | ||
VIEKIRA PAK with Ribavirin in GT1-infected Subjects with Compensated Cirrhosis
VIEKIRA PAK with ribavirin was assessed in 380 subjects with genotype 1 infection and compensated cirrhosis who were treated with VIEKIRA PAK plus ribavirin for 12 (n=208) or 24 (n=172) weeks duration (TURQUOISE-II) [see Clinical Studies (14.1, 14.3)]. The type and severity of adverse events in subjects with compensated cirrhosis was comparable to non-cirrhotic subjects in other phase 3 trials. Fatigue, skin reactions and dyspnea occurred at least 5% more often in subjects treated for 24 weeks. The majority of adverse events occurred during the first 12 weeks of dosing in both treatment arms. Most of the adverse events were mild to moderate in severity. The proportion of subjects treated with VIEKIRA PAK for 12 and 24 weeks with SAEs was 6% and 5%, respectively and 2% of subjects permanently discontinued treatment due to adverse events in each treatment arm.
VIEKIRA PAK without Ribavirin in GT1b-infected Subjects with Compensated Cirrhosis
VIEKIRA PAK without ribavirin for 12 weeks was assessed in 60 subjects with genotype 1b infection and compensated cirrhosis (TURQUOISE-III) [see Clinical Studies (14.1, 14.3)]. The type and severity of adverse events and laboratory abnormalities in genotype 1b-infected subjects with compensated cirrhosis were comparable to subjects in other trials without ribavirin.
Skin Reactions
In PEARL-II, -III and -IV, 7% of subjects receiving VIEKIRA PAK alone and 10% of subjects receiving VIEKIRA PAK with ribavirin reported rash-related events. In SAPPHIRE-I and -II 16% of subjects receiving VIEKIRA PAK with ribavirin and 9% of subjects receiving placebo reported skin reactions. In TURQUOISE-II, 18% and 24% of subjects receiving VIEKIRA PAK with ribavirin for 12 or 24 weeks reported skin reactions. The majority of events were graded as mild in severity.
Laboratory Abnormalities
Serum ALT Elevations
Approximately 1% of subjects treated with VIEKIRA PAK experienced post-baseline serum ALT levels greater than 5 times the upper limit of normal (ULN) after starting treatment. The incidence increased to 25% (4/16) among women taking a concomitant ethinyl estradiol containing medication [see Contraindications (4) and Warnings and Precautions (5.3)]. The incidence of clinically relevant ALT elevations among women using estrogens other than ethinyl estradiol, such as estradiol and conjugated estrogens used in hormone replacement therapy was 3% (2/59).
ALT elevations were typically asymptomatic, generally occurred during the first 4 weeks of treatment (mean time 20 days, range 8-57 days) and most resolved with ongoing therapy. The majority of these ALT elevations were assessed as drug-related liver injury. Elevations in ALT were generally not associated with bilirubin elevations. Cirrhosis was not a risk factor for elevated ALT [see Warnings and Precautions (5.3)].
Serum Bilirubin Elevations
Post-baseline elevations in bilirubin at least 2 x ULN were observed in 15% of subjects receiving VIEKIRA PAK with ribavirin compared to 2% in those receiving VIEKIRA PAK alone. These bilirubin increases were predominately indirect and related to the inhibition of the bilirubin transporters OATP1B1/1B3 by paritaprevir and ribavirin-induced hemolysis. Bilirubin elevations occurred after initiation of treatment, peaked by study Week 1, and generally resolved with ongoing therapy. Bilirubin elevations were not associated with serum ALT elevations.
Anemia/Decreased Hemoglobin
Across all Phase 3 studies, the mean change from baseline in hemoglobin levels in subjects treated with VIEKIRA PAK in combination with ribavirin was -2.4 g/dL and the mean change in subjects treated with VIEKIRA PAK alone was -0.5 g/dL. Decreases in hemoglobin levels occurred early in treatment (Week 1-2) with further reductions through Week 3. Hemoglobin values remained low during the remainder of treatment and returned towards baseline levels by post-treatment Week 4. Less than 1% of subjects treated with VIEKIRA PAK with ribavirin had hemoglobin levels decrease to less than 8.0 g/dL during treatment. Seven percent of subjects treated with VIEKIRA PAK in combination with ribavirin underwent a ribavirin dose reduction due to a decrease in hemoglobin levels; three subjects received a blood transfusion and five required erythropoietin. One patient discontinued therapy due to anemia. No subjects treated with VIEKIRA PAK alone had a hemoglobin level less than 10 g/dL.
VIEKIRA PAK in HCV/HIV-1 Co-infected Subjects
VIEKIRA PAK with ribavirin was assessed in 63 subjects with HCV/HIV-1 co-infection who were on stable antiretroviral therapy. The most common adverse events occurring in at least 10% of subjects were fatigue (48%), insomnia (19%), nausea (17%), headache (16%), pruritus (13%), cough (11%), irritability (10%), and ocular icterus (10%).
Elevations in total bilirubin greater than 2 x ULN (mostly indirect) occurred in 34 (54%) subjects. Fifteen of these subjects were also receiving atazanavir at the time of bilirubin elevation and nine also had adverse events of ocular icterus, jaundice or hyperbilirubinemia. None of the subjects with hyperbilirubinemia had concomitant elevations of aminotransferases [see Warnings and Precautions (5.6), Adverse Reactions (6.1) and Clinical Studies (14.6)]. No subject experienced a grade 3 ALT elevation.
Seven subjects (11%) had at least one post-baseline hemoglobin value of less than 10 g/dL, and six of these subjects had a ribavirin dose modification; no subject in this small cohort required a blood transfusion or erythropoietin.
Median declines in CD4+ T-cell counts of 47 cells/mm3 and 62 cells/mm3 were observed at the end of 12 and 24 weeks of treatment, respectively, and most returned to baseline levels post-treatment. Two subjects had CD4+ T-cell counts decrease to less than 200 cells/mm3 during treatment without a decrease in CD4%. No subject experienced an AIDS-related opportunistic infection.
VIEKIRA PAK in Selected Liver Transplant Recipients
VIEKIRA PAK with ribavirin was assessed in 34 post-liver transplant subjects with recurrent HCV infection. Adverse events occurring in more than 20% of subjects included fatigue 50%, headache 44%, cough 32%, diarrhea 26%, insomnia 26%, asthenia 24%, nausea 24%, muscle spasms 21% and rash 21%. Ten subjects (29%) had at least one post-baseline hemoglobin value of less than 10 g/dL. Ten subjects underwent a ribavirin dose modification due to decrease in hemoglobin and 3% (1/34) had an interruption of ribavirin. Five subjects received erythropoietin, all of whom initiated ribavirin at the starting dose of 1000 to 1200 mg daily. No subject received a blood transfusion [see Clinical Studies (14.5)].
6.2 Post-Marketing Adverse Reactions
The following adverse reactions have been identified during post approval use of VIEKIRA PAK. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune System Disorders: Anaphylactic reactions and other hypersensitivity reactions (including angioedema).
Hepatobiliary Disorders: Hepatic decompensation, hepatic failure [see Warnings and Precautions (5.2)].
Skin and Subcutaneous Tissue Disorders: Erythema multiforme (EM).
7 DRUG INTERACTIONS
7.1 Potential for VIEKIRA PAK to Affect Other Drugs
Ombitasvir, paritaprevir, and dasabuvir are inhibitors of UGT1A1, and ritonavir is an inhibitor of CYP3A4. Paritaprevir is an inhibitor of OATP1B1 and OATP1B3 and paritaprevir, ritonavir and dasabuvir are inhibitors of BCRP. Co-administration of VIEKIRA PAK with drugs that are substrates of CYP3A, UGT1A1, BCRP, OATP1B1 or OATP1B3 may result in increased plasma concentrations of such drugs [see Contraindications (4), Warnings and Precautions (5.5), and Clinical Pharmacology (12.3)].
7.2 Potential for Other Drugs to Affect One or More Components of VIEKIRA PAK
Paritaprevir and ritonavir are primarily metabolized by CYP3A enzymes. Co-administration of VIEKIRA PAK with strong inhibitors of CYP3A may increase paritaprevir and ritonavir concentrations. Dasabuvir is primarily metabolized by CYP2C8 enzymes. Co-administration of VIEKIRA PAK with drugs that inhibit CYP2C8 may increase dasabuvir plasma concentrations. Ombitasvir is primarily metabolized via amide hydrolysis while CYP enzymes play a minor role in its metabolism. Ombitasvir, paritaprevir, dasabuvir and ritonavir are substrates of P-gp. Ombitasvir, paritaprevir and dasabuvir are substrates of BCRP. Paritaprevir is a substrate of OATP1B1 and OATP1B3. Inhibition of P-gp, BCRP, OATP1B1 or OATP1B3 may increase the plasma concentrations of the various components of VIEKIRA PAK.
7.3 Established and Other Potential Drug Interactions
Clearance of HCV infection with direct-acting antivirals may lead to changes in hepatic function, which may impact the safe and effective use of concomitant medications. For example, altered blood glucose control resulting in serious symptomatic hypoglycemia has been reported in diabetic patients in postmarketing case reports and published epidemiological studies. Management of hypoglycemia in these cases required either discontinuation or dose modification of concomitant medications used for diabetes treatment.
Frequent monitoring of relevant laboratory parameters (e.g. International Normalized Ratio [INR] in patients taking warfarin, blood glucose levels in diabetic patients) or drug concentrations of concomitant medications such as CYP P450 substrates with a narrow therapeutic index (e.g. certain immunosuppressants) is recommended to ensure safe and effective use. Dose adjustments of concomitant medications may be necessary.
If dose adjustments of concomitant medications are made due to treatment with VIEKIRA PAK, doses should be re-adjusted after administration of VIEKIRA PAK is completed. Dose adjustment is not required for VIEKIRA PAK.
Table 4 provides the effect of co-administration of VIEKIRA PAK on concentrations of concomitant drugs and the effect of concomitant drugs on the various components of VIEKIRA PAK. See Contraindications (4) for drugs that are contraindicated with VIEKIRA PAK. Refer to the ritonavir prescribing information for other potentially significant drug interactions with ritonavir.
| Concomitant Drug Class: Drug Name | Effect on Concentration | Clinical Comments |
| ALPHA1-ADRENORECEPTOR ANTAGONIST | ||
| alfuzosin HCl* | ↑ alfuzosin HCl | Contraindicated due to potential for hypotension [see Contraindications (4)]. |
| ANDROGEN RECEPTOR INHIBITOR | ||
| apalutamide* | ↓ ombitasvir ↓ paritaprevir ↓ ritonavir | Contraindicated due to potential loss of therapeutic activity of VIEKIRA PAK [see Contraindications (4)]. |
| ANGIOTENSIN RECEPTOR BLOCKERS e.g. | ||
| valsartan*
losartan* candesartan* | ↑ angiotensin receptor blockers | Decrease the dose of the angiotensin receptor blockers and monitor patients for signs and symptoms of hypotension and/or worsening renal function. If such events occur, consider further dose reduction of the angiotensin receptor blocker or switching to an alternative to the angiotensin receptor blocker. |
| ANTI-ANGINAL | ||
| ranolazine* | ↑ ranolazine | Contraindicated due to potential for serious and/or life-threatening reactions [see Contraindications (4)]. |
| ANTIARRHYTHMICS | ||
| dronedarone* | ↑ dronedarone | Contraindicated due to potential for serious and/or life-threatening reactions such as cardiac arrhythmias [see Contraindications (4)]. |
| amiodarone*, bepridil*, disopyramide*,flecainide*, lidocaine (systemic)*, mexiletine*, propafenone*, quinidine* | ↑ antiarrhythmics | For contraindicated antiarrhythmics [see Contraindications (4)]. Therapeutic concentration monitoring (if available) is recommended for antiarrhythmics when co-administered with VIEKIRA PAK. |
| ANTICANCER AGENTS/KINASE INHIBITORS | ||
| encorafenib* fostamatinib* ibrutinib* ivosidenib* | ↑ anticancer agents/ kinase inhibitors | Co-administration of VIEKIRA PAK with these anticancer agents/kinase inhibitors may result in increased risk for adverse events. Refer to the prescribing information of these agents for details on co-administration with strong CYP3A inhibitors. |
| ANTICONVULSANTS | ||
| carbamazepine*, phenytoin*, phenobarbital* | ↓ ombitasvir ↓ paritaprevir ↓ ritonavir | Contraindicated due to potential loss of therapeutic activity of VIEKIRA PAK [see Contraindications (4)]. |
| ANTIDIABETIC DRUGS | ||
| metformin | ↔ metformin | Monitor for signs of onset of lactic acidosis such as respiratory distress, somnolence, and non-specific abdominal distress or worsening renal function. Concomitant metformin use in patients with renal insufficiency or hepatic impairment is not recommended. Refer to the prescribing information of metformin for further guidance. |
| ANTI-GOUT | ||
| colchicine* | ↑ colchicine | Contraindicated due to potential for serious and/or life-threatening reactions in patients with renal and/or hepatic impairment [see Contraindications (4)]. |
| ANTIFUNGALS | ||
| ketoconazole | ↑ ketoconazole | When VIEKIRA PAK is co-administered with ketoconazole, the maximum daily dose of ketoconazole should be limited to 200 mg per day. |
| voriconazole* | ↓ voriconazole | Co-administration of VIEKIRA PAK with voriconazole is not recommended unless an assessment of the benefit-to-risk ratio justifies the use of voriconazole. |
| ANTIHYPERLIPIDEMIC AGENT | ||
| gemfibrozil | ↑ dasabuvir ↑ paritaprevir | Contraindicated due to increase in dasabuvir exposures by 10-fold which may increase the risk of QT prolongation [see Contraindications (4)]. |
| ANTIMYCOBACTERIAL | ||
| rifampin* | ↓ ombitasvir | Contraindicated due to potential loss of therapeutic activity of VIEKIRA PAK [see Contraindications (4)]. |
| ANTIPSYCHOTICS | ||
| lurasidone* | ↑ lurasidone | Contraindicated due to potential for serious and/or life-threatening reactions [see Contraindications (4)]. |
| pimozide* | ↑ pimozide | Contraindicated due to potential for serious and/or life-threatening reactions such as cardiac arrhythmias [see Contraindications (4)]. |
| quetiapine* | ↑ quetiapine | For contraindicated antipsychotics [see Contraindications (4)].
|
| CALCIUM CHANNEL BLOCKERS | ||
| amlodipine nifedipine* diltiazem* verapamil* | ↑ calcium channel blockers | Decrease the dose of the calcium channel blocker. The dose of amlodipine should be decreased by at least 50%. Clinical monitoring of patients is recommended for edema and/or signs and symptoms of hypotension. If such events occur, consider further dose reduction of the calcium channel blocker or switching to an alternative to the calcium channel blocker. |
| CORTICOSTEROIDS (INHALED/NASAL) | ||
| fluticasone* | ↑ fluticasone | Concomitant use of VIEKIRA PAK with inhaled or nasal fluticasone may reduce serum cortisol concentrations. Alternative corticosteroids should be considered, particularly for long term use. |
| DIURETICS | ||
| furosemide | ↑ furosemide (Cmax) | Clinical monitoring of patients is recommended and therapy should be individualized based on patient’s response. |
| ERGOT DERIVATIVES | ||
| ergotamine*, dihydroergotamine*, methylergonovine* | ↑ ergot derivatives | Contraindicated due to potential for acute ergot toxicity characterized by vasospasm. Tissue ischemia has been associated with co-administration of ritonavir and ergonovine, ergotamine, dihydroergotamine, or methylergonovine [see Contraindications (4)]. |
| ETHINYL ESTRADIOL-CONTAINING PRODUCTS | ||
| ethinyl estradiol-containing medications such as combined oral contraceptives | ↔ ethinyl estradiol | Contraindicated due to potential for ALT elevations [see Contraindications (4) and Warnings and Precautions (5.3)]. |
| GI MOTILITY AGENT | ||
| cisapride* | ↑ cisapride | Contraindicated due to potential for serious and/or life threatening reactions such as cardiac arrhythmias [see Contraindications (4)]. |
| GnRH RECEPTOR ANTAGONISTS | ||
| elagolix* | ↑ elagolix | Co-administration of VIEKIRA PAK with elagolix 200 mg twice daily for more than 1 month is not recommended. |
| HERBAL PRODUCT | ||
| St. John’s Wort* (Hypericum perforatum) | ↓ ombitasvir ↓ paritaprevir ↓ ritonavir | Contraindicated due to potential loss of therapeutic activity of VIEKIRA PAK [see Contraindications (4)]. |
| HIV-ANTIVIRAL AGENTS | ||
| efavirenz* | ↑ efavirenz ↓ paritaprevir ↓ ritonavir | Contraindicated as co-administration of efavirenz based regimens with paritaprevir and ritonavir was poorly tolerated and resulted in liver enzyme elevations [see Contraindications (4)]. |
| atazanavir/ritonavir once daily | ↑ paritaprevir | When coadministered with VIEKIRA PAK, atazanavir 300 mg (without ritonavir) should only be given in the morning. |
| darunavir/ritonavir | ↓ darunavir (Ctrough) | Treatment naïve patients or treatment experienced patients with no darunavir associated substitutions: Darunavir 800 mg once daily (without ritonavir) can be co-administered with VIEKIRA PAK. Treatment experienced patients with at least one darunavir resistance associated substitution or with no baseline resistance information: Co-administration of darunavir/ritonavir 600/100 mg twice daily with VIEKIRA PAK is not recommended. |
| lopinavir/ritonavir | ↑ paritaprevir | Co-administration of VIEKIRA PAK with lopinavir/ritonavir is not recommended. |
| rilpivirine | ↑ rilpivirine | For contraindicated non-nucleoside reverse transcriptase inhibitors [see Contraindications (4)]. Co-administration of VIEKIRA PAK with rilpivirine once daily is not recommended due to potential for QT interval prolongation with higher concentrations of rilpivirine. |
| HMG CoA REDUCTASE INHIBITORS | ||
| atorvastatin lovastatin, simvastatin | ↑ atorvastatin ↑ lovastatin, ↑ simvastatin | Contraindicated due to potential for myopathy including rhabdomyolysis [see Contraindications (4)]. |
| rosuvastatin | ↑ rosuvastatin | For contraindicated HMG-CoA Reductase Inhibitors [see Contraindications (4)]. When VIEKIRA PAK is co-administered with rosuvastatin, the dose of rosuvastatin should not exceed 10 mg per day. |
| pravastatin | ↑ pravastatin | When VIEKIRA PAK is co-administered with pravastatin, the dose of pravastatin should not exceed 40 mg per day. |
| IMMUNOSUPPRESSANTS | ||
| everolimus sirolimus tacrolimus | ↑ everolimus ↑ sirolimus ↑ tacrolimus | Contraindicated due to potential for serious and/or life threatening immunosuppressant-associated adverse events [see Contraindications (4)]. |
| cyclosporine | ↑ cyclosporine | For contraindicated immunosuppressants [see Contraindications (4)]. When initiating therapy with VIEKIRA PAK, reduce cyclosporine dose to 1/5th of the patient’s current cyclosporine dose. Measure cyclosporine blood concentrations to determine subsequent dose modifications. Upon completion of VIEKIRA PAK therapy, the appropriate time to resume pre-VIEKIRA PAK dose of cyclosporine should be guided by assessment of cyclosporine blood concentrations. Frequent assessment of renal function and cyclosporine-related side effects is recommended. |
| LONG ACTING BETA-ADRENOCEPTOR AGONIST | ||
| salmeterol* | ↑ salmeterol | Concurrent administration of VIEKIRA PAK and salmeterol is not recommended. The combination may result in increased risk of cardiovascular adverse events associated with salmeterol, including QT prolongation, palpitations and sinus tachycardia. |
| MUSCLE RELAXANTS | ||
| carisoprodol | ↓ carisoprodol ↔ mepobramate (metabolite of carisoprodol) | Increase dose if clinically indicated. |
| cyclobenzaprine | ↓cyclobenzaprine ↓norcyclobenzaprine (metabolite of cyclobenzaprine) | Increase dose if clinically indicated. |
| MICROSOMAL TRIGLYCERIDE TRANSFER PROTEIN INHIBITOR | ||
| lomitapide* | ↑ lomitapide | Contraindicated due to potential for serious adverse events including hepatotoxicity [see Contraindications (4)]. |
| NARCOTIC ANALGESICS | ||
| buprenorphine/ naloxone | ↑ buprenorphine ↑ norbuprenorphine (metabolite of buprenorphine) | Patients should be closely monitored for sedation and cognitive effects. |
| hydrocodone/acetaminophen fentanyl | ↑ hydrocodone ↔ acetaminophen ↑ fentanyl | Reduce the dose of hydrocodone by 50% and monitor patients for respiratory depression and sedation at frequent intervals. Upon completion of VIEKIRA PAK therapy, adjust the hydrocodone dose and monitor for signs of opioid withdrawal. Careful monitoring of therapeutic effects and adverse effects of fentanyl (including potentially fatal respiratory depression) is recommended when fentanyl is co-administered with VIEKIRA PAK. |
| PROTON PUMP INHIBITORS | ||
| omeprazole | ↓ omeprazole | Monitor patients for decreased efficacy of omeprazole. Consider increasing the omeprazole dose in patients whose symptoms are not well controlled; avoid use of more than 40 mg per day of omeprazole. |
| PHOSPHODIESTERASE-5 (PDE5) INHIBITOR | ||
| sildenafil* when dosed as Revatio for the treatment of pulmonary arterial hypertension (PAH) | ↑ sildenafil | Contraindicated due to potential for sildenafil-associated adverse events such as visual disturbances, hypotension, priapism, and syncope [see Contraindications (4)]. |
| SEDATIVES/HYPNOTICS | ||
| triazolam* orally administered midazolam* | ↑ triazolam ↑ midazolam | Contraindicated due to potential for serious and/or life threatening events such as prolonged or increased sedation or respiratory depression [see Contraindications (4)]. |
| SEDATIVES/HYPNOTICS | ||
| alprazolam | ↑ alprazolam | For contraindicated Sedatives/Hypnotics [see Contraindications (4)]. Clinical monitoring of patients is recommended. A decrease in alprazolam dose can be considered based on clinical response. |
| diazepam | ↓ diazepam ↓ nordiazepam (metabolite of diazepam) | Increase dose if clinically indicated. |
| See Clinical Pharmacology, Tables 7 and 8. The direction of the arrow indicates the direction of the change in exposures (Cmax and AUC) (↑ = increase of more than 20%, ↓ = decrease of more than 20%, ↔ = no change or change less than 20%). *not studied. |
||
7.4 Drugs without Clinically Significant Interactions with VIEKIRA PAK
No dose adjustments are recommended when VIEKIRA PAK is co-administered with the following medications: abacavir, dolutegravir, digoxin, duloxetine, emtricitabine/tenofovir disoproxil fumarate, escitalopram, lamivudine, methadone, progestin only contraceptives, raltegravir, sofosbuvir, sulfamethoxazole, trimethoprim, and zolpidem.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
If VIEKIRA PAK is administered with ribavirin, the combination regimen is contraindicated in pregnant women and in men whose female partners are pregnant. Refer to the ribavirin prescribing information for more information on use in pregnancy.
No adequate human data are available to establish whether or not VIEKIRA PAK poses a risk to pregnancy outcomes. In animal reproduction studies, no adverse developmental effects were observed when the components of VIEKIRA PAK were administered separately during organogenesis and lactation. During organogenesis, the exposures were up to 28 and 4 times (mice and rabbits, respectively; ombitasvir), 98 and 8 times (mice and rats, respectively; paritaprevir, ritonavir), and 24 and 6 times (rats and rabbits, respectively; dasabuvir) exposures at the recommended clinical dose of VIEKIRA PAK. In rodent pre/postnatal developmental studies, maternal systemic exposures (AUC) to ombitasvir, paritaprevir and dasabuvir were approximately 25, 17 and 44 times, respectively, the exposure in humans at the recommended clinical dose [see Data].
The background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal data
Ombitasvir
Ombitasvir was administered orally to pregnant mice (0, 15, 50, or 150 mg/kg/day) and rabbits (0, 10 or 60 mg/kg/day) during the period of organogenesis (on gestation days (GD) 6 to 15, and GD 7 to 19, respectively). There were no ombitasvir-related embryofetal effects (malformations or fetal toxicity) at any dose level in either species. The systemic exposures at the highest doses were 28-times higher (mice) and 4-times higher (rabbits) than the exposures in humans at the recommended clinical dose.
In a pre- and postnatal developmental study in mice, ombitasvir was administered orally at 0, 10, 40, or 200 mg/kg/day from GD 6 to lactation day 20. There were no ombitasvir-related effects at maternal exposures 25-times higher than exposures in humans at the recommended clinical dose.
The major human metabolites of ombitasvir, M29 and M36, were tested in pregnant mice during the period of organogenesis from GD 6 to 15. M29 was administered orally at doses of 0, 1, 2.5 or 4.5 mg/kg/day. M36 was dosed orally at doses 1.5, 3, or 6 mg/kg/day. In both cases, there were no treatment related embryofetal effects (malformations or fetal toxicity) at any dose level. The highest doses produced exposures approximately 26-times higher than the exposures in humans at the recommended clinical dose.
Paritaprevir/ritonavir
Paritaprevir/ritonavir was administered orally to pregnant rats (0/0, 30/15, 100/15, 450/45 mg/kg/day) and mice (0/0, 30/30, 100/30, or 300/30 mg/kg/day) during the period of organogenesis (on GD 6 to 17, and GD 6 to 15, respectively). There were no test article-related embryofetal effects (malformations or fetal toxicity) at any dose level in either species. The highest systemic exposure of paritaprevir was 8-times higher (rats) and 98-times higher (mice) than the exposures in humans at the recommended clinical dose.
In a pre- and postnatal developmental study in rats, paritaprevir/ritonavir were administered orally at 0/0, 6/30, 30/30, or 300/30 mg/kg/day from GD 7 to lactation day 20. There were no treatment related effects at maternal exposures 17-times higher than exposures in humans at the recommended clinical dose.
Dasabuvir
Dasabuvir was administered orally to pregnant rats (0, 60, 300 and 800 mg/kg/day) and rabbits (0, 100, 200 or 400 mg/kg/day) during the period of organogenesis (on GD 6 to 17 and GD 7 to 20, respectively). There were no test article-related embryofetal effects (malformations or fetal toxicity) at any dose level in either species. The highest systemic exposure of dasabuvir was 24-times higher (rats) and 6-times higher (rabbits) than the exposures in humans at the recommended clinical dose.
In a pre- and postnatal developmental study in rats, dasabuvir was administered orally at 0, 50, 200, or 800 mg/kg/day from GD 7 to lactation day 21. There were no treatment-related effects at maternal exposures 44-times higher than exposures in humans at the recommended clinical dose.
8.2 Lactation
Risk Summary
It is not known whether VIEKIRA PAK and its metabolites are present in human breast milk, affect human milk production or have effects on the breastfed infant. Unchanged ombitasvir, paritaprevir and its hydrolysis product M13, and dasabuvir were the predominant components observed in the milk of lactating rats, without effect on nursing pups [see Data].
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for VIEKIRA PAK and any potential adverse effects on the breastfed child from VIEKIRA PAK or from the underlying maternal condition.
If VIEKIRA PAK is administered with ribavirin, the nursing mother’s information for ribavirin also applies to this combination regimen. Refer to the ribavirin prescribing information for more information on use during lactation.
Data
Animal Data
Ombitasvir
No effects of ombitasvir on growth and postnatal development were observed in nursing pups at the highest dose tested (200 mg/kg/day) in mice. Maternal systemic exposure (AUC) to ombitasvir was approximately 25 times the exposure in humans at the recommended clinical dose. Although not measured directly, ombitasvir was likely present in the milk of lactating mice in this study, since systemic exposure was observed in nursing pups on post-natal day 21 (approximately 16% of maternal exposure).
When ombitasvir was administered to lactating rats (5 mg/kg on post-partum day 10 to 11), milk exposure (AUC) was 4 times higher than that in plasma, with unchanged parent drug (91%) accounting for the majority of drug-related material in milk.
Paritaprevir/ritonavir
No effects of paritaprevir/ritonavir on growth and postnatal development were observed in nursing pups at the highest dose tested (300/30 mg/kg/day) in rats. Maternal systemic exposure (AUC) to paritaprevir was approximately 17 times the exposure in humans at the recommended clinical dose. Although not measured directly, paritaprevir was likely present in the milk of lactating rats at the high dose in this study, since systemic exposure was observed in nursing pups on post-natal day 15 (approximately 0.3 % of maternal exposure).
When paritaprevir/ritonavir was administered to lactating rats (30/15 mg/kg on post-partum day 10 to 11), milk exposure (AUC) was half that in plasma, with the hydrolysis product M13 (84%) and unchanged parent drug (16%) accounting for all paritaprevir-related material in milk.
Dasabuvir
No effects of dasabuvir on growth and postnatal development were observed in nursing pups at the highest dose tested (800 mg/kg/day) in rats. Maternal systemic exposure (AUC) to dasabuvir was approximately 44 times the exposure in humans at the recommended clinical dose. Although not measured directly, dasabuvir was likely present in the milk of lactating rats in this study, since systemic exposure was observed in nursing pups on post-natal day 14 (approximately 14% of maternal exposure).
When dasabuvir was administered to lactating rats (5 mg/kg on post-partum day 10 to 11), milk exposure (AUC) was 2 times higher than that in plasma, with unchanged parent drug (78%) accounting for the majority of drug-related material in milk.
8.3 Females and Males of Reproductive Potential
If VIEKIRA PAK is administered with ribavirin, the information for ribavirin with regard to pregnancy testing, contraception, and infertility also applies to this combination regimen. Refer to ribavirin prescribing information for additional information.
8.4 Pediatric Use
Safety and effectiveness of VIEKIRA PAK in pediatric patients less than 18 years of age have not been established.
8.5 Geriatric Use
No dosage adjustment of VIEKIRA PAK is warranted in geriatric patients. Of the total number of subjects in clinical studies of VIEKIRA PAK, 8.5% (174/2053) were 65 and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger subjects, but greater sensitivity of some older individuals cannot be ruled out.
8.6 Hepatic Impairment
No dosage adjustment of VIEKIRA PAK is required in patients with mild hepatic impairment (Child-Pugh A). VIEKIRA PAK is contraindicated in patients with moderate to severe (Child-Pugh B and C) hepatic impairment [see Contraindications (4), Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)].
8.7 Renal Impairment
No dosage adjustment of VIEKIRA PAK is required in patients with mild, moderate or severe renal impairment, including those on dialysis. For patients that require ribavirin, refer to the ribavirin prescribing information for information regarding use in patients with renal impairment [see Clinical Pharmacology (12.3)].
10 OVERDOSAGE
In case of overdose, it is recommended that the patient be monitored for any signs or symptoms of adverse reactions and appropriate symptomatic treatment instituted immediately.
11 DESCRIPTION
VIEKIRA PAK is ombitasvir, paritaprevir, ritonavir fixed dose combination tablets copackaged with dasabuvir tablets.
Ombitasvir, paritaprevir, ritonavir fixed dose combination tablet includes a hepatitis C virus NS5A inhibitor (ombitasvir), a hepatitis C virus NS3/4A protease inhibitor (paritaprevir), and a CYP3A inhibitor (ritonavir) that inhibits CYP3A mediated metabolism of paritaprevir, thereby providing increased plasma concentration of paritaprevir. Dasabuvir is a hepatitis C virus non-nucleoside NS5B palm polymerase inhibitor, which is supplied as separate tablets in the copackage. Both tablets are for oral administration.
Ombitasvir
The chemical name of ombitasvir is Dimethyl ([(2S,5S)-1-(4-tert-butylphenyl) pyrrolidine-2,5-diyl]bis{benzene-4,1-diylcarbamoyl(2S)pyrrolidine-2,1-diyl[(2S)-3-methyl-1-oxobutane-1,2-diyl]})biscarbamate hydrate. The molecular formula is C50H67N7O8•4.5H2O (hydrate) and the molecular weight for the drug substance is 975.20 (hydrate). The drug substance is white to light yellow to light pink powder, and is practically insoluble in aqueous buffers but is soluble in ethanol. Ombitasvir has the following molecular structure:
![the following molecular structure for The chemical name of ombitasvir is Dimethyl ([(2S,5S)-1-(4-tert-butylphenyl) pyrrolidine-2,5-diyl]bis{benzene-4,1-diylcarbamoyl(2S)pyrrolidine-2,1-diyl[(2S)-3-methyl-1-oxobutane-1,2-diyl]})biscarbamate hydrate. The molecular formula is C50H67N7O8•4.5H2O (hydrate) and the molecular weight for the drug substance is 975.20 (hydrate). The drug substance is white to light yellow to light pink powder, and is practically insoluble in aqueous buffers but is soluble in ethanol.](/dailymed/image.cfm?name=viekira-pak-spl-01.jpg&archiveid=718743)
Paritaprevir
The chemical name of paritaprevir is (2R,6S,12Z,13aS,14aR,16aS)-N-(cyclopropylsulfonyl)-6-{[(5-methylpyrazin-2-yl)carbonyl]amino}-5,16-dioxo-2-(phenanthridin-6-yloxy)-1,2,3,6,7,8,9,10,11,13a,14,15,16,16a-tetradecahydrocyclopropa[e]pyrrolo[1,2-a][1,4] diazacyclopentadecine-14a(5H)-carboxamide dihydrate. The molecular formula is C40H43N7O7S•2H2O (dihydrate) and the molecular weight for the drug substance is 801.91 (dihydrate). The drug substance is white to off-white powder with very low water solubility. Paritaprevir has the following molecular structure:
![the following molecular structure for The chemical name of paritaprevir is (2R,6S,12Z,13aS,14aR,16aS)-N-(cyclopropylsulfonyl)-6-{[(5-methylpyrazin-2-yl)carbonyl]amino}-5,16-dioxo-2-(phenanthridin-6-yloxy)-1,2,3,6,7,8,9,10,11,13a,14,15,16,16a-tetradecahydrocyclopropa[e]pyrrolo[1,2-a][1,4] diazacyclopentadecine-14a(5H)-carboxamide dihydrate. The molecular formula is C40H43N7O7S•2H2O (dihydrate) and the molecular weight for the drug substance is 801.91 (dihydrate). The drug substance is white to off-white powder with very low water solubility.](/dailymed/image.cfm?name=viekira-pak-spl-02.jpg&archiveid=718743)
Ritonavir
The chemical name of ritonavir is [5S-(5R*,8R*,10R*,11R*)]10-Hydroxy-2-methyl-5-(1-methyethyl)-1-[2-(1-methylethyl)-4-thiazolyl]-3,6-dioxo-8,11-bis(phenylmethyl)-2,4,7,12-tetraazatridecan-13-oic acid,5-thiazolylmethyl ester. The molecular formula is C37H48N6O5S2 and the molecular weight for the drug substance is 720.95. The drug substance is white to off white to light tan powder practically insoluble in water and freely soluble in methanol and ethanol. Ritonavir has the following molecular structure:
![the following molecular structure for The chemical name of ritonavir is [5S-(5R*,8R*,10R*,11R*)]10-Hydroxy-2-methyl-5-(1-methyethyl)-1-[2-(1-methylethyl)-4-thiazolyl]-3,6-dioxo-8,11-bis(phenylmethyl)-2,4,7,12-tetraazatridecan-13-oic acid,5-thiazolylmethyl ester. The molecular formula is C37H48N6O5S2 and the molecular weight for the drug substance is 720.95. The drug substance is white to off white to light tan powder practically insoluble in water and freely soluble in methanol and ethanol.](/dailymed/image.cfm?name=viekira-pak-spl-03.jpg&archiveid=718743)
Ombitasvir, Paritaprevir, Ritonavir Fixed-Dose Combination Tablets
Ombitasvir, paritaprevir, and ritonavir film-coated tablets are co-formulated immediate release tablets. The tablet contains copovidone, K value 28, vitamin E polyethylene glycol succinate, propylene glycol monolaurate Type I, sorbitan monolaurate, colloidal silicon dioxide/colloidal anhydrous silica, sodium stearyl fumarate, polyvinyl alcohol, polyethylene glycol 3350/macrogol 3350, talc, titanium dioxide, and iron oxide red. The strength for the tablet is 12.5 mg ombitasvir, 75 mg paritaprevir, 50 mg ritonavir.
Dasabuvir
The chemical name of dasabuvir is Sodium 3-(3-tert-butyl-4-methoxy-5-{6-[(methylsulfonyl)amino]naphthalene-2-yl}phenyl)-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-ide hydrate (1:1:1). The molecular formula is C26H26N3O5S•Na•H2O (salt, hydrate) and the molecular weight of the drug substance is 533.57 (salt, hydrate). The drug substance is white to pale yellow to pink powder, slightly soluble in water and very slightly soluble in methanol and isopropyl alcohol. Dasabuvir has the following molecular structure:
![the following molecular structure for The chemical name of dasabuvir is Sodium 3-(3-tert-butyl-4-methoxy-5-{6-[(methylsulfonyl)amino]naphthalene-2-yl}phenyl)-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-ide hydrate (1:1:1). The molecular formula is C26H26N3O5S•Na•H2O (salt, hydrate) and the molecular weight of the drug substance is 533.57 (salt, hydrate). The drug substance is white to pale yellow to pink powder, slightly soluble in water and very slightly soluble in methanol and isopropyl alcohol.](/dailymed/image.cfm?name=viekira-pak-spl-04.jpg&archiveid=718743)
Dasabuvir is formulated as a 250 mg film-coated, immediate release tablet containing microcrystalline cellulose (D50-100 um), microcrystalline cellulose (D50-50 um), lactose monohydrate, copovidone, croscarmellose sodium, colloidal silicon dioxide/anhydrous colloidal silica, magnesium stearate, polyvinyl alcohol, titanium dioxide, polyethylene glycol 3350/macrogol 3350, talc, and iron oxide yellow, iron oxide red and iron oxide black. Each tablet contains 270.3 mg dasabuvir sodium monohydrate equivalent to 250 mg dasabuvir.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
VIEKIRA PAK combines three direct-acting hepatitis C virus antiviral agents with distinct mechanisms of action [see Microbiology (12.4)].
Ritonavir is not active against HCV. Ritonavir is a potent CYP3A inhibitor that increases peak and trough plasma drug concentrations of paritaprevir and overall drug exposure (i.e., area under the curve).
12.2 Pharmacodynamics
Cardiac Electrophysiology
The effect of a combination of ombitasvir, paritaprevir, ritonavir, and dasabuvir on QTc interval was evaluated in a randomized, double blind, placebo and active-controlled (moxifloxacin 400 mg) 4-way crossover thorough QT study in 60 healthy subjects. At concentrations approximately 6, 1.8 and 2 times the therapeutic concentrations of paritaprevir, ombitasvir, and dasabuvir, the combination did not prolong QTc to any clinically relevant extent.
12.3 Pharmacokinetics
The pharmacokinetic properties of the components of VIEKIRA PAK are provided in Table 5. Based on the population pharmacokinetic analysis, the median steady-state pharmacokinetic parameters of ombitasvir, paritaprevir, ritonavir and dasabuvir in HCV-infected subjects are provided in Table 6.
| Ombitasvir | Paritaprevir | Ritonavir | Dasabuvir | |
| Absorption | ||||
| Tmax (hr) | ~ 5 | ~ 4-5 | ~ 4-5 | ~ 4 |
| Absolute bioavailability (%) | 48 | 53 | NA | 70 |
| Effect of moderate fat meal (relative to fasting)a | 1.82 (1.61-2.05) | 3.11 (2.16-4.46) | 1.49 (1.23-1.79) | 1.30 (1.08-1.55) |
| Effect of high fat meal (relative to fasting)a | 1.76 (1.56-1.99) | 2.80 (1.95-4.02) | 1.44 (1.19-1.73) | 1.22 (1.01-1.46) |
| Accumulationb | 0.90- to 1.03-fold | 1.5- to 2-fold | 0.96-fold | |
| Distribution | ||||
| % Bound to human plasma proteins | 99.9 | 97-98.6 | >99 | >99.5 |
| Blood-to-plasma ratio | 0.49 | 0.7 | 0.6 | 0.7 |
| Volume of distribution at steady state (Vss) (L) | 173 | 103 | 21.5c | 149 |
| Metabolism | ||||
| Metabolism | amide hydrolysis followed by oxidative metabolism | CYP3A4 (major), CYP3A5 | CYP3A (major), CYP2D6 | CYP2C8 (major), CYP3A |
| Eliminationd | ||||
| Major route of elimination | biliary excretion | metabolism | metabolism | metabolism |
| t1/2 (hr)e | 21-25 | 5.5 | 4 | 5.5-6 |
| % of dose excreted in fecesf | 90.2 | 88 | 86.4 | 94.4 |
| % of dose excreted unchanged in fecesf | 87.8 | 1.1 | 33.8 | 26.2 |
| % of dose excreted in urinef | 1.91 | 8.8 | 11.3 | ~ 2 |
| % of dose excreted unchanged in urinef | 0.03 | 0.05 | 3.5 | 0.03 |
NA - data not available
|
||||
| Pharmacokinetic Parametera | Ombitasvir | Paritaprevir | Ritonavir | Dasabuvir |
| Cmax (ng/mL) | 68 | 262 | 682 | 667 |
| AUCtau (ng*h/mL)b | 1000 | 2220 | 6180 | 3240 |
|
||||
Specific Populations
Hepatic Impairment
The single dose pharmacokinetics of ombitasvir, paritaprevir, ritonavir and dasabuvir were evaluated in non-HCV infected subjects with mild hepatic impairment (Child-Pugh Category A; score of 5-6), moderate hepatic impairment (Child-Pugh Category B, score of 7-9) and severe hepatic impairment (Child-Pugh Category C, score of 10-15).
Relative to subjects with normal hepatic function, ombitasvir, paritaprevir and ritonavir AUC values decreased by 8%, 29% and 34%, respectively, and dasabuvir AUC values increased by 17% in subjects with mild hepatic impairment.
Relative to subjects with normal hepatic function, ombitasvir, ritonavir and dasabuvir AUC values decreased by 30%, 30% and 16%, respectively, and paritaprevir AUC values increased by 62% in subjects with moderate hepatic impairment.
Relative to subjects with normal hepatic function, paritaprevir, ritonavir and dasabuvir AUC values increased by 945%, 13%, and 325% respectively, and ombitasvir AUC values decreased by 54% in subjects with severe hepatic impairment [see Dosage and Administration (2.4), Contraindications (4), Warnings and Precautions (5.2) and Use in Specific Populations (8.6)].
Renal Impairment
The single dose pharmacokinetics of ombitasvir, paritaprevir, ritonavir and dasabuvir were evaluated in non-HCV infected subjects with mild (CLcr: 60 to 89 mL/min), moderate (CLcr: 30 to 59 mL/min), and severe (CLcr: 15 to 29 mL/min) renal impairment.
Overall, changes in exposure of ombitasvir, paritaprevir, ritonavir and dasabuvir in non-HCV infected subjects with mild-, moderate- and severe renal impairment are not expected to be clinically relevant. Pharmacokinetic data are not available on the use of VIEKIRA PAK in non-HCV infected subjects with End Stage Renal Disease (ESRD).
Relative to subjects with normal renal function, paritaprevir, ritonavir and dasabuvir AUC values increased by 19%, 42% and 21%, respectively, while ombitasvir AUC values were unchanged in subjects with mild renal impairment.
Relative to subjects with normal renal function, paritaprevir, ritonavir and dasabuvir AUC values increased by 33%, 80% and 37%, respectively, while ombitasvir AUC values were unchanged in subjects with moderate renal impairment.
Relative to subjects with normal renal function, paritaprevir, ritonavir and dasabuvir AUC values increased by 45%, 114% and 50%, respectively, while ombitasvir AUC values were unchanged in subjects with severe renal impairment [see Use in Specific Populations (8.7)].
Pediatric Population
The pharmacokinetics of VIEKIRA PAK in pediatric patients less than 18 years of age has not been established [see Use in Specific Populations (8.4)].
Sex
No dose adjustment is recommended based on sex or body weight.
Race/Ethnicity
No dose adjustment is recommended based on race or ethnicity.
Age
No dose adjustment is recommended in geriatric patients [see Use in Specific Populations (8.5)].
Drug Interaction Studies
See also Contraindications (4), Warnings and Precautions (5.5), Drug Interactions (7)
The effects of drugs discussed in Table 4 on the exposures of the individual components of VIEKIRA PAK are shown in Table 7. For information regarding clinical recommendations, see Drug Interactions (7).
| Co-administered
Drug | Dose of Co-
administered Drug (mg) | n | DAA | Ratio (with/without co-administered drug)
of DAA Pharmacokinetic Parameters (90% CI); No Effect = 1.00 |
||
| Cmax | AUC | Cmin | ||||
| Alprazolam | 0.5 single dose | 12 | ombitasvir | 0.98 (0.93, 1.04) | 1.00 (0.96, 1.04) | 0.98 (0.93, 1.04) |
| paritaprevir | 0.91 (0.64, 1.31) | 0.96 (0.73, 1.27) | 1.12 (1.02, 1.23) |
|||
| ritonavir | 0.92 (0.84, 1.02) | 0.96 (0.89, 1.03) | 1.01 (0.94, 1.09) |
|||
| dasabuvir | 0.93 (0.83, 1.04) | 0.98 (0.87, 1.11) | 1.00 (0.87, 1.15) |
|||
| Amlodipine | 5 single dose | 14 | ombitasvir | 1.00 (0.95, 1.06) | 1.00 (0.97, 1.04) | 1.00 (0.97, 1.04) |
| paritaprevir | 0.77 (0.64, 0.94) | 0.78 (0.68, 0.88) | 0.88 (0.80, 0.95) |
|||
| ritonavir | 0.96 (0.87, 1.06) | 0.93 (0.89, 0.98) | 0.95 (0.89, 1.01) |
|||
| dasabuvir | 1.05 (0.97, 1.14) | 1.01 (0.96, 1.06) | 0.95 (0.89, 1.01) |
|||
| Atazanavir/ ritonavira | Atazanavir 300 and ritonavir 100 once daily in the evening | 11 | ombitasvir | 0.83 (0.72, 0.96) | 0.90 (0.78, 1.02) | 1.00 (0.89, 1.13) |
| paritaprevir | 2.19 (1.61, 2.98) | 3.16 (2.40, 4.17) | 11.95 (8.94, 15.98) |
|||
| ritonavir | 1.60 (1.38, 1.86) | 3.18 (2.74, 3.69) | 24.65 (18.64, 32.60) |
|||
| dasabuvir | 0.81 (0.73, 0.91) | 0.81 (0.71, 0.92) | 0.80 (0.65, 0.98) |
|||
| Carbamazepine | 200 once daily followed by 200 twice daily | 12 | ombitasvir | 0.69 (0.61, 0.78) | 0.69 (0.64, 0.74) | NA |
| paritaprevir | 0.34 (0.25, 0.48) | 0.30 (0.23, 0.38) | NA | |||
| ritonavir | 0.17 (0.12, 0.24) | 0.13 (0.09, 0.17) | NA | |||
| dasabuvir | 0.45 (0.41, 0.50) | 0.30 (0.28, 0.33) | NA | |||
| Carisoprodol | 250 single dose | 14 | ombitasvir | 0.98 (0.92, 1.04) | 0.95 (0.92, 0.97) | 0.96 (0.92, 0.99) |
| paritaprevir | 0.88 (0.75, 1.03) | 0.96 (0.85, 1.08) | 1.14 (1.02, 1.27) |
|||
| ritonavir | 0.94 (0.87, 1.02) | 0.94 (0.88, 0.99) | 0.95 (0.89, 1.03) |
|||
| dasabuvir | 0.96 (0.91, 1.01) | 1.02 (0.97, 1.07) | 1.00 (0.92, 1.10) |
|||
| Cyclobenzaprine | 5 single dose | 14 | ombitasvir | 0.98 (0.92, 1.04) | 1.00 (0.97, 1.03) | 1.01 (0.98, 1.04) |
| paritaprevir | 1.14 (0.99, 1.32) | 1.13 (1.00, 1.28) | 1.13 (1.01, 1.25) |
|||
| ritonavir | 0.93 (0.87, 0.99) | 1.00 (0.95, 1.06) | 1.13 (1.05, 1.21) |
|||
| dasabuvir | 0.98 (0.90, 1.07) | 1.01 (0.96, 1.06) | 1.13 (1.07, 1.18) |
|||
| Cyclosporine | 30 single doseb | 10 | ombitasvir | 0.99 (0.92, 1.07) | 1.08 (1.05, 1.11) | 1.15 (1.08, 1.23) |
| paritaprevir | 1.44 (1.16, 1.78) | 1.72 (1.49, 1.99) | 1.85 (1.58, 2.18) |
|||
| ritonavir | 0.90 (0.78, 1.04) | 1.11 (1.04, 1.19) | 1.49 (1.28, 1.74) |
|||
| dasabuvir | 0.66 (0.58, 0.75) | 0.70 (0.65, 0.76) | 0.76 (0.71, 0.82) |
|||
| Darunavirc | 800 once daily | 9 | ombitasvir | 0.86 (0.77, 0.95) | 0.86 (0.79, 0.94) | 0.87 (0.82, 0.92) |
| paritaprevir | 1.54 (1.14, 2.09) | 1.29 (1.04, 1.61) | 1.30 (1.09, 1.54) |
|||
| ritonavir | 0.84 (0.72, 0.98) | 0.85 (0.78, 0.93) | 1.07 (0.93, 1.23) |
|||
| dasabuvir | 1.10 (0.88, 1.37) | 0.94 (0.78, 1.14) | 0.90 (0.76, 1.06) |
|||
| Darunavir/ ritonavird | Darunavir 600 twice daily and ritonavir 100 once daily in the evening | 7 | ombitasvir | 0.76 (0.65, 0.88) | 0.73 (0.66, 0.80) | 0.73 (0.64, 0.83) |
| paritaprevir | 0.70 (0.43, 1.12) | 0.59 (0.44, 0.79) | 0.83 (0.69, 1.01) |
|||
| ritonavir | 1.61 (1.30, 2.00) | 1.28 (1.12, 1.45) | 0.88 (0.79, 0.99) |
|||
| dasabuvir | 0.84 (0.67, 1.05) | 0.73 (0.62, 0.86) | 0.54 (0.49, 0.61) |
|||
| Darunavir/ ritonavire | Darunavir 800 and ritonavir 100 once daily in the evening | 12 | ombitasvir | 0.87 (0.82, 0.93) | 0.87 (0.81, 0.93) | 0.87 (0.80, 0.95) |
| paritaprevir | 0.70 (0.50, 0.99) | 0.81 (0.60, 1.09) | 1.59 (1.23, 2.05) |
|||
| ritonavir | 1.19 (1.06, 1.33) | 1.70 (1.54, 1.88) | 14.15 (11.66, 17.18) |
|||
| dasabuvir | 0.75 (0.64, 0.88) | 0.72 (0.64, 0.82) | 0.65 (0.58, 0.72) |
|||
| Diazepam | 2 single dose | 13 | ombitasvir | 1.00 (0.93, 1.08) | 0.98 (0.93, 1.03) | 0.93 (0.88, 0.98) |
| paritaprevir | 0.95 (0.77, 1.18) | 0.91 (0.78, 1.07) | 0.92 (0.82, 1.03) |
|||
| ritonavir | 1.10 (1.02, 1.19) | 1.06 (0.98, 1.14) | 0.98 (0.92, 1.03) |
|||
| dasabuvir | 1.05 (0.98, 1.13) | 1.01 (0.94, 1.08) | 1.05 (0.98, 1.12) |
|||
| Ethinyl estradiol/ Norgestimate | Ethinyl estradiol 0.035 and Norgestimate 0.25 once daily | 7f
| ombitasvir | 1.05 (0.81, 1.35) | 0.97 (0.81, 1.15) | 1.00 (0.88, 1.12) |
| paritaprevir | 0.70 (0.40, 1.21) | 0.66 (0.42, 1.04) | 0.87 (0.67, 1.14) |
|||
| ritonavir | 0.80 (0.53, 1.21) | 0.71 (0.54, 0.94) | 0.79 (0.68, 0.93) |
|||
| dasabuvir | 0.51 (0.22, 1.18) | 0.48 (0.23, 1.02) | 0.53 (0.30, 0.95) |
|||
| Everolimus | 0.75 single dose | 12 | ombitasvir | 0.99 (0.95, 1.03) | 1.02 (0.99, 1.05) | 1.02 (0.99, 1.06) |
| paritaprevir | 1.22 (1.03, 1.43) | 1.26 (1.07, 1.49) | 1.06 (0.97, 1.16) |
|||
| ritonavir | 1.07 (0.99, 1.16) | 1.05 (1.00, 1.10) | 1.07 (1.02, 1.13) |
|||
| dasabuvir | 1.03 (0.90, 1.18) | 1.08 (0.98, 1.20) | 1.14 (1.05, 1.23) |
|||
| Furosemide | 20 single dose | 12 | ombitasvir | 1.14 (1.03, 1.26) | 1.07 (1.01, 1.12) | 1.12 (1.08, 1.16) |
| paritaprevir | 0.93 (0.63, 1.36) | 0.92 (0.70, 1.21) | 1.26 (1.16, 1.38) |
|||
| ritonavir | 1.10 (0.96, 1.27) | 1.04 (0.92, 1.18) | 1.07 (0.99, 1.17) |
|||
| dasabuvir | 1.12 (0.96, 1.31) | 1.09 (0.96, 1.23) | 1.06 (0.98, 1.14) |
|||
| Gemfibrozilg | 600 twice daily | 11 | ombitasvir | NA | NA | NA |
| paritaprevir | 1.21 (0.94, 1.57) | 1.38 (1.18, 1.61) | NA | |||
| ritonavir | 0.84 (0.69, 1.03) | 0.90 (0.78, 1.04) | NA | |||
| dasabuvir | 2.01 (1.71, 2.38) | 11.25 (9.05, 13.99) | NA | |||
| Hydrocodone/ Acetaminophen | 5/300 single dose | 15 | ombitasvir | 1.01 (0.93, 1.10) | 0.97 (0.93, 1.02) | 0.93 (0.90, 0.97) |
| paritaprevir | 1.01 (0.80, 1.27) | 1.03 (0.89, 1.18) | 1.10 (0.97, 1.26) |
|||
| ritonavir | 1.01 (0.90, 1.13) | 1.03 (0.96, 1.09) | 1.01 (0.93, 1.10) |
|||
| dasabuvir | 1.13 (1.01, 1.26) | 1.12 (1.05, 1.19) | 1.16 (1.08, 1.25) |
|||
| Ketoconazole | 400 once daily | 12 | ombitasvir | 0.98 (0.90, 1.06) | 1.17 (1.11, 1.24) | NA |
| paritaprevir | 1.37 (1.11, 1.69) | 1.98 (1.63, 2.42) | NA | |||
| ritonavir | 1.27 (1.04, 1.56) | 1.57 (1.36, 1.81) | NA | |||
| dasabuvir | 1.16 (1.03, 1.32) | 1.42 (1.26, 1.59) | NA | |||
| Lopinavir/ ritonavir | 400/100 twice daily | 6 | ombitasvir | 1.14 (1.01, 1.28) | 1.17 (1.07, 1.28) | 1.24 (1.14, 1.34) |
| paritaprevir | 2.04 (1.30, 3.20) | 2.17 (1.63, 2.89) | 2.36 (1.00, 5.55) |
|||
| ritonavir | 1.55 (1.16, 2.09) | 2.05 (1.49, 2.81) | 5.25 (3.33, 8.28) |
|||
| dasabuvir | 0.99 (0.75, 1.31) | 0.93 (0.75, 1.15) | 0.68 (0.57, 0.80) |
|||
| Lopinavir/ ritonavirh | 800/200 once daily | 12 | ombitasvir | 0.87 (0.83, 0.92) | 0.97 (0.94, 1.02) | 1.11 (1.06, 1.16) |
| paritaprevir | 0.99 (0.79, 1.25) | 1.87 (1.40, 2.52) | 8.23 (5.18, 13.07) |
|||
| ritonavir | 1.57 (1.34, 1.83) | 2.62 (2.32, 2.97) | 19.46 (15.93, 23.77) |
|||
| dasabuvir | 0.56 (0.47, 0.66) | 0.54 (0.46, 0.65) | 0.47 (0.39, 0.58) |
|||
| Omeprazole | 40 once daily | 11 | ombitasvir | 1.02 (0.95, 1.09) | 1.05 (0.98, 1.12) | 1.04 (0.98, 1.11) |
| paritaprevir | 1.19 (1.04, 1.36) | 1.18 (1.03, 1.37) | 0.92 (0.76, 1.12) |
|||
| ritonavir | 1.04 (0.96, 1.12) | 1.02 (0.97, 1.08) | 0.97 (0.89, 1.05) |
|||
| dasabuvir | 1.13 (1.03, 1.25) | 1.08 (0.98, 1.20) | 1.05 (0.93, 1.19) |
|||
| Pravastatin | 10 once daily | 12 | ombitasvir | 0.95 (0.89, 1.02) | 0.94 (0.89, 0.99) | 0.94 (0.89, 0.99) |
| paritaprevir | 0.96 (0.69, 1.32) | 1.13 (0.92, 1.38) | 1.39 (1.21, 1.59) |
|||
| ritonavir | 0.89 (0.73, 1.09) | 0.95 (0.86, 1.05) | 1.08 (0.98, 1.19) |
|||
| dasabuvir | 1.00 (0.87, 1.14) | 0.96 (0.85, 1.09) | 1.03 (0.91, 1.15) |
|||
| Rilpivirine | 25 once daily (morning)i | 10 | ombitasvir | 1.11 (1.02, 1.20) | 1.09 (1.04, 1.14) | 1.05 (1.01, 1.08) |
| paritaprevir | 1.30 (0.94, 1.81) | 1.23 (0.93, 1.64) | 0.95 (0.84, 1.07) |
|||
| ritonavir | 1.10 (0.98, 1.24) | 1.08 (0.93, 1.27) | 0.97 (0.91, 1.04) |
|||
| dasabuvir | 1.18 (1.02, 1.37) | 1.17 (0.99, 1.38) | 1.10 (0.89, 1.37) |
|||
| Rosuvastatin | 5 once daily | 11 | ombitasvir | 0.92 (0.82, 1.04) | 0.89 (0.83, 0.95) | 0.88 (0.83, 0.94) |
| paritaprevir | 1.59 (1.13, 2.23) | 1.52 (1.23, 1.90) | 1.43 (1.22, 1.68) |
|||
| ritonavir | 0.98 (0.84, 1.15) | 1.02 (0.93, 1.12) | 1.00 (0.90, 1.12) |
|||
| dasabuvir | 1.07 (0.92, 1.24) | 1.08 (0.92, 1.26) | 1.15 (1.05, 1.25) |
|||
| Sirolimus | 0.5 single dosej | 11 | ombitasvir | 1.03 (0.93, 1.15) | 1.02 (0.96, 1.09) | 1.05 (0.98, 1.12) |
| paritaprevir | 1.18 (0.91, 1.54) | 1.19 (0.97, 1.46) | 1.16 (1.00, 1.34) |
|||
| ritonavir | 1.00 (0.85, 1.17) | 1.04 (0.94, 1.15) | 1.10 (1.04, 1.17) |
|||
| dasabuvir | 1.04 (0.89, 1.22) | 1.07 (0.95, 1.22) | 1.13 (1.01, 1.25) |
|||
| Tacrolimus | 2 single dose | 12 | ombitasvir | 0.93 (0.88, 0.99) | 0.94 (0.89, 0.98) | 0.94 (0.91, 0.96) |
| paritaprevir | 0.57 (0.42, 0.78) | 0.66 (0.54, 0.81) | 0.73 (0.66, 0.80) |
|||
| ritonavir | 0.76 (0.63, 0.91) | 0.87 (0.79, 0.97) | 1.03 (0.89, 1.19) |
|||
| dasabuvir | 0.85 (0.73, 0.98) | 0.90 (0.80, 1.02) | 1.01 (0.91, 1.11) |
|||
Doses of ombitasvir, paritaprevir, and ritonavir were 25 mg, 150 mg and 100 mg. Doses of dasabuvir were 250 mg or 400 mg (both doses showed similar exposures). Ombitasvir, paritaprevir and ritonavir were dosed once daily and dasabuvir was dosed twice daily in all the above studies except studies with gemfibrozil, ketoconazole and carbamazepine that used single doses. |
||||||
Table 8 summarizes the effects of VIEKIRA PAK on the pharmacokinetics of co-administered drugs which showed clinically relevant changes. For information regarding clinical recommendations, see Drug Interactions (7).
| Co-administered
Drug | Dose of Co-
administered Drug (mg) | n | Ratio (with/without VIEKIRA PAK) of
Co-administered Drug Pharmacokinetic Parameters (90% CI); No Effect = 1.00 |
||
| Cmax | AUC | Cmin | |||
| Alprazolam | 0.5 single dose | 12 | 1.09 (1.03, 1.15) | 1.34 (1.15, 1.55) | NA |
| Amlodipine | 5 single dose | 14 | 1.26 (1.11, 1.44) | 2.57 (2.31, 2.86) | NA |
| Atazanavir/ ritonavira | Atazanavir 300 and ritonavir 100 once daily in the evening | 12 | 1.02 (0.92, 1.13)b | 1.19 (1.11, 1.28)b | 1.68 (1.44, 1.95)b |
| Buprenorphine | Buprenorphine: 4 to 24 once daily and Naloxone 1 to 6 once daily | 10 | 2.18 (1.78, 2.68)c | 2.07 (1.78, 2.40)c | 3.12 (2.29, 4.27)c |
| Norbuprenorphine | 2.07 (1.42, 3.01)c | 1.84 (1.30, 2.60)c | 2.10 (1.49, 2.97)c |
||
| Naloxone | 1.18 (0.81, 1.73) | 1.28 (0.92, 1.79)c | NA | ||
| Carbamazepine | 200 once daily followed by 200 twice daily | 12 | 1.10 (1.07, 1.14) | 1.17 (1.13, 1.22) | 1.35 (1.27, 1.45) |
| Carbamazepine’s metabolite, carbamazepine- 10,11-epoxide (CBZE) | 0.84 (0.82, 0.87) | 0.75 (0.73, 0.77) | 0.57 (0.54, 0.61) |
||
| Carisoprodol | 250 single dose | 14 | 0.54 (0.47, 0.63) | 0.62 (0.55, 0.70) | NA |
| Carisoprodol's metabolite, mepobramate | 1.17 (1.10, 1.25) | 1.09 (1.03, 1.16) | NA | ||
| Cyclobenzaprine | 5 single dose | 14 | 0.68 (0.61, 0.75) | 0.60 (0.53, 0.68) | NA |
| Cyclobenzaprine's metabolite norcyclobenzaprine | 1.03 (0.87, 1.23) | 0.74 (0.64, 0.85) | NA | ||
| Cyclosporine | 30 single dosed | 10 | 1.01 (0.85, 1.20)c | 5.82 (4.73, 7.14)c | 15.80 (13.81, 18.09)c |
| Darunavire | 800 once daily | 8 | 0.92 (0.87, 0.98)b | 0.76 (0.71, 0.82)b | 0.52 (0.47, 0.58)b |
| Darunavir/ ritonavirf | Darunavir 600 twice daily and ritonavir 100 once daily in the evening | 7 | 0.87 (0.79, 0.96)b | 0.80 (0.74, 0.86)b | 0.57 (0.48, 0.67)b |
| Darunavir/ ritonavirg | Darunavir 800 and ritonavir 100 once daily in the evening | 10 | 0.79 (0.70, 0.90)b | 1.34 (1.25, 1.43)b | 0.54 (0.48, 0.62)b |
| Diazepam | 2 single dose | 13 | 1.18 (1.07, 1.30) | 0.78 (0.73, 0.82) | NA |
| Diazepam's metabolite nordiazepam | 1.10 (1.03, 1.19) | 0.56 (0.45, 0.70) | NA | ||
| Ethinyl Estradiol | Ethinyl estradiol 0.035 and Norgestimate 0.25 once daily | 8 | 1.16 (0.90, 1.50) | 1.06 (0.96, 1.17) | 1.12 (0.94, 1.33) |
| Norelgestromin | 9 | 2.01 (1.77, 2.29) | 2.60 (2.30, 2.95) | 3.11 (2.51, 3.85) |
|
| Norgestrel | 9 | 2.26 (1.91, 2.67) | 2.54 (2.09, 3.09) | 2.93 (2.39, 3.57) |
|
| Everolimus | 0.75 single dose | 12 | 4.74 (4.29, 5.25) | 27.12 (24.5, 30.1) | 16.10 (14.5, 17.9) |
| Furosemide | 20 single dose | 12 | 1.42 (1.17, 1.72) | 1.08 (1.00, 1.17) | NA |
| Ketoconazole | 400 once daily | 12 | 1.15 (1.09, 1.21) | 2.17 (2.05, 2.29) | NA |
| Hydrocodone | 5 single dose | 15 | 1.27 (1.14, 1.40) | 1.90 (1.72, 2.10) | NA |
| Lopinavir/ ritonavir | 400/100 twice daily | 6 | 0.87 (0.76, 0.99)b | 0.94 (0.81, 1.10)b | 1.15 (0.93, 1.42)b |
| Lopinavir/ ritonavirh | 800/200 once daily | 12 | 0.86 (0.80, 0.93)b | 0.94 (0.87, 1.01)b | 3.18 (2.49, 4.06)b |
| Omeprazole | 40 once daily | 11 | 0.62 (0.48, 0.80) | 0.62 (0.51, 0.75) | NA |
| Pravastatin | 10 once daily | 12 | 1.37 (1.11, 1.69) | 1.82 (1.60, 2.08) | NA |
| Rosuvastatin | 5 once daily | 11 | 7.13 (5.11, 9.96) | 2.59 (2.09, 3.21) | 0.59 (0.51, 0.69) |
| Rilpivirine | 25 once daily (morning)i | 8 | 2.55 (2.08, 3.12) | 3.25 (2.80, 3.77) | 3.62 (3.12, 4.21) |
| Sirolimus | 0.5 single dosej | 11 | 6.40 (5.34, 7.68)c | 37.99 (31.5, 45.8)c | 19.55 (16.7, 22.9)c |
| Tacrolimus | 2 single dose | 12 | 3.99 (3.21, 4.97)c | 57.13 (45.53, 71.69)c | 16.56 (12.97, 21.16)c |
Doses of ombitasvir, paritaprevir, and ritonavir were 25 mg, 150 mg and 100 mg. Doses of dasabuvir were 250 mg or 400 mg (both doses showed similar exposures). Ombitasvir, paritaprevir and ritonavir were dosed once daily and dasabuvir was dosed twice daily in all the above studies except studies with ketoconazole and carbamazepine that used single doses. |
|||||
12.4 Microbiology
Mechanism of Action
VIEKIRA PAK combines three direct-acting antiviral agents with distinct mechanisms of action and non-overlapping resistance profiles to target HCV at multiple steps in the viral lifecycle.
Ombitasvir
Ombitasvir is an inhibitor of HCV NS5A, which is essential for viral RNA replication and virion assembly. The mechanism of action of ombitasvir has been characterized based on cell culture antiviral activity and drug resistance mapping studies.
Paritaprevir
Paritaprevir is an inhibitor of the HCV NS3/4A protease which is necessary for the proteolytic cleavage of the HCV encoded polyprotein (into mature forms of the NS3, NS4A, NS4B, NS5A, and NS5B proteins) and is essential for viral replication. In a biochemical assay, paritaprevir inhibited the proteolytic activity of recombinant HCV genotype 1a and 1b NS3/4A protease enzymes with IC50 values of 0.18 nM and 0.43 nM, respectively.
Dasabuvir
Dasabuvir is a non-nucleoside inhibitor of the HCV RNA-dependent RNA polymerase encoded by the NS5B gene, which is essential for replication of the viral genome. In a biochemical assay, dasabuvir inhibited a panel of genotype 1a and 1b NS5B polymerases with median IC50 values of 2.8 nM (range 2.4 nM to 4.2 nM; n = 3) and 3.7 nM (range 2.2 nM to 10.7 nM; n = 4), respectively. Based on drug resistance mapping studies of HCV genotypes 1a and 1b, dasabuvir targets the palm domain of the NS5B polymerase, and is therefore referred to as a non-nucleoside NS5B-palm polymerase inhibitor.
Antiviral Activity
Ombitasvir
The EC50 values of ombitasvir against genotype 1a-H77 and 1b-Con1 strains in HCV replicon cell culture assays were 14.1 pM and 5 pM, respectively. The median EC50 values of ombitasvir against HCV replicons containing NS5A genes from a panel of genotype 1a and 1b isolates from treatment-naïve subjects were 0.68 pM (range 0.35 to 0.88 pM; n = 11) and 0.94 pM (range 0.74 to 1.5 pM; n = 11), respectively.
Paritaprevir
The EC50 values of paritaprevir against genotype 1a-H77 and 1b-Con1 strains in the HCV replicon cell culture assay were 1.0 nM and 0.21 nM, respectively. The median EC50 values of paritaprevir against HCV replicons containing NS3 genes from a panel of genotype 1a and 1b isolates from treatment-naïve subjects were 0.68 nM (range 0.43 nM to 1.87 nM; n = 11) and 0.06 nM (range 0.03 nM to 0.09 nM; n = 9), respectively.
Ritonavir
In HCV replicon cell culture assays, ritonavir did not exhibit a direct antiviral effect and the presence of ritonavir did not affect the antiviral activity of paritaprevir.
Dasabuvir
The EC50 values of dasabuvir against genotype 1a-H77 and 1b-Con1 strains in HCV replicon cell culture assays were 7.7 nM and 1.8 nM, respectively. The median EC50 values of dasabuvir against HCV replicons containing NS5B genes from a panel of genotype 1a and 1b isolates from treatment-naïve subjects were 0.6 nM (range 0.4 nM to 2.1 nM; n = 11) and 0.3 nM (range 0.2 nM to 2 nM; n = 10), respectively.
Combination Antiviral Activity
Evaluation of pairwise combinations of ombitasvir, paritaprevir, dasabuvir and ribavirin in HCV genotype 1 replicon cell culture assays showed no evidence of antagonism in antiviral activity.
Resistance
In Cell Culture
Exposure of HCV genotype 1a and 1b replicons to ombitasvir, paritaprevir or dasabuvir resulted in the emergence of drug resistant replicons carrying amino acid substitutions in NS5A, NS3, or NS5B, respectively. Amino acid substitutions in NS5A, NS3, or NS5B selected in cell culture or identified in Phase 2b and 3 clinical trials were phenotypically characterized in genotype 1a or 1b replicons.
For ombitasvir, in HCV genotype 1a replicons single NS5A substitutions M28T/V, Q30E/R, L31V, H58D, and Y93C/H/L/N reduced ombitasvir antiviral activity by 58- to 67,000-fold. In genotype 1b replicons, single NS5A substitutions L28T, L31F/V, and Y93H reduced ombitasvir antiviral activity by 8- to 661-fold. In general, combinations of ombitasvir resistance-associated substitutions in HCV genotype 1a or 1b replicons further reduced ombitasvir antiviral activity.
For paritaprevir, in HCV genotype 1a replicons single NS3 substitutions F43L, R155G/K/S, A156T, and D168A/E/F/H/N/V/Y reduced paritaprevir antiviral activity by 7- to 219-fold. An NS3 Q80K substitution in a genotype 1a replicon reduced paritaprevir antiviral activity by 3-fold. Combinations of V36M, Y56H, or E357K with R155K or D168 substitutions reduced the activity of paritaprevir by an additional 2- to 7-fold relative to the single R155K or D168 substitutions in genotype 1a replicons. In genotype 1b replicons single NS3 substitutions A156T and D168A/H/V reduced paritaprevir antiviral activity by 7- to 159-fold. The combination of Y56H with D168 substitutions reduced the activity of paritaprevir by an additional 16- to 26-fold relative to the single D168 substitutions in genotype 1b replicons.
For dasabuvir, in HCV genotype 1a replicons single NS5B substitutions C316Y, M414I/T, E446K/Q, Y448C/H, A553T, G554S, S556G/R, and Y561H reduced dasabuvir antiviral activity by 8- to 1,472-fold. In genotype 1b replicons, single NS5B substitutions C316H/N/Y, S368T, N411S, M414I/T, Y448C/H, A553V, S556G and D559G reduced dasabuvir antiviral activity by 5- to 1,569-fold.
In Clinical Studies
In a pooled analysis of subjects treated with regimens containing ombitasvir, paritaprevir, and dasabuvir with or without ribavirin (for 12 or 24 weeks) in Phase 2b and Phase 3 clinical trials, resistance analyses were conducted for 64 subjects who experienced virologic failure (20 with on-treatment virologic failure, 44 with post-treatment relapse). Treatment-emergent substitutions observed in the viral populations of these subjects are shown in Table 9. Treatment-emergent substitutions were detected in all 3 HCV drug targets in 30/57 (53%) HCV genotype 1a infected subjects, and 1/6 (17%) HCV genotype 1b infected subjects.
| Target | Emergent Amino Acid Substitutions | Genotype 1a
N = 58a % (n) | Genotype 1b
N = 6 % (n) |
| NS3 | Any of the following NS3 substitutions: V36A/M/T, F43L, V55I, Y56H, Q80L, I132V, R155K, A156G, D168(any), P334S, S342P, E357K, V406A/I, T449I, P470S, V23A (NS4A) | 88 (51) | 67 (4) |
| V36A/M/Tb | 7 (4) | -- | |
| V55Ib | 7 (4) | -- | |
| Y56Hb | 10 (6) | 50 (3) | |
| I132Vb | 7 (4) | -- | |
| R155K | 16 (9) | -- | |
| D168 (any)d | 72 (42) | 67 (4) | |
| D168V | 59 (34) | 50 (3) | |
| P334Sb,c | 7 (4) | -- | |
| E357Kb,c | 5 (3) | 17 (1) | |
| V406A/Ib,c | 5 (3) | -- | |
| T449Ib,c | 5 (3) | -- | |
| P470Sb,c | 5 (3) | -- | |
| NS4A V23Ab | -- | 17 (1) | |
| F43Lb, Q80Lb, A156G, S342Pb,c | <5% | -- | |
| NS5A | Any of the following NS5A substitutions: K24R, M28A/T/V, Q30E/K/R, H/Q54Y, H58D/P/R, Y93C/H/N | 78 (45) | 33 (2) |
| K24R | 5 (3) | -- | |
| M28A/T/V | 33 (19) | -- | |
| Q30E/K/R | 47 (27) | -- | |
| H/Q54Y | -- | 17(1) | |
| H58D/P/R | 7 (4) | -- | |
| Y93C/N | 5 (3) | -- | |
| Y93H | -- | 33 (2) | |
| NS5B | Any of the following NS5B substitutions: G307R, C316Y, M414I/T, E446K/Q, A450V, A553I/T/V, G554S, S556G/R, G558R, D559G/I/N/V, Y561H | 67 (38) | 33 (2) |
| C316Y | 4 (2) | 17 (1) | |
| M414I | -- | 17 (1) | |
| M414T | 5 (3) | 17 (1) | |
| A553I/T/V | 7 (4) | -- | |
| S556G/R | 39 (22) | 17 (1) | |
| D559G/I/N/V | 7 (4) | -- | |
| Y561H | 5 (3) | -- | |
| G307R, E446K/Q, A450V, G554S, G558R | <5% | -- | |
|
|||
Persistence of Resistance-Associated Substitutions
The persistence of ombitasvir, paritaprevir, and dasabuvir treatment-emergent amino acid substitutions in NS5A, NS3, and NS5B, respectively, was assessed in HCV genotype 1a-infected subjects in Phase 2 trials whose virus had at least 1 treatment-emergent resistance-associated substitution in the drug target, and with available data through at least 24 weeks post-treatment. Population and clonal nucleotide sequence analyses (assay sensitivity approximately 5-10%) were conducted to detect the persistence of viral populations with treatment-emergent substitutions.
For ombitasvir, viral populations with 1 or more resistance-associated treatment-emergent substitutions in NS5A persisted at detectable levels through at least Post-Treatment Week 24 in 24/24 (100%) subjects, and through Post-Treatment Week 48 in 18/18 (100%) subjects with available data.
For paritaprevir, viral populations with 1 or more treatment-emergent substitutions in NS3 persisted at detectable levels through at least Post-Treatment Week 24 in 17/29 (59%) subjects, and through Post-Treatment Week 48 in 5/22 (23%) subjects with available data. Resistance-associated variant R155K remained detectable in 5/8 (63%) subjects through Post-Treatment Week 24, and in 1/5 (20%) subjects through Post-Treatment Week 48. Resistance-associated D168 substitutions remained detectable in 6/22 (27%) subjects through Post-Treatment Week 24, and were no longer detectable through Post-Treatment Week 48.
For dasabuvir, viral populations with 1 or more treatment-emergent substitutions in NS5B persisted at detectable levels through at least Post-Treatment Week 24 in 11/16 (69%) subjects, and through Post-Treatment Week 48 in 8/15 (53%) subjects with available data. Treatment-emergent S556G persisted through Post-Treatment Week 48 in 6/9 (67%) subjects.
Among HCV genotype 1b infected subjects who experienced virologic failure with a regimen including ombitasvir and paritaprevir, a treatment-emergent NS5A Y93H substitution persisted through at least Post-Treatment Week 48 in 2/2 subjects, and a NS3 D168V treatment-emergent substitution persisted through Post-Treatment Week 24 in 2/4 subjects, but was no longer detectable through Post-Treatment Week 48 (0/4 subjects).
The lack of detection of virus containing a resistance-associated substitution does not indicate that the resistant virus is no longer present at clinically significant levels. The long-term clinical impact of the emergence or persistence of virus containing VIEKIRA PAK-resistance-associated substitutions is unknown.
Effect of Baseline HCV Polymorphisms on Treatment Response
A pooled analysis of subjects in the Phase 3 clinical trials of ombitasvir, paritaprevir, and dasabuvir with or without ribavirin was conducted to explore the association between baseline HCV NS5A, NS3, or NS5B resistance-associated polymorphisms and treatment outcome. Baseline samples from HCV genotype 1a infected subjects who experienced virologic failure (n=47), as well as samples from a subset of demographically matched subjects who achieved SVR (n=94), were analyzed to compare the frequencies of resistance-associated polymorphisms in these two populations. The NS3 Q80K polymorphism was detected in approximately 38% of subjects in this analysis and was enriched approximately 2-fold in virologic failure subjects compared to SVR-achieving subjects. Ombitasvir resistance-associated polymorphisms in NS5A (pooling data from all resistance-associated amino acid positions) were detected in approximately 22% of subjects in this analysis and similarly were enriched approximately 2-fold in virologic failure subjects. Dasabuvir resistance-associated polymorphisms in NS5B were detected in approximately 5% of subjects in this analysis and were not enriched in virologic failure subjects.
In contrast to the Phase 3 subset analysis, no association of NS3 or NS5A polymorphisms and treatment outcome was seen in an analysis of noncirrhotic HCV genotype 1a-infected subjects (n=174 for NS3 and n=183 for NS5A) who received ombitasvir, paritaprevir, and dasabuvir with or without ribavirin (for 12 or 24 weeks) in a Phase 2b trial.
Baseline HCV polymorphisms are not expected to have a substantial impact on the likelihood of achieving SVR when VIEKIRA PAK is used as recommended for HCV genotype 1a and 1b infected patients, based on the low virologic failure rates observed in clinical trials.
Cross-resistance
Cross-resistance is expected among NS5A inhibitors, NS3/4A protease inhibitors, and non-nucleoside NS5B-palm inhibitors by class. Dasabuvir retained full activity against HCV replicons containing a single NS5B L159F, S282T, or V321A substitution, which are associated with resistance or prior exposure to nucleos(t)ide analogue NS5B polymerase inhibitors. In clinical trials of VIEKIRA PAK, no subjects who experienced virologic failure had treatment-emergent substitutions potentially associated with resistance to nucleos(t)ide analogue NS5B polymerase inhibitors.
The impact of prior ombitasvir, paritaprevir, or dasabuvir treatment experience on the efficacy of other NS5A inhibitors, NS3/4A protease inhibitors, or NS5B inhibitors has not been studied. Similarly, the efficacy of VIEKIRA PAK has not been studied in subjects who have failed prior treatment with another NS5A inhibitor, NS3/4A protease inhibitor, or NS5B inhibitor.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis and Mutagenesis
Ombitasvir
Ombitasvir was not carcinogenic in a 6-month transgenic mouse study up to the highest dose tested (150 mg per kg per day). Similarly, ombitasvir was not carcinogenic in a 2-year rat study up to the highest dose tested (30 mg per kg per day), resulting in ombitasvir exposures approximately 16-fold higher than those in humans at 25 mg.
Ombitasvir and its major inactive human metabolites (M29, M36) were not genotoxic in a battery of in vitro or in vivo assays, including bacterial mutagenicity, chromosome aberration using human peripheral blood lymphocytes and in vivo mouse micronucleus assays.
Paritaprevir, ritonavir
Paritaprevir, ritonavir was not carcinogenic in a 6-month transgenic mouse study up to the highest dose tested (300/30 mg per kg per day). Similarly, paritaprevir, ritonavir was not carcinogenic in a 2-year rat study up to the highest dose tested (300/30 mg per kg per day), resulting in paritaprevir exposures approximately 9-fold higher than those in humans at 150 mg.
Paritaprevir was positive in an in vitro chromosome aberration test using human lymphocytes. Paritaprevir was negative in a bacterial mutation assay, and in two in vivo genetic toxicology assays (rat bone marrow micronucleus and rat liver Comet tests).
Dasabuvir
Dasabuvir was not carcinogenic in a 6-month transgenic mouse study up to the highest dose tested (2000 mg per kg per day). Similarly, dasabuvir was not carcinogenic in a 2-year rat study up to the highest dose tested (800 mg per kg per day), resulting in dasabuvir exposures approximately 19-fold higher than those in humans at 500 mg.
Dasabuvir was not genotoxic in a battery of in vitro or in vivo assays, including bacterial mutagenicity, chromosome aberration using human peripheral blood lymphocytes and in vivo rat micronucleus assays.
If VIEKIRA PAK is administered with ribavirin, refer to the prescribing information for ribavirin for information on carcinogenesis, and mutagenesis.
Impairment of Fertility
Ombitasvir
Ombitasvir had no effects on embryo-fetal viability or on fertility when evaluated in mice up to the highest dose of 200 mg per kg per day. Ombitasvir exposures at this dose were approximately 25-fold the exposure in humans at the recommended clinical dose.
Paritaprevir, ritonavir
Paritaprevir, ritonavir had no effects on embryo-fetal viability or on fertility when evaluated in rats up to the highest dose of 300/30 mg per kg per day. Paritaprevir exposures at this dose were approximately 2- to 5-fold the exposure in humans at the recommended clinical dose.
Dasabuvir
Dasabuvir had no effects on embryo-fetal viability or on fertility when evaluated in rats up to the highest dose of 800 mg per kg per day. Dasabuvir exposures at this dose were approximately 16-fold the exposure in humans at the recommended clinical dose.
If VIEKIRA PAK is administered with ribavirin, refer to the prescribing information for ribavirin for information on Impairment of Fertility.
14 CLINICAL STUDIES
14.1 Description of Clinical Trials
Table 10 presents the clinical trial design including different treatment arms that were conducted with VIEKIRA PAK with or without ribavirin in subjects with chronic hepatitis C (HCV) genotype 1 (GT1) infection. For detailed description of trial design and recommended regimen and duration [see Dosage and Administration (2) and Clinical Studies (14)].
| Trial | Population | Study Arms and Duration
(Number of Subjects Treated) |
| SAPPHIRE-I (double-blind) | GT1 (a and b) TNa without cirrhosis |
|
| SAPPHIRE-II (double-blind) | GT1 (a and b) TEb without cirrhosis |
|
| PEARL-II (open-label) | GT1b TE without cirrhosis |
|
| PEARL-III (double-blind) | GT1b TN without cirrhosis |
|
| PEARL-IV (double-blind) | GT1a TN without cirrhosis |
|
| TURQUOISE-II (open-label) | GT1 (a and b) TN & TE with compensated cirrhosis |
|
| TURQUOISE-III (open-label) | GT1b TN & TE with compensated cirrhosis |
|
|
||
VIEKIRA PAK with RBV was also evaluated in the following two studies:
- HCV GT1-infected liver transplant recipients (CORAL-I) [see Clinical Studies (14.5)].
- Subjects with HCV GT1 co-infected with HIV-1 (TURQUOISE-I) [see Clinical Studies (14.6)].
In all clinical trials, the ombitasvir, paritaprevir, ritonavir dose was 25/150/100 mg once daily and the dasabuvir dose was 250 mg twice daily. Doses of drugs in VIEKIRA PAK were not adjusted. For subjects who received RBV, the RBV dose was 1000 mg per day for subjects weighing less than 75 kg or 1200 mg per day for subjects weighing greater than or equal to 75 kg. RBV dose adjustments were performed according to the RBV labeling.
In all clinical trials, sustained virologic response was defined as HCV RNA below the lower limit of quantification (<LLOQ) 12 weeks after the end of treatment (SVR12). Plasma HCV RNA levels were measured using the COBAS TaqMan HCV test (version 2.0), for use with the High Pure System, which has an LLOQ of 25 IU per mL. Outcomes for subjects not achieving an SVR12 were recorded as on-treatment virologic failure (VF), post-treatment virologic relapse through post-treatment Week 12 or failure due to other non-virologic reasons (e.g., premature discontinuation, adverse event, lost to follow-up, consent withdrawn).
14.2 Clinical Trial Results in Adults with Chronic HCV Genotype 1a and 1b Infection without Cirrhosis
Subjects with Chronic HCV GT1a Infection without Cirrhosis
Subjects with HCV GT1a infection without cirrhosis treated with VIEKIRA PAK with RBV for 12 weeks in SAPPHIRE-I and -II and in PEARL-IV [see Clinical Studies (14.1)] had a median age of 53 years (range: 18 to 70); 63% of the subjects were male; 90% were White; 7% were Black/African American; 8% were Hispanic or Latino; 19% had a body mass index of at least 30 kg per m2; 55% of patients were enrolled in US sites; 72% had IL28B (rs12979860) non-CC genotype; 85% had baseline HCV RNA levels of at least 800,000 IU per mL.
Table 11 presents treatment outcomes for HCV GT1a treatment-naïve and treatment-experienced subjects treated with VIEKIRA PAK with RBV for 12 weeks in SAPPHIRE-I, PEARL-IV and SAPPHIRE-II.
Treatment-naïve, HCV GT1a-infected subjects without cirrhosis treated with VIEKIRA PAK in combination with RBV for 12 weeks in PEARL-IV had a significantly higher SVR12 rate than subjects treated with VIEKIRA PAK alone (97% and 90% respectively; difference +7% with 95% confidence interval, +1% to +12%). VIEKIRA PAK alone was not studied in treatment-experienced subjects with GT1a infection.
In SAPPHIRE-I and SAPPHIRE-II, no placebo subject achieved a HCV RNA <25 IU/mL during treatment.
| VIEKIRA PAK with RBV
for 12 Weeks % (n/N) |
|
| GT1a treatment-naïve | |
| SAPPHIRE-I SVR12 | 96% (308/322) |
| Outcome for subjects without SVR12 | |
| On-treatment VF | <1% (1/322) |
| Relapse | 2% (6/314) |
| Other | 2% (7/322) |
| PEARL-IV SVR12 | 97% (97/100) |
| Outcome for subjects without SVR12 | |
| On-treatment VF | 1% (1/100) |
| Relapse | 1% (1/98) |
| Other | 1% (1/100) |
| GT1a treatment-experienced | |
| SAPPHIRE-II SVR12 | 96% (166/173) |
| Outcome for subjects without SVR12 | |
| On-treatment VF | 0% (0/173) |
| Relapse | 3% (5/172) |
| Other | 1% (2/173) |
| SVR12 by Prior pegIFN Experience | |
| Null Responder | 95% (83/87) |
| Partial Responder | 100% (36/36) |
| Relapser | 94% (47/50) |
Subjects with Chronic HCV GT1b Infection without Cirrhosis
Subjects with HCV GT1b infection without cirrhosis were treated with VIEKIRA PAK with or without RBV for 12 weeks in PEARL-II and -III [see Clinical Studies (14.1)]. Subjects had a median age of 52 years (range: 22 to 70); 47% of the subjects were male; 93% were White; 5% were Black/African American; 2% were Hispanic or Latino; 21% had a body mass index of at least 30 kg per m2; 21% of patients were enrolled in US sites; 83% had IL28B (rs12979860) non-CC genotype; 77% had baseline HCV RNA levels of at least 800,000 IU per mL.
The SVR rate for HCV GT1b-infected subjects without cirrhosis treated with VIEKIRA PAK without RBV for 12 weeks in PEARL-II (treatment-experienced: null responder, n=32; partial responder, n=26; relapser, n=33) and PEARL-III (treatment-naïve, n=209) was 100%.
14.3 Clinical Trial Results in Adults with Chronic HCV Genotype 1a and 1b Infection and Compensated Cirrhosis
VIEKIRA PAK with and without ribavirin was evaluated in two clinical trials in patients with compensated cirrhosis.
TURQUOISE-II was an open-label trial that enrolled 380 HCV GT1-infected subjects with cirrhosis and mild hepatic impairment (Child-Pugh A) who were either treatment-naïve or did not achieve SVR with prior treatment with pegIFN/RBV. Subjects were randomized to receive VIEKIRA PAK in combination with RBV for either 12 or 24 weeks of treatment. Treated subjects had a median age of 58 years (range: 21 to 71); 70% of the subjects were male; 95% were White; 3% were Black/African American; 12% were Hispanic or Latino; 28% had a body mass index of at least 30 kg per m2; 43% of patients were enrolled in US sites; 82% had IL28B (rs12979860) non-CC genotype; 86% had baseline HCV RNA levels of at least 800,000 IU per mL; 69% had HCV GT1a infection, 31% had HCV GT1b infection; 42% were treatment-naïve, 36% were prior pegIFN/RBV null responders; 8% were prior pegIFN/RBV partial responders, 14% were prior pegIFN/RBV relapsers; 15% had platelet counts of less than 90 x 109 per L; 50% had albumin less than 4.0 mg per dL.
TURQUOISE-III was an open-label trial that enrolled 60 HCV GT1b-infected subjects with cirrhosis and mild hepatic impairment (Child-Pugh A) who were either treatment-naïve or did not achieve SVR with prior treatment with pegIFN/RBV. Subjects received VIEKIRA PAK without RBV for 12 weeks. Treated subjects had a median age of 61 years (range: 26 to 78); including 45% treatment-naïve and 55% pegIFN/RBV treatment-experienced; 25% were ≥65 years; 62% were male; 12% were Black; 5% were Hispanic or Latino; 28% had a body mass index of at least 30 kg per m2; 40% of patients were enrolled in US sites; 22% had platelet counts of less than 90 x 109 per L; 17% had albumin less than 35 g/L; 92% had baseline HCV RNA levels of at least 800,000 IU per mL; 83% had IL28B (rs12979860) non-CC genotype.
Table 12 presents treatment outcomes for GT1a- and GT1b-infected treatment-naïve and treatment-experienced subjects. In GT1a infected subjects, the overall SVR12 rate difference between 24 and 12 weeks of treatment with VIEKIRA PAK with RBV was +6% with 95% confidence interval, -0.1% to +13% with differences varying by pretreatment history.
| GT1a
(TURQUOISE-II) | GT1b
(TURQUOISE-III) |
||
| VIEKIRA PAK
with RBV for 24 Weeks % (n/N) | VIEKIRA PAK
with RBV for 12 Weeks % (n/N) | VIEKIRA PAK
without RBV for 12 Weeks % (n/N) |
|
| SVR12 | 95% (115/121) | 89% (124/140) | 100% (60/60) |
| Outcome for subjects without
SVR12 | |||
| On-treatment VF | 2% (3/121) | <1% (1/140) | 0 |
| Relapse | 1% (1/116) | 8% (11/135) | 0 |
| Other | 2% (2/121) | 3% (4/140) | 0 |
| SVR12 for Naïve | 95% (53/56) | 92% (59/64) | 100% (27/27) |
| SVR12 by Prior pegIFN
Experience | 100% (33/33) | ||
| Null Responder | 93% (39/42) | 80% (40/50) | 100% (7/7) |
| Partial Responder | 100% (10/10) | 100% (11/11) | 100% (5/5) |
| Relapser | 100% (13/13) | 93% (14/15) | 100% (3/3) |
14.4 Effect of Ribavirin Dose Reductions on SVR12
Seven percent of subjects (101/1551) treated with VIEKIRA PAK with RBV had a RBV dose adjustment due to a decrease in hemoglobin level; of these, 98% (98/100) achieved an SVR12.
14.5 Clinical Trial of Selected Liver Transplant Recipients (CORAL-I)
VIEKIRA PAK with RBV was administered for 24 weeks to 34 HCV GT1-infected liver transplant recipients who were at least 12 months post transplantation at enrollment with normal hepatic function and mild fibrosis (Metavir fibrosis score F2 or lower). The initial dose of RBV was left to the discretion of the investigator with 600 to 800 mg per day being the most frequently selected dose range at initiation of VIEKIRA PAK and at the end of treatment.
Of the 34 subjects (29 with HCV GT1a infection and 5 with HCV GT1b infection) enrolled, (97%) achieved SVR12 (97% in subjects with GT1a infection and 100% of subjects with GT1b infection). One subject with HCV GT1a infection relapsed post-treatment.
14.6 Clinical Trial in Subjects with HCV/HIV-1 Co-infection (TURQUOISE-I)
In an open-label clinical trial 63 subjects with HCV GT1 infection co-infected with HIV-1 were treated for 12 or 24 weeks with VIEKIRA PAK in combination with RBV. Subjects were on a stable HIV-1 antiretroviral therapy (ART) regimen that included tenofovir disoproxil fumarate plus emtricitabine or lamivudine, administered with ritonavir boosted atazanavir or raltegravir. Subjects on atazanavir stopped the ritonavir component of their HIV-1 ART regimen upon initiating treatment with VIEKIRA PAK in combination with RBV. Atazanavir was taken with the morning dose of VIEKIRA PAK. The ritonavir component of the HIV-1 ART regimen was restarted after completion of treatment with VIEKIRA PAK and RBV.
Treated subjects had a median age of 51 years (range: 31 to 69); 24% of subjects were black; 81% of subjects had IL28B (rs12979860) non-CC genotype; 19% of subjects had compensated cirrhosis; 67% of subjects were HCV treatment-naïve; 33% of subjects had failed prior treatment with pegIFN/RBV; 89% of subjects had HCV genotype 1a infection.
The SVR12 rates were 91% (51/56) for subjects with HCV GT1a infection and 100% (7/7) for those with HCV GT1b infection. Of the 5 subjects who were non-responders, 1 experienced virologic breakthrough, 1 discontinued treatment, 1 experienced relapse and 2 subjects had evidence of HCV re-infection post-treatment.
One subject had confirmed HIV-1 RNA >400 copies/mL during the post-treatment period. This subject had no evidence of resistance to the ART regimen. No subjects switched their ART regimen due to loss of plasma HIV-1 RNA suppression.
14.7 Durability of Response
In an open-label clinical trial, 92% of subjects (526/571) who received various combinations of the direct acting antivirals included in VIEKIRA PAK with or without RBV achieved SVR12, and 99% of those who achieved SVR12 maintained their response through 48 weeks post-treatment (SVR48).
16 HOW SUPPLIED/STORAGE AND HANDLING
VIEKIRA PAK is dispensed in a monthly carton for a total of 28 days of therapy. Each monthly carton contains four weekly cartons. Each weekly carton contains seven daily dose packs.
Each child resistant daily dose pack contains four tablets: two 12.5/75/50 mg ombitasvir, paritaprevir, ritonavir tablets and two 250 mg dasabuvir tablets, and indicates which tablets need to be taken in the morning and evening. The NDC number is 0074-3093-28.
Ombitasvir, paritaprevir, ritonavir 12.5/75/50 mg tablets are pink-colored, film-coated, oblong biconvex shaped, debossed with “AV1” on one side. Dasabuvir 250 mg tablets are beige-colored, film-coated, oval-shaped, debossed with “AV2” on one side.
Store at or below 30°C (86°F).
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Inform patients to review the Medication Guide for ribavirin [see Warnings and Precautions (5.4)].
Risk of Hepatitis B Virus Reactivation in Patients Coinfected with HCV and HBV
Inform patients that HBV reactivation can occur in patients coinfected with HBV during or after treatment of HCV infection. Advise patients to tell their healthcare provider if they have a history of hepatitis B virus infection [see Warnings and Precautions (5.1)].
Risk of ALT Elevations or Hepatic Decompensation and Failure
Inform patients to watch for early warning signs of liver inflammation or failure, such as fatigue, weakness, lack of appetite, nausea and vomiting, as well as later signs such as jaundice, onset of confusion, abdominal swelling, and discolored feces, and to consult their health care professional without delay if such symptoms occur [see Warnings and Precautions (5.2 and 5.3) and Adverse Reactions (6)].
Pregnancy
Advise patients that extreme care must be taken to avoid pregnancy during treatment with VIEKIRA PAK with ribavirin and within 6 months of stopping ribavirin in both female patients and in female partners of male patients. Inform patients to notify their health care provider immediately in the event of a pregnancy [see Use in Specific Populations (8.1)].
Drug Interactions
Inform patients that VIEKIRA PAK may interact with some drugs; therefore, patients should be advised to report to their healthcare provider the use of any prescription, non-prescription medication or herbal products [see Contraindications (4), Warnings and Precautions (5.5) and Drug Interactions (7)].
Inform patients that contraceptives containing ethinyl estradiol are contraindicated with VIEKIRA PAK [see Contraindications (4) and Warnings and Precautions (5.3)].
Administration
Advise patients to take VIEKIRA PAK every day at the regularly scheduled time with a meal without regard to fat or calorie content [see Dosage and Administration (2.1)].
Inform patients that it is important not to miss or skip doses and to take VIEKIRA PAK for the duration that is recommended by the healthcare provider.
Advise patients not to remove tablets from the daily dose pack until they are ready to take them.
Manufactured by AbbVie Inc., North Chicago, IL 60064.
VIEKIRA PAK and NORVIR are trademarks of AbbVie Inc.
© 2019 AbbVie Inc. All rights reserved.
03-C075
| MEDICATION GUIDE
VIEKIRA PAK (vee-KEE-rah-pak) (ombitasvir, paritaprevir, and ritonavir tablets; dasabuvir tablets) co-packaged for oral use |
|||
| Important: When taking VIEKIRA PAK in combination with ribavirin, you should also read the Medication Guide that comes with ribavirin. | |||
| What is the most important information I should know about VIEKIRA PAK?
VIEKIRA PAK can cause serious side effects, including:
|
|||
|
|
||
| For more information about side effects, see the section “What are the possible side effects of VIEKIRA PAK?” | |||
| What is VIEKIRA PAK?
VIEKIRA PAK is a prescription medicine used with or without ribavirin to treat adults with genotype 1 chronic (lasting a long time) hepatitis C virus (HCV) infection. VIEKIRA PAK can be used in people who have compensated cirrhosis. VIEKIRA PAK is not for people with advanced cirrhosis (decompensated). If you have cirrhosis, talk to your healthcare provider before taking VIEKIRA PAK. VIEKIRA PAK contains 2 different types of tablets:
|
|||
Do not take VIEKIRA PAK if you:
○ gemfibrozil ○ lomitapide ○ lovastatin ○ lurasidone ○ midazolam, when taken by mouth ○ phenytoin ○ phenobarbital ○ pimozide ○ ranolazine ○ rifampin ○ sildenafil citrate, when taken for pulmonary artery hypertension (PAH) ○ simvastatin ○ sirolimus ○ St. John’s wort (Hypericum perforatum) or a product that contains St. John’s wort ○ tacrolimus ○ triazolam
|
|||
Before taking VIEKIRA PAK, tell your healthcare provider about all of your medical conditions, including if you:
Keep a list of your medicines to show your healthcare provider and pharmacist.
|
|||
How should I take VIEKIRA PAK?
|
|||
| What are the possible side effects of VIEKIRA PAK?
VIEKIRA PAK can cause serious side effects, including: See “What is the most important information I should know about VIEKIRA PAK?” Common side effects of VIEKIRA PAK when used with ribavirin include: |
|||
|
|
||
Common side effects of VIEKIRA PAK when used without ribavirin include:
|
|||
| How should I store VIEKIRA PAK?
Store VIEKIRA PAK at or below 86°F (30°C). Do not remove tablets from the daily dose pack until you are ready to take them. Keep VIEKIRA PAK and all medicines out of the reach of children. |
|||
| General information about the safe and effective use of VIEKIRA PAK
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use VIEKIRA PAK for a condition for which it was not prescribed. Do not give VIEKIRA PAK to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about VIEKIRA PAK that is written for health professionals. |
|||
| What are the ingredients in VIEKIRA PAK?
Ombitasvir, paritaprevir, and ritonavir tablets: Active ingredients: ombitasvir, paritaprevir, and ritonavir Inactive ingredients: copovidone, K value 28, vitamin E polyethylene glycol succinate, propylene glycol monolaurate Type I, sorbitan monolaurate, colloidal silicon dioxide/colloidal anhydrous silica, sodium stearyl fumarate, polyvinyl alcohol, polyethylene glycol 3350/macrogol 3350, talc, titanium dioxide, and red iron oxide. Dasabuvir tablets: Active ingredients: dasabuvir Inactive ingredients: microcrystalline cellulose (D50-100 um), microcrystalline cellulose (D50-50 um), lactose monohydrate, copovidone, croscarmellose sodium, colloidal silicon dioxide/anhydrous colloidal silica, magnesium stearate, polyvinyl alcohol, titanium dioxide, polyethylene glycol 3350/macrogol 3350, talc and iron oxide yellow, iron oxide red and iron oxide black. |
|||
| Manufactured by AbbVie Inc., North Chicago, IL 60064. VIEKIRA PAK and NORVIR are trademarks of AbbVie Inc. For more information, go to www.viekira.com or call 1-844-484-3547. |
|||
| This Medication Guide has been approved by the U.S. Food and Drug Administration. 03-C075 | Revised: December 2019 | ||
NDC 0074-3093-28
Rx only
viekira pak™
ombitasvir, paritaprevir, ritonavir tablets 12.5 mg / 75 mg / 50 mg
Copackaged with
dasabuvir tablets 250 mg
Do not use if seal on top of carton is broken or missing
Keep out of reach of children
Store at or below 30°C (86°F)
See Package Insert for full Prescribing Information
AbbVie Inc.
North Chicago, IL 60064
©2016 AbbVie Inc.
Each tablet contains ombitsavir, paritaprevir, ritonavir 12.5 mg / 75 mg / 50 mg
Each tablet contains 270.3 mg dasabuvir sodium monohydrate equivalent to 250 mg dasabuvir
Each carton contains 28 tablets in 7 wallets for 1 week of treatment.
Each wallet contains 4 tablets:
2 ombitasvir, paritaprevir, ritonavir tablets and 2 dasabuvir tablets.
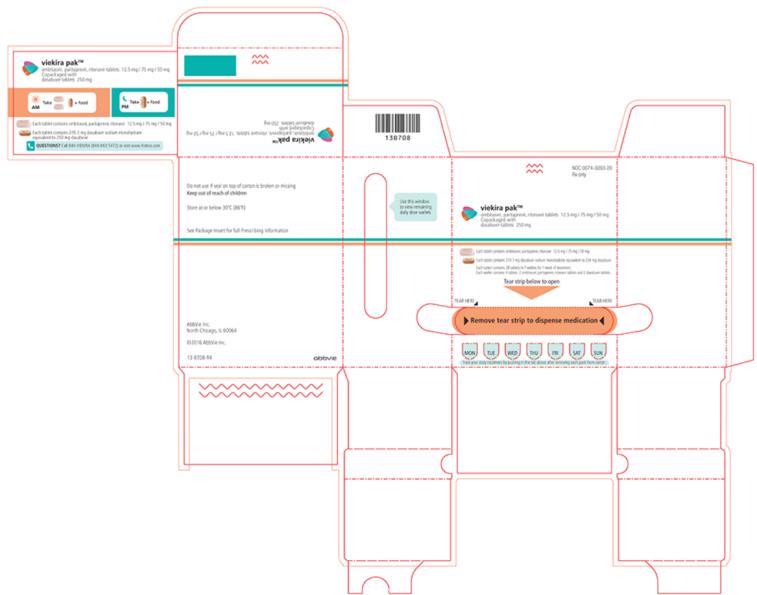
NDC 0074-3093-01
Rx only
viekira pak™
ombitasvir, paritaprevir, ritonavir tablets 12.5 mg / 75 mg / 50 mg
Copackaged with
dasabuvir tablets 250 mg
Keep out of reach of children
Each box contains: 1 wallet of 4 tablets each
2 ombitasvir, paritaprevir, ritonavir tablets and 2 dasabuvir tablets
Store at or below 30°C (86°F)
See Package Insert for full Prescribing Information
AbbVie Inc.
North Chicago, IL 60064
©2014 AbbVie Inc.
Each tablet contains ombitsavir, paritaprevir, ritonavir 12.5 mg / 75 mg / 50 mg
Each tablet contains 270.3 mg dasabuvir sodium monohydrate equivalent to 250 mg dasabuvir
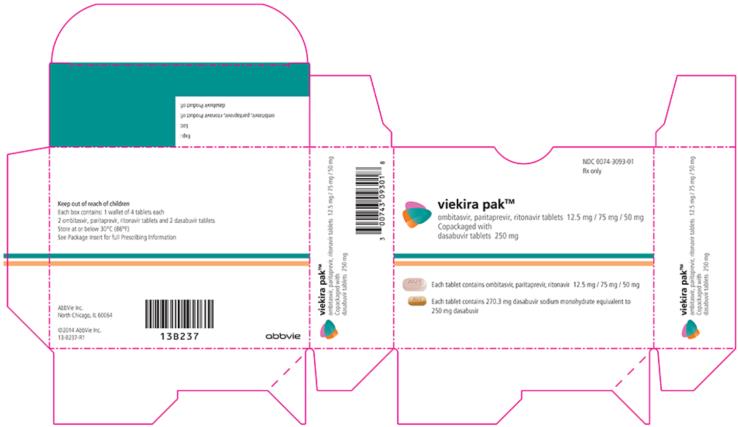
NDC 0074-3093-28
Rx only
viekira pak™
ombitasvir, paritaprevir, ritonavir tablets 12.5 mg / 75 mg / 50 mg
Copackaged with
dasabuvir tablets 250 mg
Each tablet contains ombitsavir, paritaprevir, ritonavir 12.5 mg / 75 mg / 50 mg
Each tablet contains 270.3 mg dasabuvir sodium monohydrate equivalent to 250 mg dasabuvir
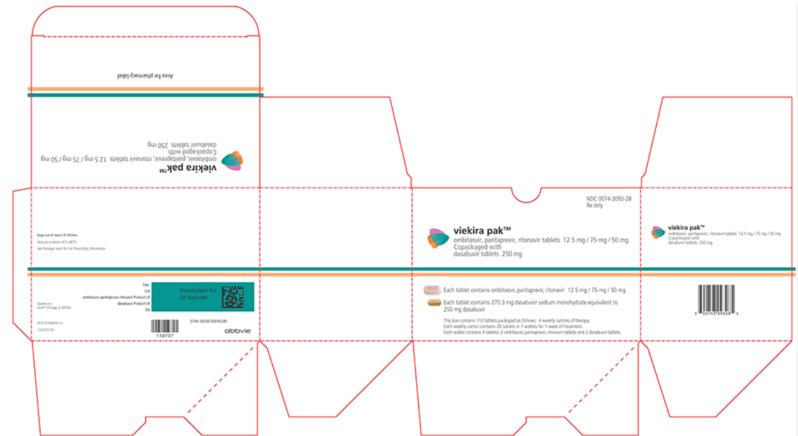
| VIEKIRA PAK
dasabuvir and ombitasvir and paritaprevir and ritonavir kit |
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| Labeler - AbbVie Inc. (078458370) |